- Bioactive Compounds
- By Signaling Pathways
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- Compound Libraries
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-Ⅱ
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Plant Extract Library
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at [email protected] to customize your library.
You could select:
- Antibodies
- Bioreagents
- qPCR
- 2x SYBR Green qPCR Master Mix
- 2x SYBR Green qPCR Master Mix(Low ROX)
- 2x SYBR Green qPCR Master Mix(High ROX)
- Protein Assay
- Protein A/G Magnetic Beads for IP
- Anti-Flag magnetic beads
- Anti-Flag Affinity Gel
- Anti-Myc magnetic beads
- Anti-HA magnetic beads
- Poly FLAG Peptide lyophilized powder
- Protease Inhibitor Cocktail
- Protease Inhibitor Cocktail (EDTA-Free, 100X in DMSO)
- Phosphatase Inhibitor Cocktail (2 Tubes, 100X)
- Cell Biology
- Cell Counting Kit-8 (CCK-8)
- Animal Experiment
- Mouse Direct PCR Kit (For Genotyping)
- New Products
- Contact Us
Pemetrexed disodium
Synonyms: LY-231514 disodium
Pemetrexed disodium is a novel antifolate and antimetabolite for TS, DHFR and GARFT with Ki of 1.3 nM, 7.2 nM and 65 nM in cell-free assays, respectively. Pemetrexed induces autophagy and apoptosis.
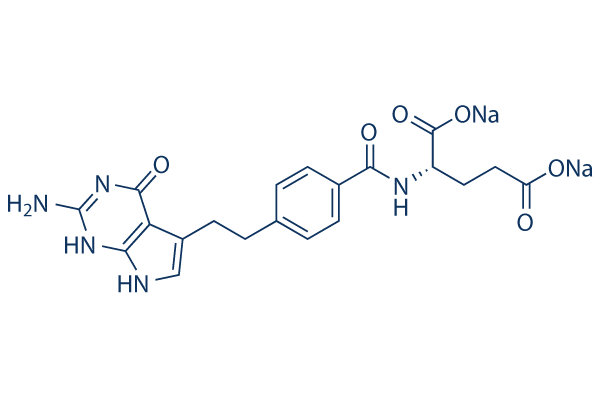
Pemetrexed disodium Chemical Structure
CAS: 150399-23-8
Selleck's Pemetrexed disodium has been cited by 42 publications
Purity & Quality Control
Batch:
Purity:
99.97%
99.97
Pemetrexed disodium Related Products
| Related Compound Libraries | Metabolism Compound Library Anti-cancer Metabolism Compound Library Glutamine Metabolism Compound Library Carbohydrate Metabolism Compound Library Lipid Metabolism Compound Library | Click to Expand |
|---|
Signaling Pathway
Choose Selective DHFR Inhibitors
Cell Data
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| PANC-1 | Function Assay | 84 μM | 24 h | enhances EGFR, HER3 and AKT phosphorylation levels | 22977607 | |
| BXPC-3 | Function Assay | 39.86 μM | 24 h | enhances EGFR, HER3 and AKT phosphorylation levels | 22977607 | |
| PANC-1 | Function Assay | 84 μM | 24 h | enhances EGFR phosphorylated levels, and the protein levels | 22977607 | |
| BXPC-3 | Function Assay | 39.86 μM | 24 h | enhances EGFR phosphorylated levels, and the protein levels | 22977607 | |
| BXPC-3 | Function Assay | 39.86 μM | 24 h | induces S arrest (P<0.05) and decreases the number of cells in the G2/M phase | 22977607 | |
| PANC-1 | Growth Inhibition Assay | IC50=83.76±0.19 μM | 22977607 | |||
| BXPC-3 | Growth Inhibition Assay | IC50=39.86±1.68 μM | 22977607 | |||
| Jurkat | Growth Inhibition Assay | 48 h | proportion of live cells=76.7±4.3 % | 23840376 | ||
| DM-3 | Growth Inhibition Assay | 48 h | proportion of live cells=101.3±2.8 % | 23840376 | ||
| JL-1 | Growth Inhibition Assay | 48 h | proportion of live cells=98.2±2.2 % | 23840376 | ||
| ZL-34 | Growth Inhibition Assay | 48 h | proportion of live cells=95.7±6.5 % | 23840376 | ||
| STAV-FCS | Growth Inhibition Assay | 48 h | proportion of live cells=88.1±8.9 % | 23840376 | ||
| M-14-K | Growth Inhibition Assay | 48 h | proportion of live cells=81.8±12.9 % | 23840376 | ||
| STAV-AB | Growth Inhibition Assay | 48 h | proportion of live cells=64.4±21.8 % | 23840376 | ||
| LoVo | Function Assay | 0.05 μM | 72 h | DMSO | increases the percentage of cells in the S phase | 23959460 |
| HT-29 | Function Assay | 0.05 μM | 72 h | DMSO | increases the percentage of cells in the S phase | 23959460 |
| WiDr | Growth Inhibition Assay | 72 h | DMSO | IC50=0.019 ± 0.002 μM | 23959460 | |
