- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at [email protected] to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly FLAG Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
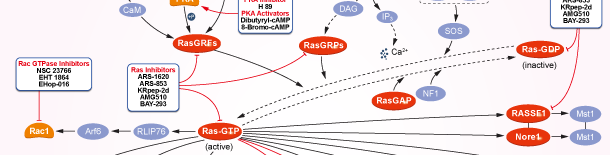
Ras
Ras Products
- All (46)
- Ras Inhibitors (43)
- New Ras Products
| Catalog No. | Product Name | Information | Product Use Citations | Product Validations |
|---|---|---|---|---|
| S1314 | Zoledronic acid (Zoledronate) | Zoledronic acid (Zoledronate), a potent osteoclast inhibitor, induces apoptosis in osteoclasts by inhibiting enzymes of the mevalonate pathway and preventing the isoprenylation of small GTP-binding proteins such as Ras and Rho. Zoledronic acid (ZA) also induces autophagy. |
|
|
| S8830 | Sotorasib (AMG510) | Sotorasib (AMG510) is a potent KRAS G12C covalent inhibitor with potential antineoplastic activity.This AMG510 is a chiral compound. | ||
| S2797 | Lonafarnib (SCH66336) | Lonafarnib (SCH66336) is an orally bioavailable FPTase inhibitor for H-ras, K-ras-4B and N-ras with IC50 of 1.9 nM, 5.2 nM and 2.8 nM in cell-free assays, respectively. Phase 3. |

|
|
| S8707 | ARS-1620 | ARS-1620 is a potent, orally bioavailable covalent inhibitor of KRASG12C and could achieve rapid and sustained in vivo target occupancy to induce tumor regression. | ||
| S5244 | Zoledronic acid monohydrate | Zoledronic acid (Zoledronate, CGP-4244) monohydrate, a nitrogen-containing bisphosphonate, is a potent osteoclast inhibitor which induces apoptosis in osteoclasts by inhibiting enzymes of the mevalonate pathway and preventing the isoprenylation of small GTP-binding proteins such as Ras and Rho. | ||
| S7684 | Salirasib | Salirasib (Farnesylthiosalicylic acid, FTS) is a potent competitive prenylated protein methyltransferase (PPMTase) inhibitor with Ki of 2.6 μM, which inhibits Ras methylation. Salirasib exerts antitumor effects and induces autophagy. Phase 2. | ||
| S8884 | Adagrasib (MRTX849) | Adagrasib (MRTX849) is a potent, selective, and covalent KRASG12C inhibitor that exhibits favorable drug-like properties, selectively modifies mutant cysteine 12 in GDP-bound KRASG12C and inhibits KRAS-dependent signaling. | ||
| S7224 | Deltarasin | Deltarasin is a small molecular inhibitor of KRAS-PDEδ interaction with Kd of 38 nM for binding to purified PDEδ. | ||
| S8156 | ARS-853 | ARS-853 is a selective, covalent KRAS(G12C) inhibitor that inhibits mutant KRAS-driven signaling by binding to the GDP-bound oncoprotein and preventing activation. ARS-853 also induces apoptosis. | ||
| S8916 | BI-3406 | BI-3406 (compound I-13) is a potent, selective and orally active inhibitor of the interaction between KRAS and Son of Sevenless 1 (SOS1) with IC50 of 5 nM. BI-3406 reduces the formation of GTP-loaded KRAS, and inhibits MAPK pathway signaling. BI-3406 exhibits anti-tumor activities. | ||
| E1051 | MRTX1133 | MRTX1133 is a highly selective inhibitor of mutant KRAS G12D and can reversibly binds to the activated and inactivated KRAS G12D mutants and inhibit their activity. The specificity of MRTX1133 to KRAS G12D is more than 1000 times that of wild-type KRAS. |
||
| S8826 | BAY-293 | BAY-293 selectively inhibits the KRAS-SOS1 interaction with an IC50 of 21 nM. | ||
| S7331 | K-Ras(G12C) inhibitor 12 | K-Ras(G12C) inhibitor 12 is an allosteric inhibitor of oncogenic K-Ras(G12C). |
-inhibitor-12-S733101W0420171213.gif)
|
|
| S7606 | RBC8 | RBC8 is a selective inhibitor of the GTPases RalA and RalB by inhibiting the binding of Ral to its effector RALBP1, no inhibition on the GTPases Ras and RhoA. |
|
|
