
- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- DNA Damage/DNA Repair
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleckchem.com to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly DYKDDDDK Tag Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
SN-38
Synonyms: NK012
Home DNA Damage/DNA Repair ADC Cytotoxin inhibitor - Topoisomerase inhibitor - Autophagy activator SN-38
For research use only.
SN-38 (NK012) is an active metabolite of CPT-11, inhibits DNA topoisomerase I, DNA synthesis and causes frequent DNA single-strand breaks. SN-38 induces autophagy.
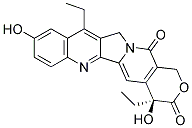
SN-38 Chemical Structure
CAS No. 86639-52-3
Purity & Quality Control
Batch:
Purity:
99.96%
99.96
SN-38 Related Products
| Related Products | Camptothecin (CPT) Triptolide (+)-Bicuculline Rutin Artemisinin BHQ Pinocembrin Lappaconite HBr Psoralen Harmine hydrochloride Lycorine hydrochloride Tetrahydropalmatine hydrochloride Luteoloside | Click to Expand |
|---|---|---|
| Related Compound Libraries | FDA-approved Drug Library Natural Product Library Apoptosis Compound Library DNA Damage/DNA Repair compound Library Cell Cycle compound library | Click to Expand |
Cell Data
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| human DU145 prostate cell line | Proliferation assay | Antiproliferative activity against human DU145 prostate cell line, IC50=13 nM | 10498216 | |||
| human MCF-7 breast cell line | Proliferation assay | Antiproliferative activity against human MCF-7 breast cell line, IC50=0.37 μM | 10498216 | |||
| human SKOV-3 ovarian cell line | Proliferation assay | Antiproliferative activity against human SKOV-3 ovarian cell line, IC50=0.72 μM | 10498216 | |||
| human breast cancer cell line (SK-BR-3) | Cytotoxic assay | In Vitro cytotoxicity against human breast cancer cell line (SK-BR-3), IC50=4 nM | 11334569 | |||
| human lung cancer cell line (H128) | Cytotoxic assay | In Vitro cytotoxicity against human lung cancer cell line (H128), IC50=6 nM | 11334569 | |||
| human stomach cancer cell line (MKN45) | Cytotoxic assay | In Vitro cytotoxicity against human stomach cancer cell line (MKN45), IC50=8 nM | 11334569 | |||
| human ovarian cancer cell line (SK-OV-3) | Cytotoxic assay | In Vitro cytotoxicity against human ovarian cancer cell line (SK-OV-3), IC50=11 nM | 11334569 | |||
| human colon cancer cell line (WiDr) | Cytotoxic assay | In Vitro cytotoxicity against human colon cancer cell line (WiDr), IC50=14 nM | 11334569 | |||
| human lung cancer cell line (A549) | Cytotoxic assay | In Vitro cytotoxicity against human lung cancer cell line (A549) | 11334569 | |||
| NSCLC-H460 | Cytotoxic assay | Cytotoxicity against human non-small-cell lung carcinoma cell line H460 (NSCLC-H460), IC50=80 nM | 11563925 | |||
| PC-3 carcinoma cell line | Growth inhibition assay | Concentration required to inhibit growth of human prostate PC-3 carcinoma cell line, IC50=35.6 nM | 15454230 | |||
| human HCT116 colon cancer cell line | Function assay | Inhibitory concentration against human HCT116 colon cancer cell line, IC50=7 nM | 15808456 | |||
| human Bel-7402 liver cancer cell line | Function assay | Inhibitory concentration against human Bel-7402 liver cancer cell line, IC50=3.19 μM | 15808456 | |||
| human tumor HL60 cell-line | Function assay | 3 days | In-vitro inhibitory concentration for human tumor HL60 cell-line was determined using SRB assay after 3 days of incubation, IC50=19 nM | 15913996 | ||
| human H460 cell | Growth inhibition assay | Inhibition of human H460 cell growth, IC50=80 nM | 16913706 | |||
| MDA-MB-435 | Function assay | Inhibition against MDA-MB-435 S human breast cancer cells in the absence of albumin, IC50 = 0.005 μM. | 11052802 | |||