| SW1116 | Growth Inhibition Assay | 72 h | DMSO | IC50=1.70 ± 0.03 μM | 23959460 | |
| HCT116 | Growth Inhibition Assay | 72 h | DMSO | IC50=0.049 ± 0.013 μM | 23959460 | |
| SW620 | Growth Inhibition Assay | 72 h | DMSO | IC50=1.09 ± 0.01 μM | 23959460 | |
| LoVo | Growth Inhibition Assay | 72 h | DMSO | IC50=0.032 ± 0.002 μM | 23959460 | |
| HT-29 | Growth Inhibition Assay | 72 h | DMSO | IC50=10.07 ± 0.94 μM | 23959460 | |
| A549/PEM-16 | Growth Inhibition Assay | 96 h | IC50=51.45 μM | 24348854 | ||
| A549/PEM-6.4 | Growth Inhibition Assay | 96 h | IC50=23.39 μM | 24348854 | ||
| A549/PEM-1.6 | Growth Inhibition Assay | 96 h | IC50=5.03 μM | 24348854 | ||
| A549 | Growth Inhibition Assay | 96 h | IC50=1.35 μM | 24348854 | ||
| MSTO-211H | Cell Viability Assay | 1-10 μg/ml | 72 h | IC50~0.04 μg/ml | 24378576 | |
| Y-meso14 | Cell Viability Assay | 1-10 μg/ml | 72 h | IC50>10 μg/ml | 24378576 | |
| H290 | Cell Viability Assay | 1-10 μg/ml | 72 h | IC50>10 μg/ml | 24378576 | |
| H226 | Cell Viability Assay | 1-10 μg/ml | 72 h | IC50>10 μg/ml | 24378576 | |
| H1299 | Growth Inhibition Assay | IC50=1.84 μM | 24418519 | |||
| H1993 | Growth Inhibition Assay | IC50=0.17 μM | 24418519 | |||
| A549 | Function Assay | 0.1/0.3/0.5/1 μM | 24/48 h | DMSO | produces the formation of AVOs in a dose-dependent manner | 24626722 |
| A549 | Cell Viability Assay | 0.1/0.3/0.5/1 μM | 24/48 h | DMSO | inhibits cell viability dose and time dependently | 24626722 |
| MES04 | Growth Inhibition Assay | IC50>100 μM | 24714722 | |||
| MES01 | Growth Inhibition Assay | IC50>100 μM | 24714722 | |||
| H2452 | Growth Inhibition Assay | IC50>100 μM | 24714722 | |||
| H2052 | Growth Inhibition Assay | IC50=0.57±0.34 μM | 24714722 | |||
| 211H | Growth Inhibition Assay | IC50=0.07±0.01 μM | 24714722 | |||
| H28 | Growth Inhibition Assay | IC50=0.07±0.02 μM | 24714722 | |||
| PC9/GR | Function Assay | 4.94 μM | 72 h | increases p-AKT levels | 24840891 | |
| PC9/GR | Function Assay | 4.94 μM | 72 h | increases p-ERK levels | 24840891 | |
| PC9/GR | Apoptosis Assay | 4.94 μM | 72 h | induces 19.54﹪ apoptosis | 24840891 | |
| PC9 | Apoptosis Assay | 16 nM | 72 h | induces 14.54﹪ apoptosis | 24840891 | |
| PC9/GR | Function Assay | 4.94 μM | 72 h | induces S-phase arrest | 24840891 | |
| PC9 | Function Assay | 16 nM | 72 h | induces S-phase arrest | 24840891 | |
| PC9/GR | Growth Inhibition Assay | 72 h | IC50=4.94±0.440 μM | 24840891 | ||
| PC9 | Growth Inhibition Assay | 72 h | IC50=16.05±1.85 nM | 24840891 | ||
| A549 | Function Assay | 0.1/0.3/1 μM | 48 h | increases the ratio of S-phase population | 24847863 | |
| A549 | Apoptosis Assay | 0.1/0.3/1 μM | 48 h | induces apoptosis in a dose dependent manner | 24847863 | |
| A549 | Function Assay | 0.1/0.3/1 μM | 48 h | increases the level of phosphorylated Akt in a dose dependent manner | 24847863 | |
| A549 | Function Assay | 1 µM | 4/8/12/24/48 h | increases the level of phosphorylated Akt in a time dependent manner | 24847863 | |
| A549 | Function Assay | 5 μM | 8 h | increases Mcl-1 ubiquitination levels | 24991768 | |
| A549 | Apoptosis Assay | 2.5 μM | 48 h | induces apoptosis | 24991768 | |
| H1792 | Function Assay | 2.5/5/10 μM | 48 h | downregulates Mcl-1 | 24991768 | |
| A549 | Function Assay | 2.5/5/10 μM | 48 h | downregulates Mcl-1 | 24991768 | |
| H1792 | Function Assay | 2.5 μM | 48 h | induces caspase-9, caspase-3 and poly (ADP-ribose) polymerase (PARP) cleavage | 24991768 | |
| A549 | Function Assay | 2.5 μM | 48 h | induces caspase-9, caspase-3 and poly (ADP-ribose) polymerase (PARP) cleavage | 24991768 | |