| S8303 | Kobe0065 | Kobe0065 is an H-Ras-cRaf1 interaction inhibitor, exhibiting potent activity to competitively inhibit the binding of H-Ras·GTP to c-Raf-1 RBD with a Ki value of 46 ± 13 μM. | ||
| S8499 | KRpep-2d | KRpep-2d is a K-Ras(G12D) selective inhibitory cyclic peptide with selectivity against wild-type (WT) K-Ras and the K-Ras(G12C) mutant. The IC50 value is 1.6 nM. | ||
| S8955 | Sotorasib (AMG510) racemate | Sotorasib (AMG510) racemate is a racemic of AMG-510 which is a potent KRAS G12C covalent inhibitor with potential antineoplastic activity. | ||
| S7607 | BQU57 | BQU57, a derivative of RBC8, is a selective GTPase Ral inhibitor relative to the GTPases Ras and RhoA. | ||
| S8959 | BI-2852 | BI-2852 is a potent inhibitor of KRAS that binds with nanomolar affinity to a pocket between switch I and II on RAS. BI-2852 blocks all GEF, GAP, and effector interactions with KRAS, leading to inhibition of downstream signaling and an antiproliferative effect in the low micromolar range in KRAS mutant cells. | ||
| E0135 | CID-1067700 (ML282) | CID-1067700 (ML282) is a pan-GTPase inhibitor with a ki of 12.89 nM for Rab7wt. |
||
| S7332 | K-Ras(G12C) inhibitor 9 | K-Ras(G12C) inhibitor 9 is an allosteric inhibitor of oncogenic K-Ras(G12C). | ||
| E1474New | BI-2865 | BI-2865 is a none-covalent pan-KRAS Inhibitor. It binds to WT, G12C, G12D, G12V and G13D mutant KRAS with KDs of 6.9, 4.5, 32, 26, 4.3 nM respectively and inhibits the proliferation of G12C, G12D or G12V mutant KRAS expressing BaF3 ce | ||
| S6996 | LC-2 | LC-2 is a first PROTAC capable of degrading endogenous KRASG12C with DC50s between 0.25 μM and 0.76 μM. LC-2 covalently binds KRASG12C with a MRTX849 warhead and recruits the E3 ligase VHL, inducing rapid and sustained KRASG12C degradation leading to suppression of MAPK signaling. | ||
| S0730 | (Rac)-Antineoplaston A10 | (rac)-Antineoplaston A10 is the racemate of Antineoplaston A10, which is a Ras inhibitor potentially for the treatment of glioma, lymphoma, astrocytoma and breast cancer. | ||
| S0023 | K-Ras-IN-1 | K-Ras-IN-1 (MDK-3017) inhibits K-Ras by binding to K-Ras in a hydrophobic pocket that is occupied by Tyr-71 in the apo-Ras crystal structure. | ||
| E1187 | Divarasib (GDC-6036) | Divarasib (GDC-6036, RG6330) is a KRAS G12C inhibitor that binds irreversibly to switch II pocket of KRAS G12C (when in the inactive GDP-bound state) thereby blocking GTP binding and activation. | ||
| E1433New | LY3537982 (KRAS G12C inhibitor 19) | LY3537982 (KRAS G12C inhibitor 19) is a highly selective and potent inhibitor of the KRAS-G12C protein. LY3537982 selectively inhibits the growth of KRAS G12C mutant cells H23 G12C, H358 G12C, and H2122 G12C with IC50s of 1.04 nM, 1.16 nM, and 11.38 nM, respectively. | ||
| E1578New | garsorasib (D-1553 ) | Garsorasib (D-1553 ) is a potent and selective oral KRASG12C inhibitor with antitumor activity. It has the potential to treat KRAS G12C-mutated non-small cell lung cancer (NSCLC). | ||
| E1606New | RMC-6291 | RMC-6291 is an orally active and covalent inhibitor of KRASG12C(ON). RMC-6291 forms a tri-complex within tumor cells between KRASG12C(ON) and cyclophilin A (CypA). | ||
| S3493 | APS6-45 | APS6-45(Compound 10) is an orally active tumor-calibrated inhibitor (TCI) that inhibits RAS/MAPK signaling and exhibits antitumor activity. | ||
| S6905 | MCP110 | MCP110 is an inhibitor of Ras/Raf-1 interaction. MCP110 disrupts the interaction of activated Ras with Raf and is potential for the treatment of human tumors. | ||
| S7330 | 6H05 | 6H05 is a selective, and allosteric inhibitor of oncogenic K-Ras(G12C). | ||