| MDA-MB-435 | Function assay | Inhibition against MDA-MB-435 S human breast cancer cells in the presence of 30 mg/mL HSA, IC50 = 0.05 μM. | 11052802 | |||
| PC-6/SN2-5H | Function assay | TP_TRANSPORTER: uptake in membrane vesicle from PC-6/SN2-5H cells, Km = 4 μM. | 11688982 | |||
| HT-29 | Function assay | In vitro inhibitory concentration required against HT-29 human colon cancer cells, IC50 = 0.0273 μM. | 12617927 | |||
| SaoS2 | Function assay | TP_TRANSPORTER: drug resistance(Imatinib mesylate) in BCRP-expressing SaoS2 cells, IC50 = 0.176 μM. | 15059881 | |||
| DU145 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human DU145 cells after 96 hrs, IC50 = 0.004 μM. | 18276141 | ||
| MIA PaCa | Antiproliferative assay | 96 hrs | Antiproliferative activity against human MIA PaCa cells after 96 hrs, IC50 = 0.006 μM. | 18276141 | ||
| HT29 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human HT29 cells after 96 hrs, IC50 = 0.012 μM. | 18276141 | ||
| H460 | Antiproliferative assay | 1 hr | Antiproliferative activity against human H460 cells after 1 hr of drug exposure measured after 72 hrs, IC50 = 0.22 μM. | 18424133 | ||
| HT29 | Antiproliferative assay | 1 hr | Antiproliferative activity against human HT29 cells after 1 hr of drug exposure measured after 72 hrs, IC50 = 0.67 μM. | 18424133 | ||
| HT29 | Antiproliferative assay | 1 hr | Antiproliferative activity against BCRP overexpressing mitoxantrone-resistant human HT29 cells after 1 hr of drug exposure measured after 72 hrs, IC50 = 11.5 μM. | 18424133 | ||
| H460 | Cytotoxicity assay | Cytotoxicity against human H460 cells, IC50 = 0.22 μM. | 18434153 | |||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells by SRB method, IC50 = 0.08 μM. | 18986807 | |||
| HT-29 | Cytotoxicity assay | Cytotoxicity against human HT-29 cells by SRB method, IC50 = 0.12 μM. | 18986807 | |||
| PC6 | Antiproliferative assay | 6 days | Antiproliferative activity against human PC6 cells carrying pRC after 6 days, IC50 = 0.00043 μM. | 19254843 | ||
| HCT116 | Antiproliferative assay | 3 days | Antiproliferative activity against human HCT116 cells after 3 days, IC50 = 0.00055 μM. | 19254843 | ||
| QG56 | Antiproliferative assay | 3 days | Antiproliferative activity against human QG56 cells after 3 days, IC50 = 0.0028 μM. | 19254843 | ||
| NCI-H460 | Antiproliferative assay | 3 days | Antiproliferative activity against human NCI-H460 cells after 3 days, IC50 = 0.0033 μM. | 19254843 | ||
| BCRP | Antiproliferative assay | 6 days | Antiproliferative activity against human PC6 expressing BCRP cells after 6 days, IC50 = 0.0051 μM. | 19254843 | ||
| KB3-1 | Cytotoxicity assay | 4 days | Cytotoxicity against human KB3-1 cells after 4 days by MTT method, IC50 = 0.004 μM. | 19303306 | ||
| KBV1 | Cytotoxicity assay | 4 days | Cytotoxicity against MDR1 overexpressing human KBV1 cells after 4 days by MTT method, IC50 = 0.046 μM. | 19303306 | ||
| KBH5.0 | Cytotoxicity assay | 4 days | Cytotoxicity against BCRP overexpressing human KBH5.0 cells after 4 days by MTT method, IC50 = 0.3 μM. | 19303306 | ||
| NCI-H460 | Antiproliferative assay | 1 hr | Antiproliferative activity against human NCI-H460 cells after short term exposure for 1 hr measured after 72 hrs in drug-free medium, IC50 = 0.13 μM. | 19530720 | ||
| A549/ATCC | Antitumor assay | Antitumor activity against human A549/ATCC cells by SRB method, IC50 = 0.088 μM. | 19541483 | |||
| HT-29 | Antitumor assay | Antitumor activity against human HT-29 cells by SRB method, IC50 = 0.17 μM. | 19541483 | |||
| gastric cancer cells | Growth inhibition assay | 72 hrs | Growth inhibition of human gastric cancer cells after 72 hrs by sulforhodamine B assay, GI50 = 0.066 μM. | 20884089 | ||