| H1792 | Function Assay | 2.5/5/10 μM | 48 h | increases Noxa expression significantly | 24991768 | |
| A549 | Function Assay | 2.5/5/10 μM | 48 h | increases Noxa expression significantly | 24991768 | |
| A549 | Function Assay | 1 μM | 48 h | leads to mitochondrial dysfunction combined with simvastatin | 25096993 | |
| MSTO-211 | Function Assay | 1 μM | 48 h | leads to mitochondrial dysfunction combined with simvastatin | 25096993 | |
| A549 | Function Assay | 1 μM | 48 h | enhances intracellular ROS production combined with simvastatin | 25096993 | |
| MSTO-211 | Function Assay | 1 μM | 48 h | enhances intracellular ROS production combined with simvastatin | 25096993 | |
| A549 | Apoptosis Assay | 1 μM | 48 h | enhances caspase-dependent apoptosis combined with simvastatin | 25096993 | |
| MSTO-211 | Apoptosis Assay | 1 μM | 48 h | enhances caspase-dependent apoptosis combined with simvastatin | 25096993 | |
| A549 | Cell Viability Assay | 1 μM | 48 h | produces a synergistic inhibitory effect on the cell growth combined with simvastatin | 25096993 | |
| MSTO-211 | Cell Viability Assay | 1 μM | 48 h | produces a synergistic inhibitory effect on the cell growth combined with simvastatin | 25096993 | |
| H1299 | Cell Viability Assay | 1-1000 nM | 72 h | IC50=178 nM | 25145669 | |
| A549 | Cell Viability Assay | 1-1000 nM | 72 h | IC50=137 nM | 25145669 | |
| MG-63 | Cell Viability Assay | 0.01-100 μM | 72 h | inhibits cell viability dosedependently | 25152399 | |
| U20S | Cell Viability Assay | 0.01-100 μM | 72 h | inhibits cell viability dosedependently | 25152399 | |
| HepG3 | Function Assay | 10 μM | 48 h | upregulates phosphorylated (p-) MEK1/2 (Ser217/221) and p-ERK1/2 (Thr202/Tyr204) | 25446102 | |
| HepG3 | Function Assay | 0.1–100 μM | 48 h | activates cyto-protective autophagy | 25446102 | |
| HepG3 | Function Assay | 0.1–100 μM | 48 h | induces p62 downregulation as well as Beclin-1 and LC3B-II upregulation | 25446102 | |
| HepG2 | Apoptosis Assay | 0.1–100 μM | 72 h | induces apoptosis slightly at high concerntration | 25446102 | |
| HepG2 | Cell Viability Assay | 0.1–100 μM | 72 h | high concentrations of pemetrexed at (10/100 μM) only slightly inhibits HepG2 cell survival | 25446102 | |
| A459 | Apoptosis Assay | 4μM | 48 h | induces apoptosis | 25743822 | |
| A459 | Function Assay | 1/2/4 μM | 48 h | decreases the levels of p-Akt | 25743822 | |
| A459 | Function Assay | 1/2/4 μM | 24/48 h | induces G1 phase arrest in dose- and time dependent manner | 25743822 | |
| T47D | Growth Inhibition Assay | 0.234 mM | 72 h | growth inhibition=30﹪ | 25975637 | |
| HeLa | Growth Inhibition Assay | 0.234 mM | 72 h | growth inhibition=20﹪ | 25975637 | |
| A549 | Growth Inhibition Assay | 0.234 mM | 72 h | growth inhibition=50﹪ | 25975637 | |
| Vero | Growth Inhibition Assay | 0.234 mM | 72 h | growth inhibition=20﹪ | 25975637 | |
| T47D | Growth Inhibition Assay | 0.234 mM | 48 h | growth inhibition=20﹪ | 25975637 | |
| HeLa | Growth Inhibition Assay | 0.234 mM | 48 h | growth inhibition=10﹪ | 25975637 | |
| A549 | Growth Inhibition Assay | 0.234 mM | 48 h | growth inhibition=30﹪ | 25975637 | |
| Vero | Growth Inhibition Assay | 0.234 mM | 48 h | growth inhibition=10﹪ | 25975637 | |
| T47D | Growth Inhibition Assay | 0.234 mM | 24 h | growth inhibition=10﹪ | 25975637 | |
| HeLa | Growth Inhibition Assay | 0.234 mM | 24 h | growth inhibition=5﹪ | 25975637 | |
| A549 | Growth Inhibition Assay | 0.234 mM | 24 h | growth inhibition=10﹪ | 25975637 | |
| Vero | Growth Inhibition Assay | 0.234 mM | 24 h | growth inhibition=5﹪ | 25975637 | |
