research use only
Lumiracoxib COX inhibitor
Cat.No.S2903
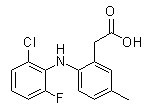
Chemical Structure
Molecular Weight: 293.72
Quality Control
Solubility
|
In vitro |
DMSO
: 59 mg/mL
(200.87 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 293.72 | Formula | C15H13ClFNO2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 220991-20-8 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | COX-189 | Smiles | CC1=CC(=C(C=C1)NC2=C(C=CC=C2Cl)F)CC(=O)O | ||
Mechanism of Action
| Targets/IC50/Ki |
COX-2
(Cell-free assay) 60 nM(Ki)
COX-1
(Cell-free assay) 3 μM(Ki)
|
|---|---|
| In vitro |
Lumumiracoxib has an IC50 of 0.14 μm in COX-2-expressing dermal fibroblasts, but caused no inhibition of COX-1 at concentrations up to 30 μm (HEK 293 cells transfected with human COX-1). In a human whole blood assay, IC50 values for this compound are 0.13 μM for COX-2 and 67 μM for COX-1 (COX-1/COX-2 selectivity ratio 515).
|
| In vivo |
Lumiracoxib is a highly selective COX-2 inhibitor with anti-inflammatory, analgesic and antipyretic activities comparable with diclofenac, the reference NSAID, but with much improved gastrointestinal safety. This compound is rapidly absorbed following oral administration in rats with peak plasma levels being reached between 0.5 and 1 h. Efficacy of this chemical in rat models of hyperalgesia, oedema, pyresis and arthritis is dose-dependent and similar to diclofenac. However, consistent with its low COX-1 inhibitory activity, it at a dose of 100 mg/kg orally causes no ulcers and is significantly less ulcerogenic than diclofenac.
|
References |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT00350155 | Completed | Healthy Volunteers |
Novartis |
May 2006 | Phase 4 |
| NCT00348491 | Completed | Pain |
Novartis Pharmaceuticals|Novartis |
February 2006 | Phase 4 |
| NCT00267176 | Completed | Osteoarthritis|Controlled Hypertension |
Novartis Pharmaceuticals|Novartis |
November 2005 | Phase 4 |
| NCT00170872 | Completed | Osteoarthritis|Rheumatoid Arthritis |
Novartis |
November 2004 | Phase 3 |
| NCT00145301 | Completed | Osteoarthritis |
Novartis |
September 2004 | Phase 3 |
| NCT00475800 | Completed | Osteoarthritis |
Novartis |
January 2004 | Phase 3 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































