research use only
Cyclosporine phosphatase inhibitor
Cat.No.S1514
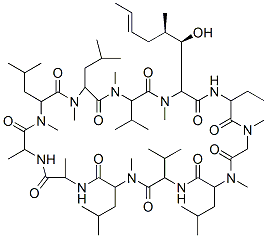
Chemical Structure
Molecular Weight: 1202.61
Quality Control
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(83.15 mM)
Ethanol : 100 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 1202.61 | Formula | C62H111N11O12 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 79217-60-0 | Download SDF | Storage of Stock Solutions |
|
|
Mechanism of Action
| Targets/IC50/Ki |
calcineurin phosphatase
|
|---|---|
| In vitro |
Cyclosporine induces phenotypic changes, including invasiveness of non-transformed cells, by a cell-autonomous mechanism. This compound's treatment of adenocarcinoma cells results in striking morphological alterations, including membrane ruffling and numerous pseudopodial protrusions, increased cell motility, and anchorage-independent (invasive) growth. It has potent immunosuppressive properties, reflecting its ability to block the transcription of cytokine genes in activated T cells. This chemical, through formation of a complex with cyclophilin, inhibits the phosphatase activity of calcineurin, which regulates nuclear translocation and subsequent activation of NFAT transcription factors. It also blocks the activation of JNK and p38 signaling pathways triggered by antigen recognition, making CsA a highly specific inhibitor of T cell activation. Its-mediated inhibition of the biliary excretion of MPAG by the Mrp2 transporter is the mechanism responsible for the interaction between this compound and mycophenolate mofetil (MMF). It inhibits biochemical and morphological differentiation of skeletal muscle cells while having a minimal effect on proliferation.
|
| In vivo |
Cyclosporine enhances tumour growth in immunodeficient SCID-beige mice. This compound inhibits muscle regeneration after induced trauma in mice. It peaks at 1 hour in blood, spleen, and kidney, with higher concentrations in spleen and kidney than in blood.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05702931 | Not yet recruiting | Hyperglycemia|Renal Transplant Complication Primary Non-Function|Diabetes |
Rigshospitalet Denmark|Aarhus University Hospital|Odense University Hospital |
April 1 2024 | Phase 4 |
| NCT05955924 | Recruiting | Non-melanoma Skin Cancer|Carcinoma Squamous Cell|Carcinoma Basal Cell|Keratinocyte Carcinoma |
Women''s College Hospital|Canadian Institutes of Health Research (CIHR)|University Health Network Toronto|NOW Foods |
August 28 2023 | Phase 3 |
| NCT04989686 | Recruiting | Immunosuppression |
Children''s Hospital of Philadelphia|Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) |
June 8 2023 | -- |
| NCT05745701 | Completed | Healthy |
Pfizer |
February 22 2023 | Phase 1 |
| NCT05056779 | Withdrawn | Moderate-to-severe Atopic Dermatitis |
Galderma R&D |
January 2023 | Phase 3 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
What is the difference between S2286 (it) and S1514 (this compound)?
Answer:
It is a mixture of Cyclosporine A, derivatives of Cyclosporine A, and salts of Cyclosporine A. Cyclosporine A is an especially useful component of this compound.






































