research use only
Pomalidomide (CC-4047) Immunomodulatory agent
Cat.No.S1567
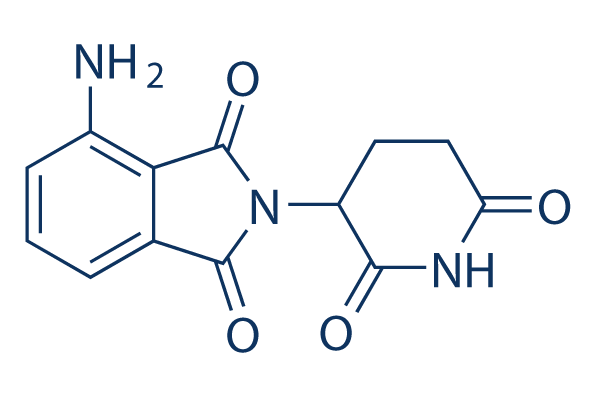
Chemical Structure
Molecular Weight: 273.24
Quality Control
| Related Targets | Proteasome E1 Activating E3 Ligase DUB SUMO p97 E2 conjugating |
|---|---|
| Other E3 ligase Ligand Inhibitors | CC-99282 |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| MOLP-8 | Cytotoxicity Assay | 10 μM | 24 h | potently augments direct and indirect MM cell killing by SAR | 26338273 | |
| J-CD38 | Cytotoxicity Assay | 10 μM | 24 h | potently augments direct and indirect MM cell killing by SAR | 26338273 | |
| R-CD38 | Cytotoxicity Assay | 10 μM | 24 h | potently augments direct and indirect MM cell killing by SAR | 26338273 | |
| BC-3 | Growth Inhibition Assay | 39-1250 nM | 5 d | DMSO | IC50=107 nM, inhibits cell IC50=107 nM, viability dose dependently | 26119939 |
| BCBL-1 | Growth Inhibition Assay | 39-1250 nM | 5 d | DMSO | IC50=74 nM, inhibits cell viability dose dependently | 26119939 |
| JSC-1 | Growth Inhibition Assay | 39-1250 nM | 5 d | DMSO | IC50=34 nM, inhibits cell viability dose dependently | 26119939 |
| VG-1 | Growth Inhibition Assay | 39-1250 nM | 5 d | DMSO | IC50=101 nM, inhibits cell viability dose dependently | 26119939 |
| UMPEL-1 | Growth Inhibition Assay | 39-1250 nM | 5 d | DMSO | IC50=32 nM, inhibits cell viability dose dependently | 26119939 |
| UMPEL-3 | Growth Inhibition Assay | 39-1250 nM | 5 d | DMSO | IC50=111 nM, inhibits cell viability dose dependently | 26119939 |
| BC-1 | Growth Inhibition Assay | 39-1250 nM | 5 d | DMSO | IC50=744 nM, inhibits cell viability dose dependently | 26119939 |
| BCP-1 | Growth Inhibition Assay | 39-1250 nM | 5 d | DMSO | IC50=396 nM, inhibits cell viability dose dependently | 26119939 |
| APK-1 | Growth Inhibition Assay | 39-1250 nM | 5 d | DMSO | IC50=226 nM, inhibits cell viability dose dependently | 26119939 |
| RPMI8226 | Growth Inhibition Assay | 0.01-50 μM | 48 h | DMSO | IC50=8 μM | 26097872 |
| OPM2 | Growth Inhibition Assay | 0.01-50 μM | 48 h | DMSO | IC50=10 μM | 26097872 |
| RPMI8226 | Function Assay | 10 μM | 48 h | DMSO | strengthens cytoplasmic-nuclear shuttling of mTOR and p-mTOR protein | 26097872 |
| OPM2 | Function Assay | 10 μM | 48 h | DMSO | strengthens cytoplasmic-nuclear shuttling of mTOR and p-mTOR protein | 26097872 |
| RPMI8226 | Function Assay | 0.1-10 μM | 4 h | DMSO | increases VEGF mRNA expression | 25053990 |
| SH-SY5Y | Apoptosis Assay | 25 μg/mL | 1 h | causes statistically significant reduction in both CPF- and CPF+CM-induced apoptosis | 24975276 | |
| JJN3 | Growth Inhibition Assay | 0.1-100 μM | 72 h | DMSO | inhibits cell growth slightly | 23178378 |
| XG-1 | Growth Inhibition Assay | 0.1-100 μM | 72 h | DMSO | inhibits cell growth | 23178378 |
| CD138+ | Growth Inhibition Assay | 0.1-100 μM | 72 h | DMSO | inhibits cell growth | 23178378 |
| XG-1 | Function Assay | 2/100 μM | 24 h | DMSO | inhibits CCL3/MIP-1α mRNA expression | 23178378 |
| U266 | Growth Inhibition Assay | 0.01-10 μM | 48 h | DMSO | inhibits cell growth dose dependently | 22552008 |
| CRBN60 | Growth Inhibition Assay | 0.01-10 μM | 48 h | DMSO | inhibits cell growth dose dependently | 22552008 |
| CRNB75 | Growth Inhibition Assay | 0.01-10 μM | 48 h | DMSO | inhibits cell growth dose dependently | 22552008 |