| A549 | Function Assay | 1 μM | 48 h | increases AMPK phosphorylation and a concomitant decrease in AKT and mTOR phosphorylation cotreated with simvastatin | 26334320 | |
| MSTO-211H | Function Assay | 1 μM | 48 h | increases AMPK phosphorylation and a concomitant decrease in AKT and mTOR phosphorylation cotreated with simvastatin | 26334320 | |
| A549 | Apoptosis Assay | 1 μM | 24 h | induces apoptosis cotreated with simvastatin | 26334320 | |
| MSTO-211H | Apoptosis Assay | 1 μM | 24 h | induces apoptosis cotreated with simvastatin | 26334320 | |
| A549 | Function Assay | 1 μM | 24 h | enhances autophagy cotreated with simvastatin | 26334320 | |
| MSTO-211H | Function Assay | 1 μM | 24 h | enhances autophagy cotreated with simvastatin | 26334320 | |
| A549 | Cell Viability Assay | 1 μM | 48 h | enhances simvastatin inhibited viability | 26334320 | |
| MSTO-211H | Cell Viability Assay | 1 μM | 48 h | enhances simvastatin inhibited viability | 26334320 | |
| KB | Function assay | IC50 = 0.03 μM | 18680275 | |||
| RT16 | Antiproliferative assay | IC50 = 0.042 μM | 18680275 | |||
| D4 | Antiproliferative assay | IC50 = 0.06 μM | 18680275 | |||
| KB | Antiproliferative assay | IC50 = 0.068 μM | 18680275 | |||
| IGROV1 | Antiproliferative assay | IC50 = 0.102 μM | 18680275 | |||
| PC43-10 | Antiproliferative assay | IC50 = 0.138 μM | 18680275 | |||
| IGROV1 | Antiproliferative assay | IC50 = 0.2 μM | 18680275 | |||
| D4 | Antiproliferative assay | IC50 = 0.254 μM | 18680275 | |||
| KB | Antiproliferative assay | IC50 = 0.327 μM | 18680275 | |||
| RT16 | Antiproliferative assay | IC50 = 0.388 μM | 18680275 | |||
| R2 | Antiproliferative assay | IC50 = 0.894 μM | 18680275 | |||
| KB | Function assay | 30 mins | IC50 = 0.03 μM | 19371039 | ||
| R2 | Function assay | Ki = 0.096 μM | 20085328 | |||
| R2 | Function assay | Ki = 1.54 μM | 20085328 | |||
| KB | Function assay | IC50 = 30 μM | 20085328 | |||
| R2 | Antiproliferative assay | 10 to 14 days | IC50 = 0.00494 μM | 21879757 | ||
| R2 | Antiproliferative assay | 96 hrs | IC50 = 0.0132 μM | 21879757 | ||
| RT16 | Antiproliferative assay | 96 hrs | IC50 = 0.042 μM | 21879757 | ||
| D4 | Antiproliferative assay | 96 hrs | IC50 = 0.06 μM | 21879757 | ||
| KB | Antiproliferative assay | 96 hrs | IC50 = 0.068 μM | 21879757 | ||
| IGROV1 | Antiproliferative assay | 96 hrs | IC50 = 0.102 μM | 21879757 | ||
| PC43-10 | Antiproliferative assay | 96 hrs | IC50 = 0.138 μM | 21879757 | ||
| IGROV1 | Antiproliferative assay | 96 hrs | IC50 = 0.2 μM | 21879757 | ||
| D4 | Antiproliferative assay | 96 hrs | IC50 = 0.254 μM | 21879757 | ||
| KB | Antiproliferative assay | 96 hrs | IC50 = 0.327 μM | 21879757 | ||
| RT16 | Antiproliferative assay | 96 hrs | IC50 = 0.388 μM | 21879757 | ||
| R2 | Antiproliferative assay | 96 hrs | IC50 = 0.894 μM | 21879757 | ||
| R2(VC) | Antiproliferative assay | 96 hrs | IC50 = 0.974 μM | 21879757 | ||
| R2 | Function assay | Ki = 0.094 μM | 22243528 | |||
| R2 | Function assay | Ki = 2.54 μM | 22243528 | |||
| R2 | Growth inhibition assay | 96 hrs | IC50 = 0.0132 μM | 24111942 | ||
| RT16 | Growth inhibition assay | 96 hrs | IC50 = 0.042 μM | 24111942 | ||
| D4 | Growth inhibition assay | 96 hrs | IC50 = 0.06 μM | 24111942 | ||
| KB | Growth inhibition assay | 96 hrs | IC50 = 0.068 μM | 24111942 | ||
| PC43-10 | Growth inhibition assay | 96 hrs | IC50 = 0.138 μM | 24111942 | ||
| D4 | Growth inhibition assay | 96 hrs | IC50 = 0.254 μM | 24111942 | ||
| KB | Growth inhibition assay | 96 hrs | IC50 = 0.327 μM | 24111942 | ||
| RT16 | Growth inhibition assay | 96 hrs | IC50 = 0.388 μM | 24111942 | ||