| lung cancer cells | Growth inhibition assay | 72 hrs | Growth inhibition of human lung cancer cells after 72 hrs by sulforhodamine B assay, GI50 = 0.066 μM. | 20884089 | ||
| colon cancer cells | Growth inhibition assay | 72 hrs | Growth inhibition of human colon cancer cells after 72 hrs by sulforhodamine B assay, GI50 = 0.066 μM. | 20884089 | ||
| HT-29 | Cytotoxicity assay | Cytotoxicity against human HT-29 cells, IC50 = 0.0059 μM. | 21470864 | |||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells, IC50 = 0.047 μM. | 21470864 | |||
| PC3 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human PC3 cells expressing alpha5beta3 integrin assessed as cell survival after 72 hrs by SRB assay, IC50 = 0.0026 μM. | 22959246 | ||
| A2780 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A2780 cells overexpressing alpha5beta3 integrin assessed as cell survival after 72 hrs by SRB assay, IC50 = 0.009 μM. | 22959246 | ||
| DU145 | Cytotoxicity assay | Cytotoxicity against human DU145 cells, IC50 = 0.03 μM. | 23578545 | |||
| HT-29 | Cytotoxicity assay | Cytotoxicity against human HT-29 cells, IC50 = 0.03 μM. | 23578545 | |||
| L1210 | Cytotoxicity assay | Cytotoxicity against mouse L1210 cells, IC50 = 0.04 μM. | 23578545 | |||
| DU145 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human DU145 cells assessed as inhibition of cell viability after 72 hrs by MTS assay, IC50 = 0.11 μM. | 23622981 | ||
| DU145 | Function assay | 0.1 to 0.5 uM | 24 hrs | Inhibition of HDAC in human DU145 cells assessed as upregulation of p21waf1 expression at 0.1 to 0.5 uM after 24 hrs by Western blot analysis | 23622981 | |
| A549 | Cytotoxicity assay | 3 days | Cytotoxicity against human A549 cells assessed as growth inhibition after 3 days by SRB assay, IC50 = 0.00272 μM. | 24529870 | ||
| HCT116 | Cytotoxicity assay | 3 days | Cytotoxicity against human HCT116 cells assessed as growth inhibition after 3 days by SRB assay, IC50 = 0.031 μM. | 24529870 | ||
| HT-29 | Cytotoxicity assay | 3 days | Cytotoxicity against human HT-29 cells assessed as growth inhibition after 3 days by SRB assay, IC50 = 0.10667 μM. | 24529870 | ||
| HCT116 | Cytotoxicity assay | Cytotoxicity against human HCT116 cells, IC50 = 0.166 μM. | 24529870 | |||
| MDA-MB-231 | Cytotoxicity assay | Cytotoxicity against human MDA-MB-231 cells, IC50 = 0.176 μM. | 24529870 | |||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells, IC50 = 0.259 μM. | 24529870 | |||
| HEK293 | Cytotoxicity assay | 72 hrs | Intrinsic cytotoxicity against HEK293 cells assessed as reduction in cell viability after 72 hrs by MTT assay, IG50 = 0.005 μM. | 25272055 | ||
| HCT116 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT116 cells after 72 hrs by sulforhodamine B assay, IC50 = 0.00428 μM. | 25835359 | ||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells after 72 hrs by sulforhodamine B assay, IC50 = 0.01854 μM. | 25835359 | ||
| HCT15 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT15 cells assessed as growth inhibition after 72 hrs by SRB assay, IC50 = 0.008 μM. | 27060760 | ||
| U251 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human U251 cells assessed as growth inhibition after 72 hrs by SRB assay, IC50 = 0.021 μM. | 27060760 | ||
| U87MG | Cytotoxicity assay | 72 hrs | Cytotoxicity against human U87MG cells assessed as growth inhibition after 72 hrs by SRB assay, IC50 = 0.03 μM. | 27060760 | ||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells assessed as growth inhibition after 72 hrs by SRB assay, IC50 = 0.054 μM. | 27060760 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells assessed as growth inhibition after 72 hrs by SRB assay, IC50 = 0.515 μM. | 27060760 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-231 cells assessed as growth inhibition after 72 hrs by SRB assay, IC50 = 1.97 μM. | 27060760 | ||