| MM.1S | Growth Inhibition Assay | 0.01-10 μM | 48 h | DMSO | significantly inhibits proliferation at concentrations as low as 0.01μM | 21389327 |
| OPM2 | Growth Inhibition Assay | 0.01-10 μM | 48 h | DMSO | significantly inhibits proliferation at concentrations as low as 0.01μM | 21389327 |
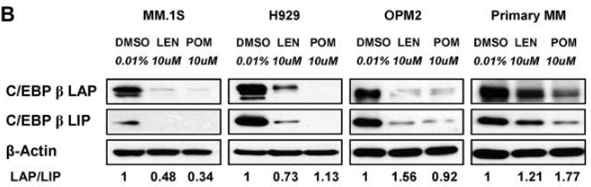
| MM.1S | Function Assay | 10 μM | 72 h | DMSO | significantly decreases the protein level of C/EBPβ isoforms | 21389327 |
| H929 | Function Assay | 10 μM | 72 h | DMSO | significantly decreases the protein level of C/EBPβ isoforms | 21389327 |
| OPM2 | Function Assay | 10 μM | 72 h | DMSO | significantly decreases the protein level of C/EBPβ isoforms | 21389327 |
| CT26 | Function Assay | 1/10 μM | 24 h | reduces the numbers of live colonies | 19638977 | |
| T-cells | Function assay | 2 to 3 days | Inhibition of IL-2 production in human T cells measured after 2 to 3 days by ELISA, EC50 = 0.008 μM. | 23168019 | ||
| DF15 | Function assay | 4 hrs | Induction of cereblon-mediated aiolos degradation in human DF15 cells expressing ePL-tagged aiolos after 4 hrs by luminometric analysis, EC50 = 0.022 μM. | 28425720 | ||
| DF15 | Function assay | 4 hrs | Induction of cereblon-mediated ikaros degradation in human DF15 cells expressing ePL-tagged ikaros after 4 hrs by luminometric analysis, EC50 = 0.024 μM. | 28425720 | ||
| DF15 | Function assay | 4 hrs | Induction of CRL4/CRBN ubiquitin ligase-mediated aiolos degradation in human DF15 cells expressing pLOC-ePL-tagged aiolos after 4 hrs by luminescence based beta-galactosidase enzyme fragmentation complementation assay, EC50 = 0.027 μM. | 28358507 | ||
| NAMALWA | Antiproliferative assay | 72 hrs | Antiproliferative activity against human NAMALWA cells assessed as inhibition of [3H]thymidine incorporation after 72 hrs by scintillation counting, IC50 = 0.03 μM. | 23168019 | ||
| HeLa | Function assay | Inhibition of IL-1-alpha-induced NF-kappaB activation in HeLa cells assessed as blocking of p50/p65 nuclear translocation, IC50 = 1.27 μM. | 17845850 | |||
| DF15 | Function assay | 0.01 to 1 uM | 5 hrs | Induction of cereblon-mediated aiolos degradation in human DF15 cells at 0.01 to 1 uM after 5 hrs by immunoblot analysis | 28425720 | |
| OPM2 | Function assay | 0.01 to 1 uM | 5 hrs | Induction of cereblon-mediated ikaros degradation in human OPM2 cells at 0.01 to 1 uM after 5 hrs by immunoblot analysis | 28425720 | |
| DF15 | Function assay | 0.01 to 1 uM | 5 hrs | Induction of cereblon-mediated ikaros degradation in human DF15 cells at 0.01 to 1 uM after 5 hrs by immunoblot analysis | 28425720 | |
| OPM2 | Function assay | 0.01 to 1 uM | 5 hrs | Induction of cereblon-mediated aiolos degradation in human OPM2 cells at 0.01 to 1 uM after 5 hrs by immunoblot analysis | 28425720 | |
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(365.97 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 273.24 | Formula | C13H11N3O4 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 19171-19-8 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | CC-4047 | Smiles | C1CC(=O)NC(=O)C1N2C(=O)C3=C(C2=O)C(=CC=C3)N | ||
Mechanism of Action
| Features |
A derivative of thalidomide and up to 10,000 times more potent than thalidomide.