| S7333 | K-Ras(G12C) inhibitor 6 | K-Ras(G12C) inhibitor 6 is an allosteric, and selective inhibitor of oncogenic K-Ras(G12C). | ||
| E1356New | BI-0474 | BI-0474 is a potent inhibitor of KRASG12C with an IC50 value of 7.0 nM. It inhibits GDP-KRAS::SOS1 protein-protein interactions. It exhibits anti-proliferative and anti-tumour activity and can be used in cancer research. | ||
| E1056 | Opnurasib (JDQ-443) | Opnurasib (JDQ-443,NVP-JDQ443) , a potent and selective, orally bioavailable covalent inhibitor of GDP-bound KRASG12C with an IC50 of 0.012 µM for inhibition of c-Raf recruitment. | ||
| S6527 | Kobe2602 | Kobe2602 is a selective Ras inhibitor that blocks H-Ras GTP binding to c-Raf-1 (Ki = 149 µM). | ||
| E1597New | RMC-6236 | RMC-6236 (RAS-IN-2) is a RAS(ON) multi-selective noncovalent inhibitor of the active, GTP-bound state of both mutant and wild-type variants of canonical RAS isoforms with broad therapeutic potential. RMC-6236 exhibits strong anticancer efficacy in RAS-addicted cell lines, especially those with mutations at codon 12 of KRAS. | ||
| E1047 | NAV-2729 | NAV-2729, an inhibitor of Arf6 activation which directly binds to Arf6, also inhibits all Arf guanine nucleotide exchange factors (ArfGEFs) with a strongest effect against BRAG2 and Arf1 . | ||
| S0323 | Antineoplaston A10 | Antineoplaston A10 is a naturally occurring substance in the human body that that can be potentially used for the treatment of glioma, lymphoma, astrocytoma and breast cancer. The main ingredient active of antineoplaston A10 (Phenylacetylglutamine, PG) inhibits RAS and promotes apoptosis. | ||
| S5279 | Fendiline hydrochloride | Fendiline hydrochloride is the hydrochloride salt form of Fendiline, which is an L-type calcium channel blocker and also a specific inhibitor of K-Ras plasma membrane targeting with no detectable effect on the localization of H- and N-Ras. | ||
| S3853 | Perillyl alcohol | Perillyl alcohol (Perilla alcohol, Isocarveol) is a monoterpene isolated from the essential oils of lavendin, peppermint, spearmint, cherries, celery seeds, and several other plants. | ||
| E1071 | ASP2453 | ASP2453, a potent, selective and covalent KRAS G12C inhibitor, inhibits the Son of Sevenless (SOS)-mediated interaction between KRAS G12C and Raf. ASP2453 is a potential therapeutic agent for KRAS G12C-mutated cancer. | ||
| E1157 | Pan-RAS-IN-1 | Pan-RAS-IN-1, a pan-Ras inhibitor, binds to KRasG12D-GppNHp with a Kd less than 20 μM in MST, ITC and NMR assays, also binds to Ras proteins and exhibits lethality in cells partially dependent on expression of Ras proteins. | ||
| S8831 | MRTX-1257 | MRTX-1257 is a potent, selective, irreversible, covalent and orally active inhibitor of KRAS G12C with IC50 of 900 pM for KRAS dependent ERK phosphorylation in H358 cells. | ||
| S0458 | KY1220 | KY1220 is a small molecule that destabilizes both β-catenin and Ras, via targeting the Wnt/β-catenin pathway with IC50 of 2.1 μM in HEK293 reporter cells. | ||
| E1564New | BI-2493 | BI-2493, a structural analogue of BI-2865, is a highly selective inhibitor of pan-KRAS. BI-2493 attenuates tumor growth and can be used for cancer research. | ||
| S1314 | Zoledronic acid (Zoledronate) | Zoledronic acid (Zoledronate), a potent osteoclast inhibitor, induces apoptosis in osteoclasts by inhibiting enzymes of the mevalonate pathway and preventing the isoprenylation of small GTP-binding proteins such as Ras and Rho. Zoledronic acid (ZA) also induces autophagy. |

|
|
| S8830 | Sotorasib (AMG510) | Sotorasib (AMG510) is a potent KRAS G12C covalent inhibitor with potential antineoplastic activity.This AMG510 is a chiral compound. | ||
