- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- DNA Damage/DNA Repair
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleckchem.com to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly DYKDDDDK Tag Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
research use only
Dolutegravir (GSK1349572) Integrase inhibitor
Dolutegravir (GSK1349572) is a two-metal-binding HIV integrase inhibitor with IC50 of 2.7 nM in a cell-free assay, and it shows modest activity against raltegravir-resistant signature mutants Y143R, Q148K, N155H, and G140S/Q148H.
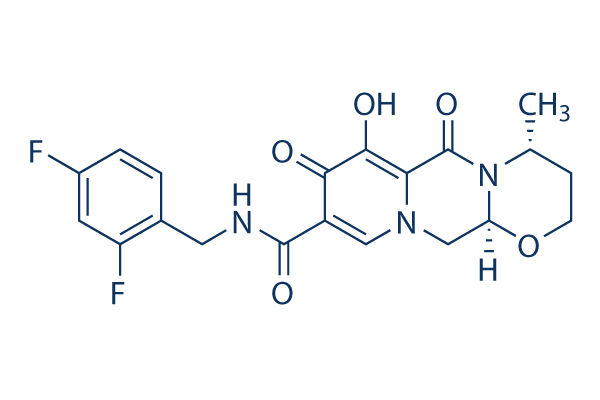
Chemical Structure
Molecular Weight: 419.38
Purity & Quality Control
Batch:
Purity:
99.76%
99.76
Related Products
| Related Targets | HIV-1 Integrase HIV-2 Integrase HIV Integrase | Click to Expand |
|---|---|---|
| Related Products | MK-2048 BMS-707035 | Click to Expand |
| Related Compound Libraries | Anti-infection Compound Library Antibiotics compound Library Antiviral Compound Library Anti-parasitic Compound Library Gut Microbial Metabolite Library | Click to Expand |
Signaling Pathway
Cell Culture and Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| MDCK2 cells | Function assay | Inhibition of human OCT2 expressed in MDCK2 cells using [14C]metformin as substrate by liquid scintillation counting analysis | 23132334 | |||
| HOS | Antiviral assay | 3 hrs | Antiviral activity against HIV-1 harboring wild type integrase infected in human HOS cells pretreated with compound for 3 hrs by single-round HIV-1 infectivity assay, EC50 = 0.0016 μM. | 24901667 | ||
| HEK293T | Antiviral assay | 2 days | Antiviral activity against pseudo Human immunodeficiency virus infected in HEK293T cells after 2 days, IC50 = 0.0017 μM. | 23845180 | ||
| MT4 | Antiviral assay | 4 to 5 days | Antiviral activity against Human immunodeficiency virus 1 3B infected in human MT4 cells after 4 to 5 days by bioluminescence assay, IC50 = 0.002 μM. | 23845180 | ||
| HOS | Antiviral assay | 3 hrs | Antiviral activity against raltegravir-resistant HIV-1 harboring integrase N155H mutant infected in human HOS cells pretreated with compound for 3 hrs by single-round HIV-1 infectivity assay, EC50 = 0.0036 μM. | 24901667 | ||
| HOS | Antiviral assay | 3 hrs | Antiviral activity against raltegravir-resistant HIV-1 harboring integrase Y143R mutant infected in human HOS cells pretreated with compound for 3 hrs by single-round HIV-1 infectivity assay, EC50 = 0.0043 μM. | 24901667 | ||
| HOS | Antiviral assay | 3 hrs | Antiviral activity against raltegravir-resistant HIV-1 harboring integrase G140S/Q148H double mutant infected in human HOS cells pretreated with compound for 3 hrs by single-round HIV-1 infectivity assay, EC50 = 0.0058 μM. | 24901667 | ||
| HOS | Antiviral assay | 3 hrs | Antiviral activity against INSTI-resistant HIV-1 harboring integrase R263K mutant infected in human HOS cells pretreated with compound for 3 hrs by single-round HIV-1 infectivity assay, EC50 = 0.011 μM. | 24901667 | ||
| HOS | Antiviral assay | 3 hrs | Antiviral activity against INSTI-resistant HIV-1 harboring integrase G118R mutant infected in human HOS cells pretreated with compound for 3 hrs by single-round HIV-1 infectivity assay, EC50 = 0.013 μM. | 24901667 | ||