|
|---|---|
| Targets/IC50/Ki |
CRBN
TNF-α
(PBMCs) 13 nM
|
| In vitro |
Pomalidomide inhibits lipopolysaccharide (LPS) stimulated TNF-alpha release in human PBMC and in human whole blood with IC50 values of 13 nM and 25 nM, respectively. This compound inhibits the growth of T regulatory cells which is stimulated by IL-2 with an IC50 of ~1 μM. Treatment with this chemical (6.4 nM-10 μM) increases the production of IL-2 in human peripheral blood T cells, and is slightly more potent in the CD4+ subset than in the CD8+ subset. It is significantly more potent than CC-5013 at elevating IL-2, IL-5, and IL-10 levels, but only slightly more potent than CC-5013 at elevating IFN-γ levels. This agent enhances SEE and Raji cells induced AP-1 transcriptional activity in Jurkat cells in a dose-dependent manner, with a maximal enhancement of 4-fold at 1 μM. Exposure of Raji cells to various concentrations of this compound (2.5-40 μg/mL) for 48 hours leads to a significant decrease in cell proliferation and DNA synthesis. There is a reduction of ~40% compared to vehicle-treated controls. |
| Kinase Assay |
Inhibition of TNF-α synthesis
|
|
TNF-α inhibitory activity is measured in lipopolysacharide (LPS) stimulated PBMC. Pomalidomide is added to human PBMCs 1 hour prior to the addition of LPS (1 μg/mL) and incubation continued for an additional 18-20 hours. Supernatants are then harvested, and the concentration of TNF-α in the supernatants is determined by ELISA. The concentration of this compound that inhibits TNF- production by 50% (IC50) is calculated by nonlinear regression analysis. The human whole blood TNF- inhibition assay is run in a similar fashion to the PBMC assay except that heparinized fresh human whole blood is plated directly into microtiter plates.
|
|
| In vivo |
Pomalidomide enhances the antitumor effect of rituximab against B-cell lymphomas in severe combined immunodeficient mice. Administration of this compound in combination with rituximab, gives the mice a median survival period of 74 days compared with 58 days of CC5013/rituximab treatment and 45 days of rituximab nonotherapy. The synergistic effect of this compound and rituximab can be completely abrogated by depletion of NK cells, supporting the proposal that NK cell expansion is one mechanism by which this compound may augment rituximab antitumor activity. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | CEBPβ IKZF1 / IKZF3 / UBE2G1 / CRBN |
|
21389327 |
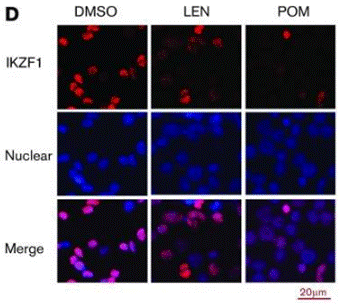
| Immunofluorescence | IKZF1 |
|
29496670 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT04902443 | Recruiting | Viral Associated Malignancies|Kaposi Sarcoma|EBV/KSHV-associated Lymphomas |
National Cancer Institute (NCI)|National Institutes of Health Clinical Center (CC) |
December 10 2021 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
Is the formulation S1567 in 1% DMSO+30% polyethylene glycol+1% Tween 80 suitable for its oral administration?
Answer:
Its suspension in 1% DMSO+30% polyethylene glycol+1% Tween 80 is for oral gavage.
Question 2:
I would like to know if it is racemic or optically active?
Answer:
Our S1567 is racemic.






































