research use only
Dapagliflozin (BMS-512148) SGLT2 inhibitor
Cat.No.S1548
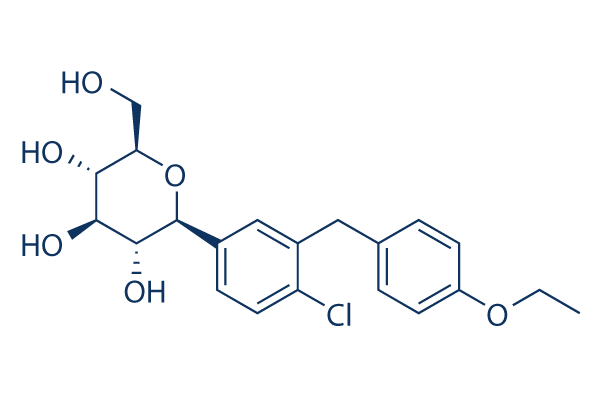
Chemical Structure
Molecular Weight: 408.87
Quality Control
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| CHO cells | Function assay | 2 h | Inhibition of human SGLT2 expressed in CHO cells assessed as [14C]alpha-methyl-D-glucopyranoside uptake after 2 hrs by liquid scintillation counting, IC50=0.00049 μM | 20434909 | ||
| CHOK1 cells | Function assay | 3 h | Inhibition of human SGLT2 expressed in CHOK1 cells assessed as inhibition of [14C]-AMG uptake after 3 hrs by microbeta scintillation counting analysis, IC50=0.001 μM | 24842618 | ||
| HEK293.ETN cells | Function assay | 1.5 h | Inhibition of human SGLT2 transfected in HEK293.ETN cells assessed as AMG uptake after 1.5 hrs by scintillation counting, IC50=0.0067 μM | 19896374 | ||
| COS-7 cells | Function assay | 2 h | Inhibition of human SGLT1 transfected in COS-7 cells assessed as AMG uptake after 2 hrs by scintillation counting, IC50=0.89 μM | 19896374 | ||
| CHO | Function assay | Inhibition of human SGLT2 expressed in CHO cells assessed as inhibition of [14C]AMG accumulation, EC50 = 0.0011 μM. | 18260618 | |||
| CHO | Function assay | Inhibition of rat SGLT2 expressed in CHO cells assessed as inhibition of [14C]AMG accumulation, EC50 = 0.003 μM. | 18260618 | |||
| CHO | Function assay | Inhibition of human SGLT1 expressed in CHO cells assessed as inhibition of [14C]AMG accumulation, EC50 = 1.39 μM. | 18260618 | |||
| HEK293.ETN | Function assay | Inhibition of human SGLT2 expressed in HEK293.ETN cells assessed as methyl-alpha-D-[U-14C]glucopyranoside uptake by scintillation counting, IC50 = 0.0067 μM. | 19700318 | |||
| COS7 | Function assay | Inhibition of human SGLT1 expressed in african green monkey COS7 cells assessed as methyl-alpha-D-[U-14C]glucopyranoside uptake by scintillation counting, IC50 = 0.885 μM. | 19700318 | |||
| HEK293 | Function assay | Inhibition of human SGLT1 expressed in HEK293 cells assessed as inhibition of [14C]alpha-methylglucopyranoside uptake, IC50 = 0.81 μM. | 19785435 | |||
| CHO | Function assay | 2 hrs | Inhibition of human SGLT2 expressed in CHO cells assessed as inhibition of sodium-dependent methyl-alpha-D-[U-14C]glucopyranoside uptake after 2 hrs, IC50 = 0.0014 μM. | 20149653 | ||
| CHO | Function assay | 2 hrs | Inhibition of human SGLT1 expressed in CHO cells assessed as inhibition of sodium-dependent methyl-alpha-D-[U-14C]glucopyranoside uptake after 2 hrs, IC50 = 1.2 μM. | 20149653 | ||
| CHO | Function assay | Inhibition of human recombinant SGLT2 expressed in CHO cells by liquid scintillation counting, IC50 = 0.00049 μM. | 20181486 | |||
| HEK293 | Function assay | 1.5 hrs | Inhibition of human SGLT2 expressed in HEK293 cells assessed as inhibition of [14C]alpha-methyl-D-glucopyranoside uptake after 1.5 hrs by liquid scintillation counting, IC50 = 0.0067 μM. | 20576578 | ||