| S2797 | Lonafarnib (SCH66336) | Lonafarnib (SCH66336) is an orally bioavailable FPTase inhibitor for H-ras, K-ras-4B and N-ras with IC50 of 1.9 nM, 5.2 nM and 2.8 nM in cell-free assays, respectively. Phase 3. |

|
|
| S8707 | ARS-1620 | ARS-1620 is a potent, orally bioavailable covalent inhibitor of KRASG12C and could achieve rapid and sustained in vivo target occupancy to induce tumor regression. | ||
| S5244 | Zoledronic acid monohydrate | Zoledronic acid (Zoledronate, CGP-4244) monohydrate, a nitrogen-containing bisphosphonate, is a potent osteoclast inhibitor which induces apoptosis in osteoclasts by inhibiting enzymes of the mevalonate pathway and preventing the isoprenylation of small GTP-binding proteins such as Ras and Rho. | ||
| S7684 | Salirasib | Salirasib (Farnesylthiosalicylic acid, FTS) is a potent competitive prenylated protein methyltransferase (PPMTase) inhibitor with Ki of 2.6 μM, which inhibits Ras methylation. Salirasib exerts antitumor effects and induces autophagy. Phase 2. | ||
| S8884 | Adagrasib (MRTX849) | Adagrasib (MRTX849) is a potent, selective, and covalent KRASG12C inhibitor that exhibits favorable drug-like properties, selectively modifies mutant cysteine 12 in GDP-bound KRASG12C and inhibits KRAS-dependent signaling. | ||
| S7224 | Deltarasin | Deltarasin is a small molecular inhibitor of KRAS-PDEδ interaction with Kd of 38 nM for binding to purified PDEδ. | ||
| S8156 | ARS-853 | ARS-853 is a selective, covalent KRAS(G12C) inhibitor that inhibits mutant KRAS-driven signaling by binding to the GDP-bound oncoprotein and preventing activation. ARS-853 also induces apoptosis. | ||
| S8916 | BI-3406 | BI-3406 (compound I-13) is a potent, selective and orally active inhibitor of the interaction between KRAS and Son of Sevenless 1 (SOS1) with IC50 of 5 nM. BI-3406 reduces the formation of GTP-loaded KRAS, and inhibits MAPK pathway signaling. BI-3406 exhibits anti-tumor activities. | ||
| E1051 | MRTX1133 | MRTX1133 is a highly selective inhibitor of mutant KRAS G12D and can reversibly binds to the activated and inactivated KRAS G12D mutants and inhibit their activity. The specificity of MRTX1133 to KRAS G12D is more than 1000 times that of wild-type KRAS. |
||
| S8826 | BAY-293 | BAY-293 selectively inhibits the KRAS-SOS1 interaction with an IC50 of 21 nM. | ||
| S7331 | K-Ras(G12C) inhibitor 12 | K-Ras(G12C) inhibitor 12 is an allosteric inhibitor of oncogenic K-Ras(G12C). |
-inhibitor-12-S733101W0420171213.gif)
|
|
| S7606 | RBC8 | RBC8 is a selective inhibitor of the GTPases RalA and RalB by inhibiting the binding of Ral to its effector RALBP1, no inhibition on the GTPases Ras and RhoA. |

|
|
| S8303 | Kobe0065 | Kobe0065 is an H-Ras-cRaf1 interaction inhibitor, exhibiting potent activity to competitively inhibit the binding of H-Ras·GTP to c-Raf-1 RBD with a Ki value of 46 ± 13 μM. | ||
| S8499 | KRpep-2d | KRpep-2d is a K-Ras(G12D) selective inhibitory cyclic peptide with selectivity against wild-type (WT) K-Ras and the K-Ras(G12C) mutant. The IC50 value is 1.6 nM. | ||
| S8955 | Sotorasib (AMG510) racemate | Sotorasib (AMG510) racemate is a racemic of AMG-510 which is a potent KRAS G12C covalent inhibitor with potential antineoplastic activity. | ||
| S7607 | BQU57 | BQU57, a derivative of RBC8, is a selective GTPase Ral inhibitor relative to the GTPases Ras and RhoA. | ||
