- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- DNA Damage/DNA Repair
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleckchem.com to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly DYKDDDDK Tag Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
research use only
U73122 Phospholipase (e.g. PLA) inhibitor
U73122 is a potent phospholipase C (PLC) inhibitor, which reduces agonist-induced Ca2+ increases in platelets and PMN. This compound potently inhibits human 5-lipoxygenase (5-LO).
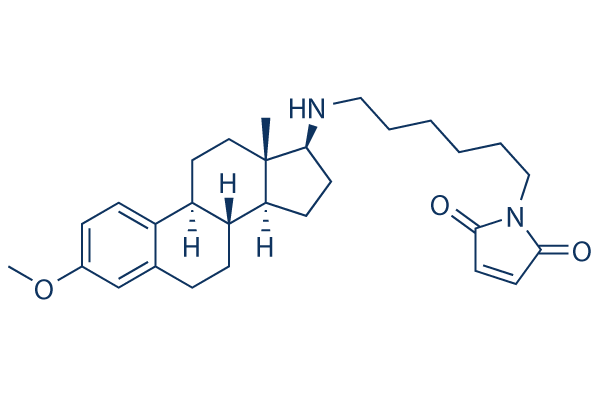
Chemical Structure
Molecular Weight: 464.64
Purity & Quality Control
Batch:
Purity:
99.22%
99.22
Related Products
| Related Targets | PLA2 PLC PLD LYPLA1 Smase | Click to Expand |
|---|---|---|
| Related Products | GW4869 Varespladib (LY315920) Tanshinone I Darapladib Quinacrine 2HCl CAY10593 (VU0155069) m-3M3FBS Melittin Trigonelline | Click to Expand |
| Related Compound Libraries | Metabolism Compound Library Anti-cancer Metabolism Compound Library Glutamine Metabolism Compound Library Carbohydrate Metabolism Compound Library Lipid Metabolism Compound Library | Click to Expand |
Signaling Pathway
Cell Culture and Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| human Jurkat cells | Function assay | Inhibition of human telomerase activity in human Jurkat cells by TRAP assay, IC50=0.2 μM | 24053596 | |||
| Click to View More Cell Line Experimental Data | ||||||
Mechanism of Action
| Targets |
|
|---|
In vitro |
||||
| In vitro | U73122 significantly inhibits aggregation of human platelets induced by a variety of agonists, including collagen, thrombin, ADP, arachidonic acid, with IC50 of 1-5 μM. This compound (10 μM) inhibits the production of IP3 and the subsequent rapid increase in cytosolic Ca2+ induced by either thrombin or U-46619 through inhibiting hydrolysis of phosphatidyl[3H]inositol and phosphatldyl[3H]inosito1 4,5-bisphosphate catalyzed by a soluble fraction from platelets (Ki=9 and 40 μM, respectively). It inhibits thromboxane B production induced by collagen through inhibiting receptor-coupled mobilization of arachidonic acid. This chemical inhibits also FMLP-induced aggregation of human polymorphonuclear neutrophils and the associated production of IP3 and diacylglycerol. [1] This compound causes a concentration-dependent inhibition of C5a, FMLP, PAF and LTB4-induced MPO and B12-BP release from cyto-chalasin B-treated PMNs. The IC50 values are 60 (FMLP), 110 (LTB4), 115 (C5a) and 120 (PAF) nM for MPO release; and 105 (FMLP), 110 (LTB4), 120 (C5a) and 140 (PAY) nM for B12-BP release. It is also a potent inhibitor of superoxide anion production by cytochalasin B-treated PMNs activated with either C5a or FMLP with IC50 of 160 and 300 nM, respectively. This chemical causes suppression of the rise in [Ca2+]i, IP3 production and DAG production in FMLP-stimulated PMNs with IC50 of 500 nM, 2 μM, and 2 μM, respectively. 3 μM of this compound causes 100% inhibition of FMLP-induced GTPase activity. It causes a concentration-dependent inhibition of the FMLP-evoked association of PKC with the extractable particulate fraction of PMNs, but not a soluble preparation of PMN PKC. [2] It significantly inhibits recombinant human PLC-β2, with an IC50 of ~6 μM. This compound has little effect on PLC-β1, PLC-β3, or PLC-β4. It reduces interleukin-8 and leukotriene B4-induced Ca2+ flux and chemotaxis in human neutrophils with IC50 of ~6 μM and ~5 μM, respectively. [3] 1 μM of this compound blocks bradykinin (BK)-induced increases in the intracellular free Ca2+ concentration in undifferentiated NG108-15 cell. The IC50 for a 20-min exposure is approximately 200 nM. 1 μM of this chemical produces a small but significant increase in [Ca2+]i which results from Ca 2+ release from an intracellular store. In differentiated NG108-15 cells it blocks completely depolarization-induced Ca2+ influx. In contrast, in DRG neurons this compound inhibits only slightly voltage-sensitive Ca2+ channels. [4] It is a relatively specific