| P4R5 MAGI | Antiviral assay | 24 hrs | Antiviral activity against HIV1 infected in CD4/CXCR4/CCR5 expressing human P4R5 MAGI cells preincubated with cells for 24 hrs followed by viral infection measured after 48 hrs by beta-galactosidase reporter gene assay, EC50 = 0.02 μM. | 30031976 | ||
| P4R5 | Antiviral assay | 24 hrs | Antiviral activity against HIV1 infected in CD4/CXCR4/CCR5 expressing human P4R5 cells assessed as inhibition of viral replication preincubated with cells for 24 hrs followed by viral infection measured after 48 hrs by beta-galactosidase reporter gene ass, EC50 = 0.02 μM. | 28525279 | ||
| HEK293T | Antiviral assay | 2 days | Antiviral activity against pseudo Human immunodeficiency virus infected in HEK293T cells after 2 days in presence of human serum albumin, IC50 = 0.022 μM. | 23845180 | ||
| MDCK2 | Function assay | Inhibition of human OCT2 expressed in MDCK2 cells using [14C]metformin as substrate by liquid scintillation counting analysis, IC50 = 1.9 μM. | 23132334 | |||
| Vero E6 | Antiviral assay | 2 days | Antiviral efficacy against SARS-CoV-2 (strain BavPat1) in Vero E6 cells assessed by inhibition of viral RNA replication measured by RT-PCR after 2 days, EC50 = 22.04 μM. | ChEMBL | ||
| Vero E6 | Antiviral assay | 2 days | Antiviral efficacy against SARS-CoV-2 (strain BavPat1) in Vero E6 cells assessed by inhibition of viral RNA replication measured by RT-PCR after 2 days, EC90 = 42.81 μM. | ChEMBL | ||
| Click to View More Cell Line Experimental Data | ||||||
Mechanism of Action
| Features | A next-generation and two-metal-binding HIV integrase strand transfer inhibitor. | ||
|---|---|---|---|
| Targets |
|
In vitro |
||||
| In vitro | Dolutegravir (GSK1349572) shows the potent inhibitory effect on nine clinical isolates from integrase inhibitor-naive HIV-2-infected patients with EC50 ranging from 0.2 nM -1.4 nM. [1] In vitro, it inhibits recombinant HIV-1 integrase-catalyzed strand transfer with IC50 of 2.7 nM. Furthermore, this compound potently inhibits HIV replication in cells such as peripheral blood mononuclear cells (PBMCs), MT-4 cells and CIP4 cells infected with a self-inactivating PHIV lentiviral vector with EC50 of 0,51 nM, 0.71 nM and 2.2 nM, respectively. [2] In vitro, it exhibits potent activity against five different nonnucleoside reverse transcription inhibitor--resistant or nucleoside reverse transcription inhibitor--resistant viruses with EC50 ranging from 1.3 nM -2.1 nM. Similarly to that against wild-type virus, this compound shows equivalent activity against two protease inhibitor-resistant viruses with EC50 of 0.36 nM and 0.37 nM, respectively. [2] |
|||
|---|---|---|---|---|
| Kinase Assay | In vitro strand transfer assay | |||
| The inhibitory potencies of this compound and other INIs are measured in a strand transfer assay using recombinant HIV integrase. A complex of integrase and biotinylated preprocessed donor DNA-streptavidin-coated scintillation proximity assay (SPA) beads is formed by incubating 2 μM purified recombinant integrase with 0.66 μM biotinylated donor DNA-4 mg/mL streptavidin-coated SPA beads in 25 mM sodium morpholinepropanesulfonic acid (MOPS) (pH 7.2), 23 mM NaCl, and 10 mM MgCl2 for 5 minutes at 37 °C. These beads are spun down and preincubated with diluted INIs for 60 minutes at 37 °C. Then a 3H-labeled target DNA substrate is added to give a final concentration of 7 nM substrate, and the strand transfer reaction mixture is incubated at 37 °C for 25 to 45 minutes, which allows for a linear increase in the strand transfer of donor DNA to radiolabeled target DNA. The signal is read using a Wallac MicroBeta scintillation plate reader. | ||||