| S8959 | BI-2852 | BI-2852 is a potent inhibitor of KRAS that binds with nanomolar affinity to a pocket between switch I and II on RAS. BI-2852 blocks all GEF, GAP, and effector interactions with KRAS, leading to inhibition of downstream signaling and an antiproliferative effect in the low micromolar range in KRAS mutant cells. | ||
| E0135 | CID-1067700 (ML282) | CID-1067700 (ML282) is a pan-GTPase inhibitor with a ki of 12.89 nM for Rab7wt. |
||
| S7332 | K-Ras(G12C) inhibitor 9 | K-Ras(G12C) inhibitor 9 is an allosteric inhibitor of oncogenic K-Ras(G12C). | ||
| E1474New | BI-2865 | BI-2865 is a none-covalent pan-KRAS Inhibitor. It binds to WT, G12C, G12D, G12V and G13D mutant KRAS with KDs of 6.9, 4.5, 32, 26, 4.3 nM respectively and inhibits the proliferation of G12C, G12D or G12V mutant KRAS expressing BaF3 ce | ||
| S0730 | (Rac)-Antineoplaston A10 | (rac)-Antineoplaston A10 is the racemate of Antineoplaston A10, which is a Ras inhibitor potentially for the treatment of glioma, lymphoma, astrocytoma and breast cancer. | ||
| S0023 | K-Ras-IN-1 | K-Ras-IN-1 (MDK-3017) inhibits K-Ras by binding to K-Ras in a hydrophobic pocket that is occupied by Tyr-71 in the apo-Ras crystal structure. | ||
| E1187 | Divarasib (GDC-6036) | Divarasib (GDC-6036, RG6330) is a KRAS G12C inhibitor that binds irreversibly to switch II pocket of KRAS G12C (when in the inactive GDP-bound state) thereby blocking GTP binding and activation. | ||
| E1433New | LY3537982 (KRAS G12C inhibitor 19) | LY3537982 (KRAS G12C inhibitor 19) is a highly selective and potent inhibitor of the KRAS-G12C protein. LY3537982 selectively inhibits the growth of KRAS G12C mutant cells H23 G12C, H358 G12C, and H2122 G12C with IC50s of 1.04 nM, 1.16 nM, and 11.38 nM, respectively. | ||
| E1578New | garsorasib (D-1553 ) | Garsorasib (D-1553 ) is a potent and selective oral KRASG12C inhibitor with antitumor activity. It has the potential to treat KRAS G12C-mutated non-small cell lung cancer (NSCLC). | ||
| E1606New | RMC-6291 | RMC-6291 is an orally active and covalent inhibitor of KRASG12C(ON). RMC-6291 forms a tri-complex within tumor cells between KRASG12C(ON) and cyclophilin A (CypA). | ||
| S3493 | APS6-45 | APS6-45(Compound 10) is an orally active tumor-calibrated inhibitor (TCI) that inhibits RAS/MAPK signaling and exhibits antitumor activity. | ||
| S6905 | MCP110 | MCP110 is an inhibitor of Ras/Raf-1 interaction. MCP110 disrupts the interaction of activated Ras with Raf and is potential for the treatment of human tumors. | ||
| S7330 | 6H05 | 6H05 is a selective, and allosteric inhibitor of oncogenic K-Ras(G12C). | ||
| S7333 | K-Ras(G12C) inhibitor 6 | K-Ras(G12C) inhibitor 6 is an allosteric, and selective inhibitor of oncogenic K-Ras(G12C). | ||
| E1356New | BI-0474 | BI-0474 is a potent inhibitor of KRASG12C with an IC50 value of 7.0 nM. It inhibits GDP-KRAS::SOS1 protein-protein interactions. It exhibits anti-proliferative and anti-tumour activity and can be used in cancer research. | ||
| E1056 | Opnurasib (JDQ-443) | Opnurasib (JDQ-443,NVP-JDQ443) , a potent and selective, orally bioavailable covalent inhibitor of GDP-bound KRASG12C with an IC50 of 0.012 µM for inhibition of c-Raf recruitment. | ||
| S6527 | Kobe2602 | Kobe2602 is a selective Ras inhibitor that blocks H-Ras GTP binding to c-Raf-1 (Ki = 149 µM). | ||
| E1597New | RMC-6236 | RMC-6236 (RAS-IN-2) is a RAS(ON) multi-selective noncovalent inhibitor of the active, GTP-bound state of both mutant and wild-type variants of canonical RAS isoforms with broad therapeutic potential. RMC-6236 exhibits strong anticancer efficacy in RAS-addicted cell lines, especially those with mutations at codon 12 of KRAS. | ||