inhibitor of G-protein-mediated phospholipase C activation in pancreatic acini. This compound inhibits phosphatidylinositol (PI) hydrolysis on stimulation with either cholecystokinin (by 81%) or carbachol (by 73%) at a maximal effect concentration of 10 μM. This chemical (10 μM) inhibits the increases in [Ca2+]i stimulated by high concentrations of secretagogues in fura-2-loaded acini. It also inhibits the [Ca2+]i signal generated by directly stimulating G-proteins with sodium fluoride. This compound rapidly inhibits the oscillating [Ca2+]i signal elicited by low concentrations of cholecystokinin or carbachol. [5] It increases the activity of hPLCβ3 in a concentration-and time-dependent manner in a cell-free micellar system, with up to an 8-fold increase in enzyme activity and a EC50 of 13.6 μM. Activation of hPLCβ3 by this compound requires covalent modification of cysteines. This chemical (10 μM) also activates hPLCγ1(>10-fold) and hPLCβ2(~2-fold); PLC δ1 is neither activated nor inhibited. [6] |
|||
|---|---|---|---|---|
| Kinase Assay | Phosphoinositide-specific phospholipase C activity assay | |||
| Assay mixtures (0.2 mL) contains assay buffer (without leupeptin), CaCl2 (an amount calculated to produce a Ca-EGTA buffer with the required free Ca2+ concentration), platelet soluble fraction (5-15 μg of protein) and 4 nmol of either phosphatidyl[3H]inositol or phosphatidyl[3H]inositol 4,5-bisphosphate. Substrata mixtures consists of either phosphatidyl[3H]inositol (3.4 Ci/mol) plus 1-pahnitoyl-2-oleoyl-sn-glycero-3-phosphoethanolamine (1:0.4 M ratio) or phosphatidyl[3H]inositol4,5-bisphosphate (7 Ci/mol) plus 1-palmitoyl 2-oleoyl-sn-giycero-3-phosphoethanolamine (1:4 M ratio). Substrate mixtures are prepared as l0× suspensions of small unilamellar vesicles by consonication of the lipids in assay buffer (Vibra Cell microprobe sonicator, 75 W, 2 times 60 sec). U-73122 is added to assay mixtures in 2 μL of DMSO. This amount of DMSO has a negligible effect on phospholipase C activity under these conditions. This compound is incubated in glass tubes at 37 ℃ for 10 min and then quenched with 1.6 mL of chloroform/rnethanol/HC1 (1 N) (108:108:25, by vol). After vigorous mixing and centrifugation (500 g for 10 min), a portion (0.7 mL) of the aqueous upper layer (0.9 mL) is transferred to a scintillation vial that contains 10 mL of ACS scintillation fluid. Tritium in water-soluble products (>90% being inositol phosphates) is measured by liquid scintillation spectrornetry. Phosphoinositide hydrolysis under these conditions never exceeds 10% of total added and proceeded at a constant rate throughout the incubation period. Blank values are determined from assay mixtures in which platelet soluble fraction is added after the stopping reagent. | ||||
In Vivo |
||
| In vivo | U73122 (30 mg/kg i.p.) blocks swelling of rats hind paw by 65 and 80% at 1 and 3 h postcarrageenan challenge. This compound (0.1 mg/mL) inhibits carrageenan-induced macrophage and lymphocyte accumulation into subcutaneous chambers in dogs by 65 and 74%, respectively. This chemical (30 mg/kg i.p.) totally inhibits the LPS-induced increase in macrophage and lymphocyte infiltration and prostaglandin E2 production (by 80%) in a mouse peritonitis model, and inhibits and 12- O-tetradecanoyl-phorbol-13-acetate-induced ear edema in mice. [3] |
|
|---|---|---|
| Animal Research | Animal Models | Swiss-Webster mic |
| Dosages | 30 mg/kg | |
| Administration | i.p. | |
References |
|
Chemical Information
| Molecular Weight | 464.64 | Formula | C29H40N2O3 |
| CAS No. | 112648-68-7 | SDF | Download SDF |
| Synonyms | N/A | ||
| Smiles | CC12CCC3C(C1CCC2NCCCCCCN4C(=O)C=CC4=O)CCC5=C3C=CC(=C5)OC | ||
Storage and Stability
| Storage (From the date of receipt) | |||
|
In vitro |
DMSO : 93 mg/mL ( (200.15 mM) Moisture-absorbing DMSO reduces solubility. Please use fresh DMSO.) Water : Insoluble Ethanol : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
|||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field