| COS7 | Function assay | 2 hrs | Inhibition of human SGLT1 expressed in african green monkey COS7 cells assessed as inhibition of [14C]alpha-methyl-D-glucopyranoside uptake after 2 hrs by liquid scintillation counting, IC50 = 0.89 μM. | 20576578 | ||
| CHO | Function assay | 2 hrs | Inhibition of human SGLT2 expressed in CHO cells assessed as sodium-dependent [14C]-alpha-methyl-D-glucopyranoside uptake after 2 hrs by liquid scintillation counting, EC50 = 0.003 μM. | 21128592 | ||
| CHO | Function assay | 2 hrs | Inhibition of human SGLT1 expressed in CHO cells assessed as sodium-dependent [14C]-alpha-methyl-D-glucopyranoside uptake after 2 hrs by liquid scintillation counting, EC50 = 0.4285 μM. | 21128592 | ||
| CHO | Function assay | 2 hrs | Inhibition of human SGLT2 expressed in CHO cells assessed as inhibition of [14C]alpha-methyl-D-glucopyranoside uptake after 2 hrs by liquid scintillation counting, IC50 = 0.00049 μM. | 21193308 | ||
| CHO | Function assay | 60 mins | Inhibition of human SGLT2 expressed in CHO cells assessed as inhibition of [14C]-alpha-methyl-D-glucopyranoside transport after 60 mins by scintillation counting, IC50 = 0.004 μM. | 21398124 | ||
| CHO | Function assay | 60 mins | Inhibition of human SGLT2 expressed in CHO cells assessed as inhibition of [14C]-alpha-methyl-D-glucopyranoside transport after 60 mins by scintillation counting in presence of 100% plasma, IC50 = 0.022 μM. | 21398124 | ||
| CHO | Function assay | 60 mins | Inhibition of human SGLT1 expressed in CHO cells assessed as inhibition of [14C]-alpha-methyl-D-glucopyranoside transport after 60 mins by scintillation counting, IC50 = 0.37 μM. | 21398124 | ||
| CHO | Function assay | 60 mins | Inhibition of SGLT6 expressed in CHO cells assessed as inhibition of [14C]-alpha-methyl-D-glucopyranoside transport after 60 mins by scintillation counting, IC50 = 0.38 μM. | 21398124 | ||
| CHO | Function assay | 2 hrs | Inhibition of human SGLT2 expressed in CHO cells assessed as [14C]-alpha-methyl-D-glucopyranoside uptake after 2 hrs by liquid scintillation counter, IC50 = 0.00049 μM. | 21514014 | ||
| CHO | Function assay | 60 mins | Inhibition of human SGLT2 expressed in CHO cells assessed as inhibition of [14C]-Glucose uptake using [14C]-methyl glucopyranoside after 60 mins by microbeta plate counting, IC50 = 0.003 μM. | 21565497 | ||
| CHO | Function assay | Inhibition of human SGLT2 expressed in CHO cells using methyl-alpha-D-glucopyranoside by liquid scintillation counting, IC50 = 0.00135 μM. | 21592794 | |||
| COS7 | Function assay | 2 hrs | Inhibition of human SGLT2 expressed in african green monkey COS7 cells assessed as inhibition of [14C]-methyl-alpha-D-glucopyranoside uptake after 2 hrs by scintillation counting in presence of 25 % human plasma, IC50 = 0.0032 μM. | 21737266 | ||
| 293 | Function assay | 1.5 hrs | Inhibition of human SGLT1 expressed in human 293 cells assessed as inhibition of [14C]-methyl-alpha-D-glucopyranoside uptake after 1.5 hrs by scintillation counting in presence of 25 % human plasma, IC50 = 3.1 μM. | 21737266 | ||
