research use only
Ketoprofen COX inhibitor
Cat.No.S1645
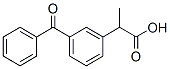
Chemical Structure
Molecular Weight: 254.28
Quality Control
Solubility
|
In vitro |
DMSO
: 51 mg/mL
(200.56 mM)
Ethanol : 51 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 254.28 | Formula | C16H14O3 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 22071-15-4 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | RP-19583 | Smiles | CC(C1=CC(=CC=C1)C(=O)C2=CC=CC=C2)C(=O)O | ||
Mechanism of Action
| Targets/IC50/Ki |
COX-1
COX-2
|
|---|---|
| In vitro |
Ketoprofen combined with UVB irradiation induces the cytotoxicity and suppresses DNA synthesis in HaCaT cells in a concentration-dependent manner. This compound combined with UVB irradiation inhibits the cell growth and induces G2/M cell cycle arrest by modulating the levels of cdc2, cyclin B1, Chk1, Tyr15-phosphorylated cdc2 and p21. It also provokes a striking accumulation of cyclin B1-cdc2-p21 complexes, concomitantly with an increase in the levels of Tyr15-phosphorylated cdc2 and p21 protein. This chemical combined with UVB irradiation accentuates the apoptotic response to UVB radiation in HaCaT cells as evidenced by DAPI staining.
|
| In vivo |
Ketoprofen at 1% level in suitable topical vehicles can effectively inhibit GCF-LTB4 and GCF-PGE2 and positively alter alveolar bone activity in the ligature-induced model of periodontitis in the monkey. This compound (3.63 mg/kg bwt) reduces hoof pain andlameness to a greater extent than the 2.2 mg/kg dose and phenylbutazone. It is more effective than local anesthesia (LA), or caudal epidural anesthesia (EPI) in decreasing cortisol and partially reverses the reduction in average daily gain (ADG) following castration. This chemical (40 and 80 mg/kg diet) reduces the incidence of transitional cell carcinoma of the urinary bladder by >70% from that seen in dietary mice.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT04678076 | Completed | Healthy |
Aziende Chimiche Riunite Angelini Francesco S.p.A|Cross Research S.A. |
July 23 2018 | Phase 1 |
| NCT02796547 | Withdrawn | Episiotomy |
Brugmann University Hospital |
July 14 2016 | Phase 2 |
| NCT02350296 | Completed | Pain Disorders |
Dompé Farmaceutici S.p.A|Cross Research S.A. |
November 26 2014 | Phase 1 |
| NCT01541059 | Completed | Third Molar Extraction |
Centre Hospitalier Universitaire de Nīmes |
June 2012 | Phase 4 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