| NCI-H460 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human NCI-H460 cells pretreated for 24 hrs under normoxic condition followed by compound washout measured after 72 hrs by MTT assay, IC50 = 0.05 μM. | 28350997 | ||
| NCI-H460 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human NCI-H460 cells pretreated for 24 hrs under hypoxic condition followed by compound washout measured after 72 hrs by MTT assay, IC50 = 0.06 μM. | 28350997 | ||
| HT-29 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HT-29 cells pretreated for 24 hrs under normoxic condition followed by compound washout measured after 72 hrs by MTT assay, IC50 = 2.74 μM. | 28350997 | ||
| HT-29 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HT-29 cells pretreated for 24 hrs under hypoxic condition followed by compound washout measured after 72 hrs by MTT assay, IC50 = 3.71 μM. | 28350997 | ||
| NCI-H460 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human NCI-H460 cells assessed as growth reduction under hypoxic condition after 72 hrs by MTT assay, IC50 = 0.047 μM. | 28740613 | ||
| NCI-H460 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human NCI-H460 cells assessed as growth reduction under normoxic condition after 72 hrs by MTT assay, IC50 = 0.05 μM. | 28740613 | ||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells assessed as growth reduction under normoxic condition after 72 hrs by MTT assay, IC50 = 0.096 μM. | 28740613 | ||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells assessed as growth reduction under hypoxic condition after 72 hrs by MTT assay, IC50 = 0.119 μM. | 28740613 | ||
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for TC32 cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-12 cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for MG 63 (6-TG R) cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for BT-12 cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for NB-EBc1 cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for SK-N-SH cells | 29435139 | |||
| Rh30 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh30 cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for A673 cells) | 29435139 | |||
| Rh30 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Rh30 cells | 29435139 | |||
| U-2 OS | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for U-2 OS cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Rh41 cells | 29435139 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | |||
| HCT116 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT116 cells after 72 hrs by SRB assay, IC50 = 0.00078 μM. | 30115492 | ||
| A549 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human A549 cells after 72 hrs by SRB assay, IC50 = 0.00162 μM. | 30115492 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells after 72 hrs by sulforhodamine B assay, IC50 = 0.0001 μM. | 30169038 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells after 72 hrs by sulforhodamine B assay, IC50 = 0.2 μM. | 30169038 | ||
| Click to View More Cell Line Experimental Data | ||||||
Biological Activity
| Description | SN-38 (NK012) is an active metabolite of CPT-11, inhibits DNA topoisomerase I, DNA synthesis and causes frequent DNA single-strand breaks. SN-38 induces autophagy. | |
|---|---|---|
| Targets |
|
| In vitro | ||||