| CHO | Function assay | 2 hrs | Inhibition of human SGLT2 expressed in CHO cells assessed as reduction of [14C]-labeled AMG after 2 hrs by liquid scintillation counting, IC50 = 0.00135 μM. | 21868239 | ||
| CHO | Function assay | 2 hrs | Inhibition of human SGLT2 expressed in CHO cells assessed as [14C]-alpha-methyl-D-glucopyranoside uptake after 2 hrs by liquid scintillation counting, IC50 = 0.00049 μM. | 21906953 | ||
| CHO-K1 | Function assay | Inhibition of human SGLT2 expressed in CHO-K1 cells by [14C]AMG uptake assay, IC50 = 0.0013 μM. | 22652255 | |||
| CHO-K1 | Function assay | Inhibition of human SGLT1 expressed in CHO-K1 cells by [14C]AMG uptake assay, IC50 = 0.8 μM. | 22652255 | |||
| CHO-K1 | Function assay | 120 mins | Inhibition of human SGLT2 expressed in CHO-K1 cells incubated for 120 mins at 37 degC by [14C]-AMG uptake assay, EC50 = 0.0024 μM. | 22818040 | ||
| CHO-K1 | Function assay | 120 mins | Inhibition of human SGLT1 expressed in CHO-K1 cells incubated for 120 mins at 37 degC by [14C]-AMG uptake assay, EC50 = 0.593 μM. | 22818040 | ||
| CHO | Function assay | 45 mins | Inhibition of human SGLT2 expressed in CHO cells assessed as inhibition of sodium-dependent [14C]methyl-alpha-D-glucopyranoside uptake after 45 mins, IC50 = 0.0013 μM. | 22889351 | ||
| CHO | Function assay | 45 mins | Inhibition of human SGLT1 expressed in CHO cells assessed as inhibition of sodium-dependent [14C]methyl-alpha-D-glucopyranoside uptake after 45 mins, IC50 = 0.8 μM. | 22889351 | ||
| CHOK1 | Function assay | 3 hrs | Inhibition of human SGLT1 expressed in CHOK1 cells assessed as inhibition of [14C]-AMG uptake after 3 hrs by microbeta scintillation counting analysis, IC50 = 0.891 μM. | 24842618 | ||
| CHO | Function assay | 2 hrs | Inhibition of human SGLT2 expressed in CHO cells assessed as [14C]-alpha-methyl-D-glucopyranoside uptake after 2 hrs by liquid scintillation counting, IC50 = 0.00049 μM. | 24900297 | ||
| CHO | Function assay | 2 hrs | Competitive inhibition of full-length human SGLT2 expressed in CHO cells assessed as inhibition of sodium-dependent [14C]-alpha-methyl-D-glucopyranoside uptake after 2 hrs by scintillation counting, IC50 = 0.005 μM. | 25650254 | ||
| CHO | Function assay | 120 mins | Inhibition of human SGLT2 expressed in CHO cells assessed as decrease in uptake of [14C]AMG after 120 mins by TopCount method, IC50 = 0.0015 μM. | 28447791 | ||
| CHO | Function assay | 120 mins | Inhibition of human SGLT1 expressed in CHO cells assessed as decrease in uptake of [14C]AMG after 120 mins by TopCount method, IC50 = 0.629 μM. | 28447791 | ||
| CHO | Function assay | 1 hr | Inhibition of human SGLT2 expressed in CHO cells assessed as reduction in [14C]AMG uptake after 1 hr by microbeta counting method, EC50 = 0.002 μM. | 29216560 | ||
| CHO | Function assay | 1 hr | Inhibition of human SGLT1 expressed in CHO cells assessed as reduction in [14C]AMG uptake after 1 hr by microbeta counting method, EC50 = 0.86 μM. | 29216560 | ||
