Smoothened (smo), a G protein-coupled receptor, drives the Hedgehog signalling pathway and is involved in gene expression and Gli family transcription factors. The core members of the Smo signalling pathway include the ligand (Hedgehog, Hh), the receptor (Patched, Ptc), several transducers such as Smoothened (Smo) and the ytoplasmic complex formed by the kinesin-like protein Costal-2 (Cos2), the serine/threonine kinase Fused (Fu), the novel protein Suppressor of fused [Su(fu)], and the transcription factor Cubitus interruptus (Ci). SMO is classified as a proto-oncogene because the tumour suppressor PTCH represses signalling by SMO, and SHH relieves this repression.
Hedgehog/Smoothened Inhibitors
Other GPCR & G Protein Related Inhibitors
Isoform-selective Products
| Cat.No. | Product Name | Information | Product Use Citations | Product Validations |
|---|---|---|---|---|
| S7779 | SAG (Smoothened Agonist) Hydrochloride | SAG (Smoothened Agonist) HCl is a cell-permeable Smoothened (Smo) agonist with EC50 of 3 nM in Shh-LIGHT2 cells. |
|

|
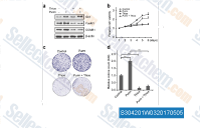
| S3042 | Purmorphamine | Purmorphamine (Shh Signaling Antagonist VI), which directly binds and activates Smoothened, blocks BODIPY-cyclopamine binding to Smo with IC50 of ~ 1.5 μM in HEK293T cell and also is an inducer of osteoblast differentiation with EC50 of 1 μM. Purmorphamine can reduce both basal and induced autophagy. |
|
|
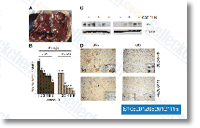
| S1082 | Vismodegib (GDC-0449) | Vismodegib (GDC-0449) is a potent, novel and specific hedgehog inhibitor with IC50 of 3 nM and also inhibits P-gp with IC50 of 3.0 μM in a cell-free assay. |
|
|
| S1146 | Cyclopamine (11-Deoxojervine) | Cyclopamine (11-deoxojervine) is a specific Hedgehog (Hh) signaling pathway antagonist of Smoothened (Smo) with IC50 of 46 nM in TM3Hh12 cells. |
|

|
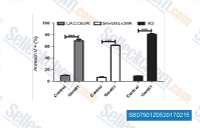
| S8075 | GANT 61 (NSC 136476) | GANT61 (NSC 136476) is an inhibitor for GLI1 as well as GLI2-induced transcription, inhibits hedgehog with IC50 of 5 μM in GLI1 expressing HEK293T cell, displays selectivity over other pathways, such as TNF and glucocorticoid receptor gene transactivation. GANT61 induces apoptosis and activates protective autophagy in LX-2 cells. |
|
|
| S2151 | Sonidegib (NVP-LDE225, Erismodegib) | Sonidegib is a Smoothened (Smo) antagonist, inhibiting Hedgehog (Hh) signaling with IC50 of 1.3 nM (mouse) and 2.5 nM (human) in cell-free assays, respectively. Phase 3. |
|

|
| S2476 | Itraconazole | Itraconazole is a relatively potent inhibitor of CYP3A4 with IC50 of 6.1 nM, used as a triazole antifungal agent. This compound is a potent antagonist of the Hedgehog (Hh) signaling pathway. It also suppresses the growth of glioblastoma through induction of autophagy. |
|

|
| S6384 | SAG (Smoothened Agonist) | SAG (Smoothened Agonist) is a potent agonist of Smoothened (Smo). Smoothened Agonist induces firefly luciferase expression in Shh-LIGHT2 cells with an EC50 of 3 nM. |
|
|
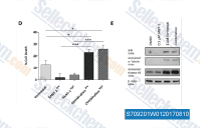
| S7092 | SANT-1 | SANT-1 directly binds to Smoothened (Smo) receptor with Kd of 1.2 nM and inhibits Smo agonist effects with IC50 of 20 nM. |
|
|
| S8249 | HPI-4 (Ciliobrevin A) | HPI-4 (Ciliobrevin A, Hedgehog Pathway Inhibitor 4) is a hedgehog (Hh) pathway antagonist. It blocks Sonic hedgehog (Shh)-induced Hh pathway activation (IC50 = 7 μM) downstream of Smo. |
|
|





































