research use only
Retigabine 2HCl Potassium Channel activator
Cat.No.S4734
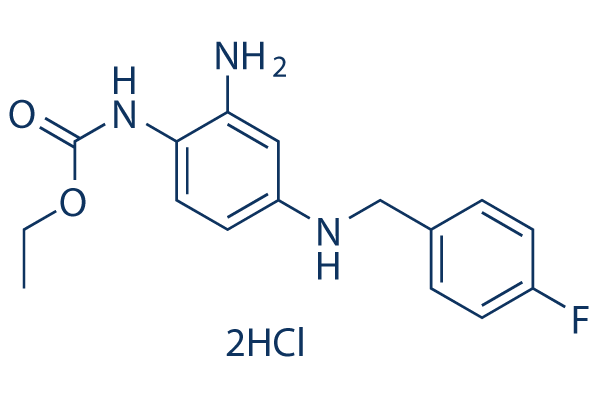
Chemical Structure
Molecular Weight: 376.25
Quality Control
| Related Targets | CFTR CRM1 CD markers AChR Calcium Channel Sodium Channel GABA Receptor TRP Channel ATPase GluR |
|---|---|
| Other Potassium Channel Inhibitors | Nigericin TRAM-34 Nicorandil ML133 HCl Hydralazine HCl Sophocarpine Senicapoc (ICA-17043) Gliquidone PAP-1 E-4031 dihydrochloride |
Solubility
|
In vitro |
DMSO
: 75 mg/mL
(199.33 mM)
Water : 75 mg/mL Ethanol : 25 mg/mL |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 376.25 | Formula | C16H18FN3O2.2HCl |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 150812-13-8 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N-(2-amino-4-[fluorobenzylamino]-phenyl) carbamic acid (2HCl), D-23129 2HCl | Smiles | CCOC(=O)NC1=C(C=C(C=C1)NCC2=CC=C(C=C2)F)N.Cl.Cl | ||
Mechanism of Action
| Targets/IC50/Ki |
KCNQ3/5
(Cell-based assay) 1.4 μM(EC50)
KCNQ2/Q3 channels
(Cell-based assay) 1.6 μM(EC50)
KCQN4
(Cell-based assay) 5.2 μM(EC50)
|
|---|---|
| In vitro |
Retigabine is a novel antiepileptic drug whose mechanism of action involves potassium channel opening activity in neuronal cells. Retigabine can markedly enhance KCNQ2/Q3 currents. In addition, retigabine also enhances slow channel deactivation. Retigabine has been shown to increase the synthesis of GABA in rat hippocampal slices and to enhance GABA-induced chloride currents in cultured rat cortical neurons. Retigabine has been shown to induce membrane hyperpolarization in neurones in rat hippocampal-entorhinal cortex slices and to exert potassium channel opening activity in neuronal cells. Retigabine enhances a linopirdine-sensitive current in differentiated PC12 cells. concentrations of RTG/EZG ≥10 μM are required to cause significant augmentation of the GABAA receptor response. The potency of RTG/EZG differs somewhat depending on the GABAA receptor subunit combination, with the following rank order: α1β3γ2=α1β2γ2 >α3β2γ2=α2β2γ2>α5β2γ2=α1β2(N265S)γ2=α1β1γ2. RTG/EZG exhibits only weak inhibitory effects at voltage-gated Nav and Cav channel currents at predominantly supratherapeutic concentrations. RTG/EZG does not interact significantly with glutamate receptors.
|
| In vivo |
Retigabine enhances γ-aminobutyric acid (GABA)-ergic transmission in the central nervous system. Retigabine is rapidly absorbed and distributed with an oral bioavailability of 60% and a high volume of distribution of approximately 6.2 L/kg. Tolerability is good in humans when titrated up to its therapeutic dose range (600-1200 mg/day). Plasma protein binding of the drug is approximately 80%. The relatively high systemic bioavailability after oral administration suggests that retigabine is resistant to first-pass metabolism, a finding confirmed in multiple species. Retigabine is metabolized exclusively via phase II hepatic glucuronidation and acetylation. Gender differences in exposure have been noted, with female subjects exhibiting higher plasma concentrations of the drug after oral administration than male subjects. Excretion of retigabine appears to be predominantly renal. Depending on the behavioral endpoints analyzed, it appears that retigabine has a relatively poor therapeutic index (ratio between TD50 obtained in rotarod and ED50 obtained in maximal electroshock where maximal tonic extension of the hindlimbs was used as endpoint) in both mice (TD50/ED50 = 2.2) and rats (TD50/ED50 = 1.9) after i.p. administration. However, after p.o. administration in rats retigabine shows a therapeutic index of 28.8, which compares favorably with that reported for other antiepileptics, such as carbamazepine.
|
References |
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