| CHO | Function assay | 1 hr | Inhibition of full-length human SGLT2 expressed in CHO cells assessed as decrease in [14C]-methyl alpha-D-glucopyranoside uptake after 1 hr by liquid scintillation counting method, IC50 = 0.00105 μM. | 29764742 | ||
| HEK293 | Function assay | 10 mins | Inhibition of human SGLT2 expressed in HEK293 cells assessed as decrease in [14C]-AMG uptake preincubated for 10 mins followed by [14C]-AMG addition and measured after 2 hrs by liquid scintillation counting method, IC50 = 0.0014 μM. | 29954682 | ||
| HEK293 | Function assay | 10 mins | Inhibition of human SGLT1 expressed in HEK293 cells assessed as decrease in [14C]-AMG uptake preincubated for 10 mins followed by [14C]-AMG addition and measured after 2 hrs by liquid scintillation counting method, IC50 = 1.83 μM. | 29954682 | ||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(244.57 mM)
Ethanol : 100 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 408.87 | Formula | C21H25ClO6 |
Storage (From the date of receipt) | 3 years -20°C powder (seal) |
|---|---|---|---|---|---|
| CAS No. | 461432-26-8 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | BMS-512148 | Smiles | CCOC1=CC=C(C=C1)CC2=C(C=CC(=C2)C3C(C(C(C(O3)CO)O)O)O)Cl | ||
Mechanism of Action
| Features |
More potent stimulator of glucosuria than other SGLT2 inhibitors.
|
|---|---|
| Targets/IC50/Ki |
hSGLT2
(CHO cells) 1.1 nM(EC50)
|
| In vitro |
Dapagliflozin is not sensitive to hSGLT1 with a 1200-fold IC50. This compound is 32-fold more potent than phlorizin against hSGLT2 but 4-fold less than phlorizin against hSGLT1. It is highly selective versus GLUT transporters and displays 8–9% inhibition in protein-free buffer at 20 μM and virtually no inhibition in the presence of 4% bovine serum albumin. This chemical has good permeability across Caco-2 cell membranes and is a substrate for P-glycoprotein (P-gp) but not a significant P-gp inhibitor. It is stable in rat, dog, monkey, and human serum at 10 μM. It shows no inhibitory responses or induction to human P450 enzymes. The in vitro metabolic pathways of this compound are glucuronidation, hydroxylation, and O-deethylation. |
| Kinase Assay |
SGLT Binding Assays
|
|
Chinese hamster ovary (CHO) cells stably expressing human SGLT2 (hSGLT2) and human SGLT1 (\hSGLT1) are utilized for the development of transport assays using the selective SGLT substrate α-methyl-D-glucopyranoside (AMG). Dapagliflozin is assayed for the ability to inhibit [14C]AMG uptake in a protein- free buffer over a 2 hours incubation period. The response curve is fitted to an empirical four-parameter model to determine the inhibitor concentration at half maximal response, reported as EC50. Protein-free buffer is used to simulate the low-protein conditions of the glomerular filtrate, which bathes the SGLT targets on the lumenal surface of the proximal tubule in the kidney.