| MTXRII-OuaR2-4 | Growth inhibition assay | 96 hrs | IC50 = 0.894 μM | 24111942 | ||
| R2(VC) | Growth inhibition assay | 96 hrs | IC50 = 0.974 μM | 24111942 | ||
| KB | Cytotoxicity assay | 96 hrs | IC50 = 0.00994 μM | 24256410 | ||
| KB | Function assay | 30 mins | IC50 = 0.01174 μM | 24256410 | ||
| RT16 | Cytotoxicity assay | 96 hrs | IC50 = 0.0182 μM | 24256410 | ||
| R2 | Cytotoxicity assay | 96 hrs | IC50 = 0.0223 μM | 24256410 | ||
| PC43-10 | Cytotoxicity assay | 96 hrs | IC50 = 0.0306 μM | 24256410 | ||
| KB | Cytotoxicity assay | 96 hrs | IC50 = 0.69 μM | 24256410 | ||
| R2/PCFT4 | Function assay | 96 hrs | IC50 = 0.0132 μM | 25234128 | ||
| RT16 | Function assay | 96 hrs | IC50 = 0.042 μM | 25234128 | ||
| D4 | Function assay | 96 hrs | IC50 = 0.06 μM | 25234128 | ||
| KB | Cytotoxicity assay | 96 hrs | IC50 = 0.068 μM | 25234128 | ||
| PC43-10 | Function assay | 96 hrs | IC50 = 0.138 μM | 25234128 | ||
| D4 | Function assay | 96 hrs | IC50 = 0.254 μM | 25234128 | ||
| KB | Cytotoxicity assay | 96 hrs | IC50 = 0.327 μM | 25234128 | ||
| RT16 | Function assay | 96 hrs | IC50 = 0.388 μM | 25234128 | ||
| R2 | Cytotoxicity assay | 96 hrs | IC50 = 0.894 μM | 25234128 | ||
| R2 | Cytotoxicity assay | 96 hrs | IC50 = 0.974 μM | 25234128 | ||
| KB | Function assay | 1 hr | IC50 = 0.01174 μM | 25602637 | ||
| R2/PCFT4 | Function assay | 96 hrs | IC50 = 0.0132 μM | 25602637 | ||
| RT16 | Function assay | 96 hrs | IC50 = 0.042 μM | 25602637 | ||
| R2 | Cytotoxicity assay | 96 hrs | IC50 = 0.042 μM | 25602637 | ||
| KB | Antiproliferative assay | 96 hrs | IC50 = 0.068 μM | 25602637 | ||
| PC43-10 | Function assay | 96 hrs | IC50 = 0.138 μM | 25602637 | ||
| R2(VC) | Cytotoxicity assay | 96 hrs | IC50 = 0.974 μM | 25602637 | ||
| KB | Cytotoxicity assay | 72 hrs | IC50 = 0.07 μM | 25668494 | ||
| A549 | Cytotoxicity assay | 72 hrs | IC50 = 0.08 μM | 25668494 | ||
| HepG2 | Cytotoxicity assay | 72 hrs | IC50 = 1.26 μM | 25668494 | ||
| R2/PCFT4 | Function assay | 2 mins | K = 0.044 μM | 26317331 | ||
| R2/PCFT4 | Function assay | 2 mins | K = 0.27 μM | 26317331 | ||
| KB | Antiproliferative assay | 72 hrs | IC50 = 0.07 μM | 27017552 | ||
| SW620 | Antiproliferative assay | 72 hrs | IC50 = 0.08 μM | 27017552 | ||
| A549 | Antiproliferative assay | 72 hrs | IC50 = 1.26 μM | 27017552 | ||
| KB | Function assay | 72 hrs | IC50 = 0.07 μM | 28830032 | ||
| SW620 | Antiproliferative assay | 72 hrs | IC50 = 0.09 μM | 28830032 | ||
| MCF7 | Antiproliferative assay | 72 hrs | IC50 = 0.65 μM | 28830032 | ||
| R2/PCFT4 | Function assay | 96 hrs | IC50 = 0.0132 μM | 29425443 | ||
| RT16 | Function assay | 96 hrs | IC50 = 0.042 μM | 29425443 | ||
| D4 | Function assay | 96 hrs | IC50 = 0.06 μM | 29425443 | ||
| KB | Antiproliferative assay | 96 hrs | IC50 = 0.068 μM | 29425443 | ||
| PC43-10 | Function assay | 96 hrs | IC50 = 0.138 μM | 29425443 | ||
| D4 | Function assay | 96 hrs | IC50 = 0.254 μM | 29425443 | ||
| R2/PCFT4 | Function assay | 2 mins | Ki = 0.259 μM | 29425443 | ||
| KB | Antiproliferative assay | 96 hrs | IC50 = 0.327 μM | 29425443 | ||
| R2 | Cytotoxicity assay | 96 hrs | IC50 = 0.849 μM | 29425443 | ||
| RT16 | Function assay | 96 hrs | IC50 = 0.894 μM | 29425443 | ||
| R2(VC) | Growth inhibition assay | 96 hrs | IC50 = 0.974 μM | 29425443 | ||
| A549 | Antiproliferative assay | 24 hrs | IC50 = 3.31 μM | 29807332 | ||
| MDA-MB-231 | Antiproliferative assay | 24 hrs | IC50 = 3.85 μM | 29807332 | ||
| OVCAR3 | Antiproliferative assay | 24 hrs | IC50 = 6.9 μM | 29807332 | ||
| SGC7901 | Antiproliferative assay | 24 hrs | IC50 = 9.08 μM | 29807332 | ||