| In vitro | SN-38, a biological active metabolite of CPT-11. SN-38 causes the strongest inhibition of DNA topoisomerase I, followed by CPT and then CPT-11. CPT-11 dose dependently shifts the position of relaxed DNA in the direction of nicked DNA, but SN-38 and CPT shows no effect on the position of relaxed DNA. SN-38 dose-dependently and time-dependently inhibit DNA synthesis. Respective IC50 values of SN-38, in DNA synthesis is 0.077 μM. The inhibitory effect of SN-38 on RNA synthesis is less than that on DNA synthesis and it does not inhibit protein synthesis. SN-38 caused frequent DNA single-strand breaks in P388 cells. [1] |
|||
|---|---|---|---|---|
| Kinase Assay | Topoisomerase I Assay | |||
| One unit (the minimum amount for full relaxation of 0.5 μg SV40 DNA under the conditions of this study) of topoisomerase I, 0.5 μL of the test compounds, and 0.5μg SV40 DNA are added sequentially to the reaction buffer, which is composed of 25 mM Tris-HCl (pH 7.5), 50 mM KC1, 5 mM MgCl2, 0.25 mM EDTA disodium salt, 0.25 mM dithiothreitol, 15μg /mL bovine serum albumin, and 5% glycerol. Then, the reaction mixture (50 μL) is incubated for 10 min at 37 °C, and the reaction is terminated by treatment with 7.5 μL of a solution consisting of 1% sodium dodecyl sulfate, 20 mM EDTA disodium salt, and 0.5 mg/mL proteinase K for an additional 30 min at 37°C. The samples are mixed with 5 μL of the loading buffer containing 10 mM Na2HPO4, 31.3% sucrose, and 0.3% bromophenol blue. Relaxed (form Ir) DNA is separated from supercoiled (form I) and nicked (form II) DNA by electrophoresis on 0.8% agarose gel at 50 mA and 20 V for 17 h in the presence of 2 μg/mL CHQ, 10 mM EDTA, 30 mM NaH2PO4, and 36 Mm Tris-HCl (pH 7.8). After electrophoresis, the gel is stained with 0.05% ethidium bromide and photographed with UV light (302 nm). The amount of DNA is quantified using a densitometer. | ||||
| Cell Research | Cell lines | A-172, U-87, and LA-567 | ||
| Concentrations | 0 -1000 nM | |||
| Incubation Time | 48 h | |||
| Method | MTT assay |
|||
| Experimental Result Images | Methods | Biomarkers | Images | PMID |
| Western blot | p-Chk1 / Chk1 / Topo I |
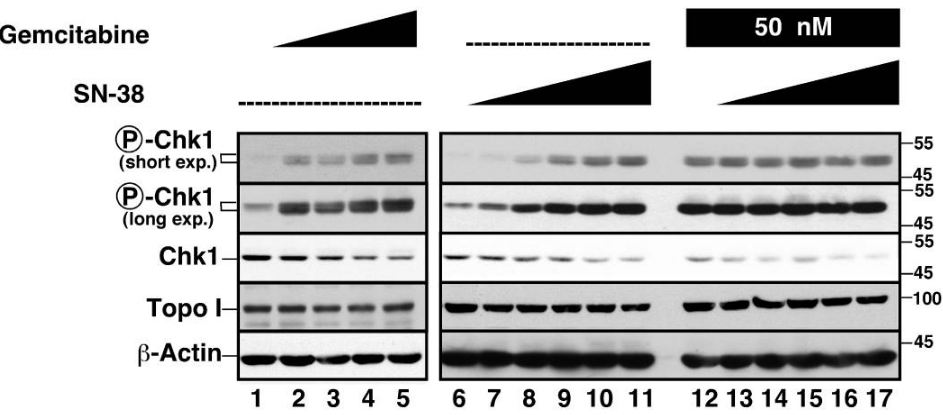
|
18509065 | |
| Growth inhibition assay | Cell viability |
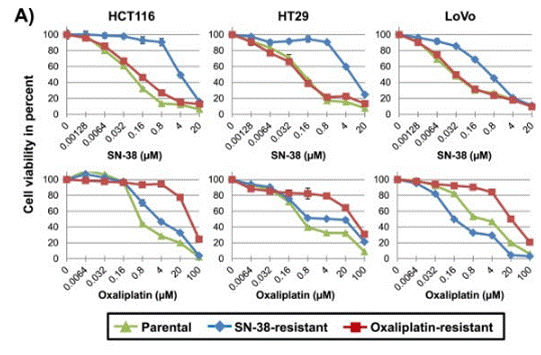
|
25759163 | |
| In Vivo | ||
| In vivo | After oral dosing, peak SN-38 concentrations occurrs within 1 h, and the The percent unbound SN-38 lactone in murine plasma at 1000 ng/mL is 3.4 +/- 0.67%, whereas at 100 ng/mL the percent unbound is 1.18 +/- 0.14%. SN-38 lactone AUCs in micebearing human neuroblastoma xenografts are greater than in nontumor-bearing animals. [2] |
|
|---|---|---|
| Animal Research | Animal Models | Male Sprague-Dawley rats |
| Dosages | 10 mg/kg | |
| Administration | i.v. | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06143774 | Recruiting | Advanced Solid Tumor |
TaiRx Inc. |
October 31 2023 | Phase 1 |
| NCT05101096 | Recruiting | Advanced Solid Tumor|Metastatic Triple-Negative Breast Cancer|HR+/HER2- Metastatic Breast Cancer|Metastatic Urothelial Cancer |
Gilead Sciences |
October 20 2021 | Phase 1|Phase 2 |
| NCT05119907 | Recruiting | Solid Tumor |
Gilead Sciences |
October 12 2021 | Phase 2 |
| NCT04866641 | Recruiting | Advanced Solid Tumor |
Taivex Therapeutics Corporation |
June 24 2021 | Phase 1 |
| NCT04617522 | Recruiting | Advanced or Metastatic Solid Tumor|Liver Failure |
Gilead Sciences |
April 6 2021 | Phase 1 |
|
Chemical Information & Solubility
| Molecular Weight | 392.4 | Formula | C22H20N2O5 |
| CAS No. | 86639-52-3 | SDF | Download SN-38 SDF |
| Smiles | CCC1=C2CN3C(=CC4=C(C3=O)COC(=O)C4(CC)O)C2=NC5=C1C=C(C=C5)O | ||
| Storage (From the date of receipt) | |||
|
In vitro |
DMSO : 19 mg/mL ( (48.41 mM) Moisture-absorbing DMSO reduces solubility. Please use fresh DMSO.) Water : Insoluble Ethanol : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
|||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field





