| E1047 | NAV-2729 | NAV-2729, an inhibitor of Arf6 activation which directly binds to Arf6, also inhibits all Arf guanine nucleotide exchange factors (ArfGEFs) with a strongest effect against BRAG2 and Arf1 . | ||
| S0323 | Antineoplaston A10 | Antineoplaston A10 is a naturally occurring substance in the human body that that can be potentially used for the treatment of glioma, lymphoma, astrocytoma and breast cancer. The main ingredient active of antineoplaston A10 (Phenylacetylglutamine, PG) inhibits RAS and promotes apoptosis. | ||
| S5279 | Fendiline hydrochloride | Fendiline hydrochloride is the hydrochloride salt form of Fendiline, which is an L-type calcium channel blocker and also a specific inhibitor of K-Ras plasma membrane targeting with no detectable effect on the localization of H- and N-Ras. | ||
| E1071 | ASP2453 | ASP2453, a potent, selective and covalent KRAS G12C inhibitor, inhibits the Son of Sevenless (SOS)-mediated interaction between KRAS G12C and Raf. ASP2453 is a potential therapeutic agent for KRAS G12C-mutated cancer. | ||
| E1157 | Pan-RAS-IN-1 | Pan-RAS-IN-1, a pan-Ras inhibitor, binds to KRasG12D-GppNHp with a Kd less than 20 μM in MST, ITC and NMR assays, also binds to Ras proteins and exhibits lethality in cells partially dependent on expression of Ras proteins. | ||
| S8831 | MRTX-1257 | MRTX-1257 is a potent, selective, irreversible, covalent and orally active inhibitor of KRAS G12C with IC50 of 900 pM for KRAS dependent ERK phosphorylation in H358 cells. | ||
| E1564New | BI-2493 | BI-2493, a structural analogue of BI-2865, is a highly selective inhibitor of pan-KRAS. BI-2493 attenuates tumor growth and can be used for cancer research. | ||
| E1474New | BI-2865 | BI-2865 is a none-covalent pan-KRAS Inhibitor. It binds to WT, G12C, G12D, G12V and G13D mutant KRAS with KDs of 6.9, 4.5, 32, 26, 4.3 nM respectively and inhibits the proliferation of G12C, G12D or G12V mutant KRAS expressing BaF3 ce | ||
| E1433New | LY3537982 (KRAS G12C inhibitor 19) | LY3537982 (KRAS G12C inhibitor 19) is a highly selective and potent inhibitor of the KRAS-G12C protein. LY3537982 selectively inhibits the growth of KRAS G12C mutant cells H23 G12C, H358 G12C, and H2122 G12C with IC50s of 1.04 nM, 1.16 nM, and 11.38 nM, respectively. | ||
| E1578New | garsorasib (D-1553 ) | Garsorasib (D-1553 ) is a potent and selective oral KRASG12C inhibitor with antitumor activity. It has the potential to treat KRAS G12C-mutated non-small cell lung cancer (NSCLC). | ||
| E1606New | RMC-6291 | RMC-6291 is an orally active and covalent inhibitor of KRASG12C(ON). RMC-6291 forms a tri-complex within tumor cells between KRASG12C(ON) and cyclophilin A (CypA). | ||
| E1356New | BI-0474 | BI-0474 is a potent inhibitor of KRASG12C with an IC50 value of 7.0 nM. It inhibits GDP-KRAS::SOS1 protein-protein interactions. It exhibits anti-proliferative and anti-tumour activity and can be used in cancer research. | ||
| E1597New | RMC-6236 | RMC-6236 (RAS-IN-2) is a RAS(ON) multi-selective noncovalent inhibitor of the active, GTP-bound state of both mutant and wild-type variants of canonical RAS isoforms with broad therapeutic potential. RMC-6236 exhibits strong anticancer efficacy in RAS-addicted cell lines, especially those with mutations at codon 12 of KRAS. | ||
| E1564New | BI-2493 | BI-2493, a structural analogue of BI-2865, is a highly selective inhibitor of pan-KRAS. BI-2493 attenuates tumor growth and can be used for cancer research. |
Choose Selective Ras Inhibitors
Tags: Ras inhibitor|Ras agonist|Ras activator|Ras inducer|Ras antagonist|Ras signaling pathway|Ras assay kit