| KB | Function assay | 1 uM | 24 hrs | Induction of apoptotic activity in human KB cells at 1 uM after 24 hrs | 18680275 | |
| R2 | Antiproliferative assay | Antiproliferative activity against chinese hamster R2 cells expressing human PCFT assessed as growth inhibition in the presence of 10 uM thymidine and 320 uM AICA | 21879757 | |||
| R2 | Antiproliferative assay | Antiproliferative activity against chinese hamster R2 cells expressing human PCFT assessed as growth inhibition in the presence of 60 uM adenosine | 21879757 | |||
| R2 | Function assay | Inhibition of GARFTase in chinese hamster R2 cells expressing human PCFT assessed as incorporation of [14C]glycine into [14C]formyl GAR in the presence of 4 uM azaserine | 21879757 | |||
| R2 | Function assay | 10 uM | Inhibition of human PCFT-mediated [3H]MTX uptake ectopically expressed in chinese hamster R2 cells at 10 uM at pH 5.5 to 7.2 | 21879757 | ||
| PC43-10 | Function assay | 10 uM | 2 mins | Inhibition of human RFC-mediated [3H]MTX uptake in chinese hamster PC43-10 cells at 10 uM after 2 mins relative to control | 21879757 | |
| KB | Function assay | 1 uM | 48 hrs | Inhibition of AICARFTase in human KB cells assessed as phosphorylated AMPK at 1 uM after 48 hrs by Western blot analysis | 24256410 | |
| KB | Cytotoxicity assay | 96 hrs | Cytotoxicity against human KB cells expressing human RFC/FRalpha/PCFT after 96 hrs by CellTitre-Blue fluorescence assay in presence of adenosine/AICA/thymidine | 24256410 | ||
| KB | Cell cycle arrest assay | 1 uM | 48 hrs | Cell cycle arrest in human KB cells assessed as accumulation at G1/G0 phase at 1 uM after 48 hrs by propidium iodide staining-based flow cytometry relative to control | 24256410 | |
| KB | Function assay | 48 hrs | Inhibition of AICARFTase in human KB cells assessed as accumulation of ZMP after 48 hrs by HPLC analysis | 24256410 | ||
| R2/PCFT4 | Function assay | 0.5 uM | 5 mins | Binding affinity to human PCFT expressed in Chinese hamster R2/PCFT4 cells assessed as intracellular drug level at 0.5 uM at 37 degC at pH 5.5 measured over 5 mins | 26317331 | |
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for TC32 cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for A673 cells) | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for NB1643 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for SK-N-MC cells | 29435139 | |||
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for TC32 cells | 29435139 | |||
| IGROV1 | Function assay | Effect on TS protein expression in human IGROV1 cells by Western blot analysis | 30035541 | |||
| IGROV1 | Function assay | Effect on DHFR protein expression in human IGROV1 cells by Western blot analysis | 30035541 | |||
| IGROV1 | Function assay | Effect on HSP90 protein expression in human IGROV1 cells by Western blot analysis | 30035541 | |||
| Click to View More Cell Line Experimental Data | ||||||
Biological Activity
| Description | Pemetrexed disodium is a novel antifolate and antimetabolite for TS, DHFR and GARFT with Ki of 1.3 nM, 7.2 nM and 65 nM in cell-free assays, respectively. Pemetrexed induces autophagy and apoptosis. | ||||||
|---|---|---|---|---|---|---|---|
| Targets |
|
| In vitro | ||||
| In vitro | Pemetrexed disodium shows the antiproliferative activity in CCRF-CEM leukemia, GC3/C1 colon carcinoma, and HCT-8 ileocecal carcinoma cells with IC50 of 25 nM, 34 nM and 220 nM, respectively. [1] A recent study shows that cisplatin plus Pemetrexed combined with SOCS-1 gene delivery shows the antitumor effect by inhibition of cell proliferation, invasiveness, and induction of apoptosis in MPM cells infected with adenovirus-expressing SOCS-1 vector. [2] | |||