|
|
| In vivo |
Dapagliflozin reduces blood glucose levels by 55% after 0.1 mg/kg oral dose in hyperglycemic streptozotocin (STZ) rats, which is in part to the metabolic stability conferred by the C-glucoside linkage. This compound displays a favorable absorption, distribution, metabolism, and excretion (ADME) profile and is orally bioavailable. This chemical (1 mg/kg) causes significant dose-dependent glucosuria and increase in urine volume in normal rats over 24 hours post-dose. It induces increase in urine glucose and urine volume excretion at 6 hours post-dose in Zucker diabetic fatty (ZDF) rats. This agent lowers fasting and fed glucose levels in ZDF rats even by 2 weeks of treatment, without any marker of renal or liver toxicity. It significantly reduces the development of hyperglycaemia, with lowered blood glucose. This compound could improve the insulin sensitivity, reduce β-cell mass and the development of impaired pancreatic function. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
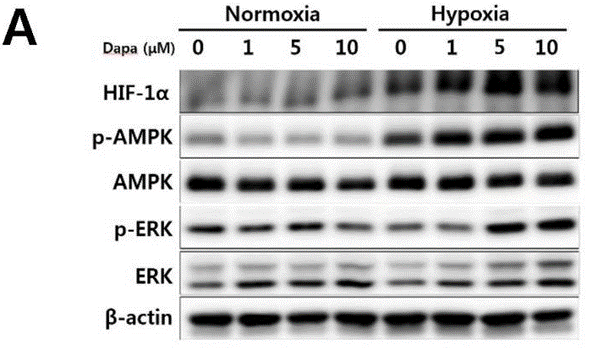
| Western blot | HIF-1α / p-AMPK / AMPK / p-ERK / ERK / β-actin DDR1 HIF-1α / PHD2 / tubulin p-elf2α / t-elf2α / tubulin-α Bax / Bcl2 / PARP / β-actin |
|
27391020 |
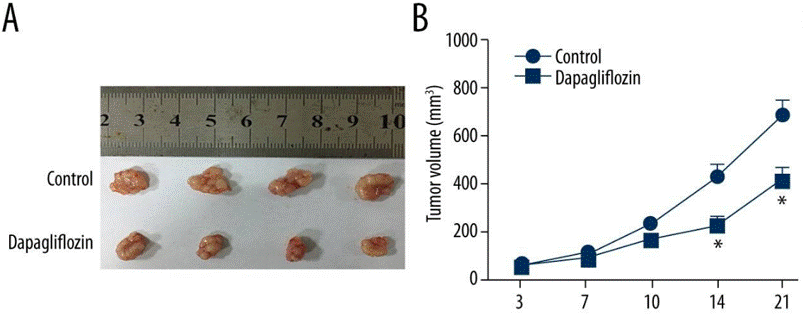
| Growth inhibition assay | Tumor volume |
|
28763435 |
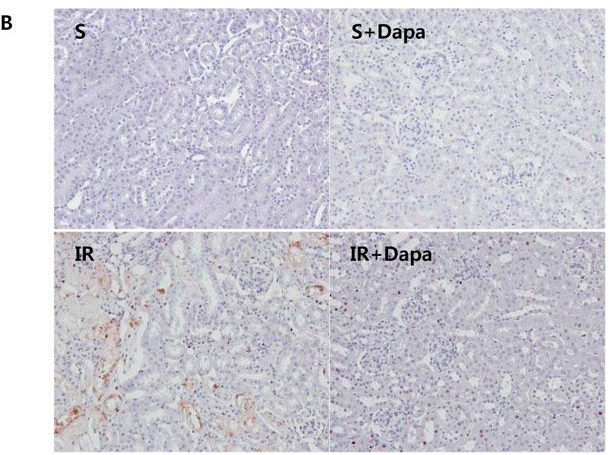
| IHC | TUNEL-positive cells HIF1 TUNEL-positive cells mice kidney sections HE staining / SGLT2 |
|
27391020 |
| Immunofluorescence | Metabolic switch in diabetic kidneys F-actin EdU PECAM-1 / α-SMA podocytes |
|
32444604 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06012266 | Not yet recruiting | Heart Failure |
Centre Hospitalier Universitaire Vaudois|Great Ormond Street Hospital for Children NHS Foundation Trust|University College London |
August 2024 | Phase 2 |
| NCT06336330 | Recruiting | Heart Diseases|Cardiovascular Diseases|Heart Failure |
AstraZeneca |
April 25 2024 | -- |
| NCT04887935 | Not yet recruiting | Prostate Cancer|Cancer of Prostate |
Washington University School of Medicine|The Foundation for Barnes-Jewish Hospital |
April 30 2024 | Phase 1 |
| NCT06398977 | Recruiting | Peritoneal Dialysis Complication|Renal Function Aggravated|Sodium-glucose Co-transporter-2 Inhibitors |
Sichuan Academy of Medical Sciences |
March 11 2024 | Not Applicable |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































