research use only
Ambroxol HCl Sodium Channel inhibitor
Cat.No.S3064
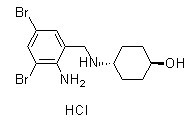
Chemical Structure
Molecular Weight: 414.56
Quality Control
| Related Targets | CFTR CRM1 CD markers AChR Calcium Channel Potassium Channel GABA Receptor TRP Channel ATPase GluR |
|---|---|
| Other Sodium Channel Inhibitors | Camostat Mesilate A-803467 cariporide Veratramine Tolperisone HCl Bulleyaconi cine A Vinpocetine Tenapanor PF-06869206 Sparteine |
Solubility
|
In vitro |
DMSO
: 6 mg/mL
(14.47 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 414.56 | Formula | C13H18Br2N2O.HCl |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 23828-92-4 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | C1CC(CCC1NCC2=C(C(=CC(=C2)Br)Br)N)O.Cl | ||
Mechanism of Action
| Targets/IC50/Ki |
Sodium channel
35.2 μM-22.5 μM
|
|---|---|
| In vitro |
Ambroxol inhibits Na+ channels in sensory neurons. The potency for tonic block of TTX-r channels is relatively high. Ambroxol affects the Na+ current kinetics of TTX-r and TTX-s channels differently. In CNaIIA cells, the compound behaves like a charged local anesthetic: the block is dependent on stimulus number and increases with higher frequencies in a train of depolarizing stimuli. In CNaIIA cells, ambroxol inhibits inactivated channels 5.5-fold more potently than resting channels. The corresponding factor for TTX-r channels is only 3.3. Ambroxol inhibits the release of histamine, leukotrienes and cytokines from human leukocytes and mast cells.
|
| In vivo |
Ambroxol inhibited histamine release by more than 50% from human adenoidal mast cells (1000 microM ambroxol) and skin mast cells (100 microM ambroxol) stimulated by Con A and compound 48/80, respectively. Ambroxol (100 microM) strikingly inhibited anti-IgE induced release of both histamine, LTC4, IL-4 and IL-13 from basophils and reduced both histamine and LTB4 release induced by C5a or Zymosan in monocytes. The drug also reduced LTB4 and superoxide anion production in granulocytes stimulated by zymosan or fMLP.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT04405596 | Not yet recruiting | Lewy Body Disease |
Lawson Health Research Institute |
January 2025 | Phase 1|Phase 2 |
| NCT05778617 | Not yet recruiting | Parkinson Disease |
University College London |
September 2023 | Phase 3 |
| NCT05558878 | Not yet recruiting | Diabetic Neuropathy Peripheral |
Ain Shams University |
October 1 2022 | Not Applicable |
| NCT05287503 | Active not recruiting | Parkinson Disease|GBA Gene Mutation |
Fondazione I.R.C.C.S. Istituto Neurologico Carlo Besta|IRCCS National Neurological Institute C. Mondino Foundation|University of Campania Luigi Vanvitelli |
February 15 2022 | Phase 2 |
| NCT04388969 | Recruiting | Gaucher Disease|Parkinson Disease|GBA Gene Mutation |
Shaare Zedek Medical Center |
May 6 2020 | -- |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