|---|---|---|---|---|
| Kinase Assay | Enzyme Assays and Methods. | |||
| TS activity is assayed using a spectrophotometric method, which involved monitoring the increase in absorbance at 340 nm resulting from formation of the product, 7,8-dihydrofolate. The assay buffer contains 50 mM N-tris[hydroxymethyljmethyl-2-aminoethanesulfonic acid, 25 mM MgC12, 6.5 mM formaldehyde, 1 mM EDTA, and 75 mM 2-mercaptoethanol, pH 7.4. The concentrations of deoxyuridylate monophosphate, 6R-MTHF, and hIS are 100 μM, 30μM and 30 nM (1.7 milliunits/mL), respectively. At the 6R-MTHF concentration, an uninhibited reaction and six concentrations of inhibitor are assayed. Ki app values are determined by fitting the data to the Morrison equation using nonlinear regression analysis with the aid of the program ENZFITTER. Ki values are calculated using the equation: Ki app= Ki(1 + [S]/Km), where [S] is equal to 30 μM and Km is equal to 3 μM. DHFR activity is assayed spectrophotometrically by monitoring the dis appearance of the substrates NADPH and 7,8-dihydrofolate at 340 nm. The reaction takes place at 25°C in 0.5 mL of 50 mM potassium phosphate buffer, which contains 150 mM KC1 and 10 nM 2-mercaptoethanol, pH 7.5, and 14 nM (0.34 milliunitlmL) DHFR. The NADPH concentration is 10 μM and 7,8-dihydrofolate is varied at 5, 10, or 15 μM. At each 7,8-dihydrofolate concentration, an uninhibited reaction and seven concentrations of inhibitor are assayed. The ENZFITI'ER microcomputer program is used to obtain Ki app values by fitting the data to the Morrison equation by nonlinear regression analysis. Ki app= Ki(1 + [S]/Km), where [S] is equal to the concentration of 7,8-dihydrofolate used and Km of 7,8-dihydrofolate is equal to 0.15 μM. GARFT activity is assayed spectrophotometrically by monitoring the increase of absorbance resulting from formation of the product 5,8-dideazafolate at 295 nm. The reaction solvent contains 75 mM HEPES, 20% glycerol, and 50 mM a-thioglygerol, pH 7.5, at 25°C. The concentrations of substrates and enzyme used are 10 μM α,β-glycinamide ribonucleotide, 0-10 μM 10-formyl-5,8-dideazafolic acid, and 10 nM (1.9 milliunits/mL) GARFT. Ki values are calculated using the Enzyme Mechanism program of the Beckman DU640 spectrophotometer, which uses nonlinear regression analysis to fit data to the Michaelis-Menten equation for competitive inhibition. | ||||
| Cell Research | Cell lines | CCRF-CEM leukemia, GC3/C1 colon carcinoma, and HCT-8 ileocecal carcinoma cells. | ||
| Concentrations | 0-30 μM | |||
| Incubation Time | 72 hours | |||
| Method | Dose-response curves are generated to determine the concentration required for 50% inhibition of growth (IC50). Pemetrexed disodium is dissolved initially in DMSO at a concentration of 4 mg/mL and further diluted with cell culture medium to the desired concentration. CCRF-CEM leukemia cells in complete medium are added to 24-well Cluster plates in a total volume of 2.0 mL. Pemetrexed disodium at various concentrations are added to duplicate wells so that the final volume of DMSO is 0.5%. The plates are incubated for 72 hour at 37 °C in an atmosphere of 5% CO2 in air. At the end of the incubation, cell numbers are determined on a ZBI Coulter counter. For several studies, IC50s are determined for each compound in the presence of either 300 μM AICA, 5 μM thymidine, 100 μM hypoxanthine, or combination of 5 μM hymidine plus 100 μM hypoxanthine. For adherent tumor cells, a modification of the original