| Cell Research | Cell lines | MT-4 | ||
| Concentrations | 0 to 10 μM | |||
| Incubation Time | 4 days or 5 days | |||
| Method | Dolutegravir (GSK1349572) is evaluated using MT-4 cells growing exponentially at a density of 500000 or 600000 /mL, which are infected with HIV-1 strain IIIB at a viral multiplicity of infection of 0.001 or a 50% tissue culture infective dose of 4 to 10. The cells are then aliquoted to 96-well plates in the presence of varying concentrations of this compound. After incubation for 4 or 5 days, antiviral activity is determined by a cell viability assay that either measured bioluminescence with a CellTiter-Glo luminescent reagent or measured absorbance at 560 and 690 nm using the yellow tetrazolium MTT reagent [3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyltetrazolium bromide]. |
|||
| Experimental Result Images | Methods | Biomarkers | Images | PMID |
| Dose-response infectivity curves | NL4.3IN(WT) / NL4.3IN(S230R) virus |
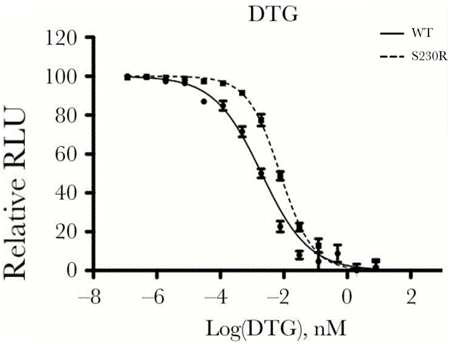
|
29617824 | |
In Vivo |
||
| In vivo | Dolutegravir (GSK1349572), also known as DTG, is a first-line antiretroviral drug (ARV) used in combination therapy for HIV-1. It inhibits MMP activity and has the potential to affect prenatal and postnatal neurodevelopment. |
|
|---|---|---|
| Animal Research | Animal Models | C3H/HeJ mice |
| Dosages | 50 mg/kg | |
| Administration | o.g. | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06281834 | Not yet recruiting | Pediatric HIV Infection|Latent Tuberculosis |
Brigham and Women''s Hospital|APIN Public Health Initiatives|University of Cape Town |
May 2024 | Phase 1 |
| NCT05122026 | Recruiting | HIV Seropositivity|Pregnancy|Tuberculosis Infection |
The Aurum Institute NPC|Johns Hopkins University|Weill Medical College of Cornell University|University of Washington |
January 17 2024 | Phase 1|Phase 2 |
| NCT05069688 | Recruiting | Pediatric HIV Infection|Tuberculosis Infection |
Brigham and Women''s Hospital|APIN Public Health Initiatives|University of Cape Town |
July 7 2023 | Phase 1 |
| NCT05122767 | Recruiting | Tuberculosis|HIV |
The Aurum Institute NPC|Johns Hopkins University |
May 24 2023 | Phase 1|Phase 2 |
References |
|
Chemical Information
| Molecular Weight | 419.38 | Formula | C20H19F2N3O5 |
| CAS No. | 1051375-16-6 | SDF | Download SDF |
| Synonyms | GSK1349572,S/GSK1349572 | ||
| Smiles | CC1CCOC2N1C(=O)C3=C(C(=O)C(=CN3C2)C(=O)NCC4=C(C=C(C=C4)F)F)O | ||
Storage and Stability
| Storage (From the date of receipt) | |||
|
In vitro |
DMSO : 83 mg/mL ( (197.91 mM) Moisture-absorbing DMSO reduces solubility. Please use fresh DMSO.) Water : Insoluble Ethanol : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
|||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field