research use only
Thiabendazole P450 (e.g. CYP17) inhibitor
Cat.No.S1739
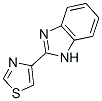
Chemical Structure
Molecular Weight: 201.25
Quality Control
| Related Targets | Dehydrogenase HSP Transferase PDE phosphatase PPAR Vitamin Carbohydrate Metabolism Mitochondrial Metabolism Drug Metabolite |
|---|---|
| Other P450 (e.g. CYP17) Inhibitors | Apigenin Baicalein Naringenin Diosmetin Alizarin Sodium Danshensu Naringin Benzbromarone Orteronel Isosilybin |
Solubility
|
In vitro |
DMSO
: 40 mg/mL
(198.75 mM)
Ethanol : 1 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 201.25 | Formula | C10H7N3S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 148-79-8 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | C1=CC=C2C(=C1)NC(=N2)C3=CSC=N3 | ||
Mechanism of Action
| Targets/IC50/Ki |
CYP1A2
|
|---|---|
| In vitro |
Thiabendazole provokes a strong DNA-damaging activity in the human lymphoblastoid cell line that constitutively expresses human CYP1A1 cDNA, but not in the parental line, indicating that CYP1A1 is chiefly implicated in carbaryl and thiabendazole genotoxicity. This compound provokes a dose- and time-dependent increase in CYP1A1 (EROD activity, protein and mRNA levels) in primary culture of rat hepatocytes. It results in the induction of 7-ethoxyresorufin O-deethylase and 7-pentoxyresorufin O-depentylase activities, CYP1A1, CYP1A2, CYP2B1 and CYP2B1/2 mRNA levels and CYP1A2 and CYP2B1/2 apoprotein levels. This chemical markedly induces GSTP1 mRNA levels, but has only a small effect on GSTT1 mRNA levels. It is rapidly transported by passive diffusion through the human intestinal cells by comparison with the protein-bound residues which are not able to cross the intestinal barrier. It will be firstly metabolized to 5OH-TBZ and subsequently converted to a chemically reactive metabolic intermediate binding to proteins.
|
| In vivo |
Thiabendazole results in nephrosis or hydronephrosis and this organ toxicity may lead to the high dose-dependent mortality in treated mice. This compound results in hormone imbalance and this imbalance may play an important role in the changes of the reproductive or endocrine system in treated mice.
|
References |
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































