research use only
SRPIN340 Serine/threonin kinase inhibitor
Cat.No.S7270
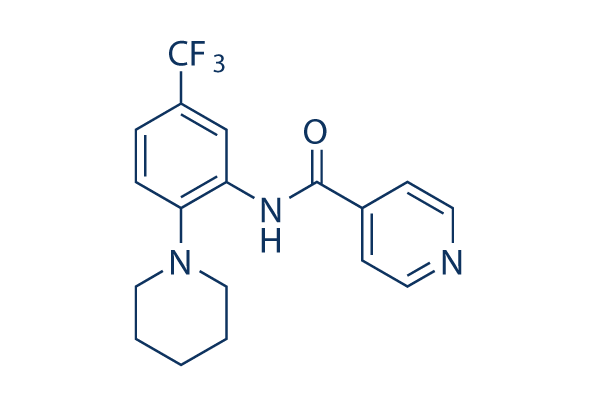
Chemical Structure
Molecular Weight: 349.35
Quality Control
| Related Targets | Dehydrogenase HSP Transferase P450 (e.g. CYP17) PDE phosphatase PPAR Vitamin Carbohydrate Metabolism Mitochondrial Metabolism |
|---|---|
| Other Serine/threonin kinase Inhibitors | WNK463 GCN2iB Benzamidine HCl SPHINX31 ML281 SGC-GAK-1 BAY-1816032 DCLK1-IN-1 WNK-IN-11 MKI-1 |
Solubility
|
In vitro |
DMSO
: 70 mg/mL
(200.37 mM)
Ethanol : 70 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 349.35 | Formula | C18H18F3N3O |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 218156-96-8 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | C1CCN(CC1)C2=C(C=C(C=C2)C(F)(F)F)NC(=O)C3=CC=NC=C3 | ||
Mechanism of Action
| Features |
The only specific SRPK1 and SRPK2 inhibitor. Potential use in congenital diseases and viral diseases including HIV, HCV and HBV.
|
|---|---|
| Targets/IC50/Ki |
SRPK1
0.89 μM(Ki)
|
| In vitro |
SRPIN340 inhibits SR phosphorylation by SRPK in Flp-In293 cells and promotes degradation of SRp75 in a dose-dependent manner, which subsequently inhibits HIV production. This compound dose (5 mg/mL) produces no abnormalities in chromosomal structure and chromosome number of CHO cells. It suppresses in a dose-dependent fashion expression of a HCV subgenomic replicon and replication of the HCV-JFH1 clone in vitro.
|
| In vivo |
SRPIN340 inhibits CNV formation in a dose-dependent manner in vivo. This compound significantly decreases the protein levels of VEGF, MCP-1, ICAM-1, and consequently inhibits macrophage infiltration.
|
References |
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