MTT colorimetric assay is used to measure cell cytotoxicity. The human tumor cells are seeded in 100 μL assay medium/well in 96-well flat-bottomed tissue culture plates. The assay medium contains folic acid-free RPMI 1640 supplemented with 10% FCS and either 2 nM folinic acid or 2.3 μM folic acid as the sole folate source. Well 1A is left blank. Stock solutions of antifolates are prepared in Dulbecco's PBS at 1 mg/mL, and a series of 2-fold dilutions are subsequently made in PBS. Ten-μL aliquots of each concentration are added to triplicate wells. Plates are incubated for 72 hours at 37 °C in a humidified atmosphere of 5% CO2-in-air. MTT is dissolved in PBS at 5 mg/mL, 10 μL of stock MTF solution are added to each well of an assay, and the plates are incubated at 37 °C for 2 additional hours. Following incubation, 100 μL of DMSO are added to each well. After thorough formazan solubilization, the plates are read on a Dynatech MR600 reader, using a test wavelength of 570 nm and a reference wavelength of 630 nm. The IC50 is determined as the concentration of drug required to inhibit cell growth by 50% compared to an untreated controls. | |||
| Experimental Result Images | Methods | Biomarkers | Images | PMID |
| Western blot | p-Chk1 / Chk1 / Cyclin D / Cyclin E / p-Histone H3 / Histone H3 / Cyclin B1 / p-Cdc2 / Cdc2 Topo IIα / Topo I / γH2AX / Cleaved PARP / Survivin AKT / p-AKT / GSK3β / p-GSK3β EGFR / p-EGFR |
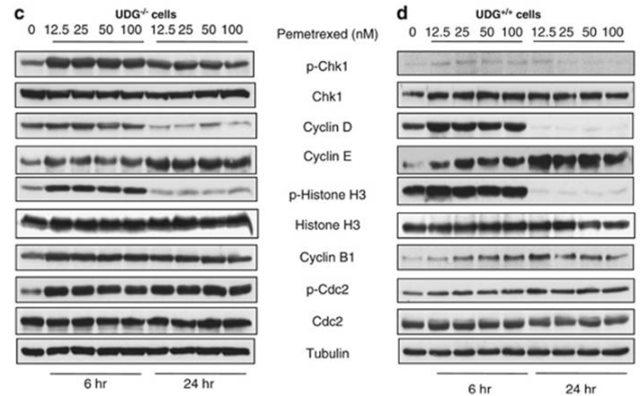
|
22237209 | |
| Immunofluorescence | p-AKT |
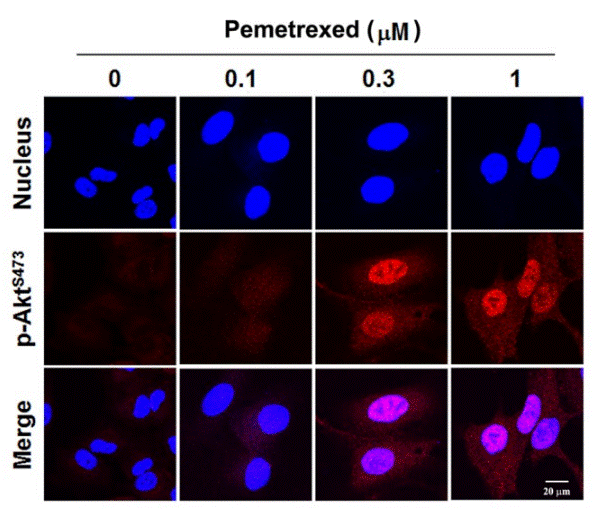
|
24847863 | |
| Growth inhibition assay | Cell viability |
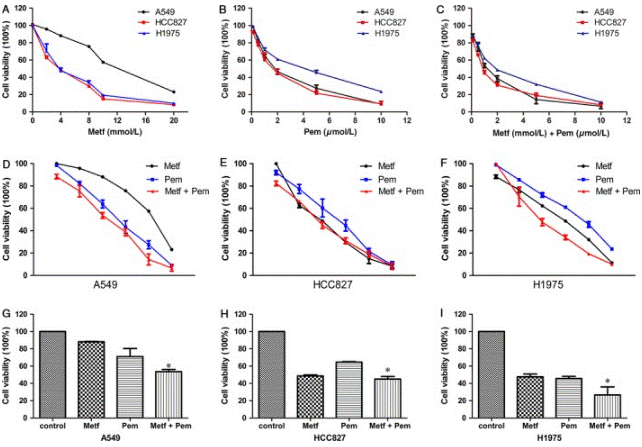
|
28719077 | |
| In Vivo | ||
| In vivo | In the human H460 non-small cell lung carcinoma xenograft, Pemetrexed disodium produces a duration-dependent tumor growth delay (TGD). [3] | |
|---|---|---|
| Animal Research | Animal Models | EMT-6 mammary carcinoma, the human HCT 116 colon carcinoma, and the human H460 non-small cell lung carcinoma are injected s.c. into the nude mice. |
| Dosages | 100 mg/kg or 150 mg/kg | |
| Administration | Administered via i.p. | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06010277 | Recruiting | NSCLC|Mesothelioma|Thymoma |
Amphia Hospital|Albert Schweitzer Hospital |
February 6 2023 | Phase 4 |
Chemical Information & Solubility
| Molecular Weight | 471.37 | Formula | C20H19N5Na2O6 |
| CAS No. | 150399-23-8 | SDF | Download Pemetrexed disodium SDF |
| Smiles | C1=CC(=CC=C1CCC2=CNC3=C2C(=O)NC(=N3)N)C(=O)NC(CCC(=O)[O-])C(=O)[O-].[Na+].[Na+] | ||
| Storage (From the date of receipt) | |||
|
In vitro |
Water : 94 mg/mL DMSO : Insoluble ( Moisture-absorbing DMSO reduces solubility. Please use fresh DMSO.) Ethanol : Insoluble |
Molecular Weight Calculator |
|
In vivo Add solvents to the product individually and in order. |
In vivo Formulation Calculator |
||||
Preparing Stock Solutions
Molarity Calculator
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
mg/kg
g
μL
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Tech Support
Answers to questions you may have can be found in the inhibitor handling instructions. Topics include how to prepare stock solutions, how to store inhibitors, and issues that need special attention for cell-based assays and animal experiments.
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
* Indicates a Required Field
Tags: buy Pemetrexed disodium | Pemetrexed disodium supplier | purchase Pemetrexed disodium | Pemetrexed disodium cost | Pemetrexed disodium manufacturer | order Pemetrexed disodium | Pemetrexed disodium distributor