research use only
Ibrutinib (PCI-32765) Btk inhibitor
Cat.No.S2680
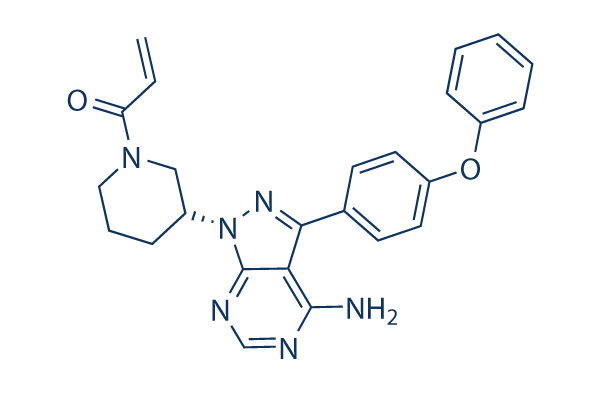
Chemical Structure
Molecular Weight: 440.5
Quality Control
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| human Rec1 cells | Function assay | 2.5 μM | 6 h | Inhibition of Lyn phosphorylation in human Rec1 cells at 2.5 uM incubated for 6 hrs by Western blotting method | 25222877 | |
| human WSU-NHL cells | Cytotoxic assay | 72 h | Cytotoxicity against human WSU-NHL cells assessed as growth inhibition after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50=1.09 μM | 24915291 | ||
| human SU-DHL6 cells | Cytotoxic assay | 72 h | Cytotoxicity against human SU-DHL6 cells assessed as growth inhibition after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50=0.58 μM. | 24915291 | ||
| human DOHH2 cells | Cytotoxic assay | 72 h | Cytotoxicity against human DOHH2 cells assessed as growth inhibition after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50=0.41 μM. | 24915291 | ||
| Sf9 cells | Function assay | 1 h | Inhibition of LYN-A expressed in Sf9 cells after 60 mins by TR-FRET Assay, IC50=0.2 μM. | 21958547 | ||
| human Ramos cells | Function assay | 1 h | Inhibition of Btk in human Ramos cells assessed as inhibition of PLC-gamma2 phosphorylation at Tyr1217 after 1 hr by Western blot analysis, IC50=14 nM. | 24915291 | ||
| human Pfeiffer cells | Function assay | 72 h | Cytotoxicity against human Pfeiffer cells assessed as growth inhibition after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50=2 nM. | 24915291 | ||
| Sf9 cells | Function assay | 1 h | Inhibition of human full-length BTK expressed in Sf9 cells using FAM-Srctide peptide as substrate after 60 mins by TR-FRET Assay, IC50=0.5 nM. | 21958547 | ||
| Sf9 insect cells | Function assay | 60 mins | IC50 = 0.0003 μM | 29146136 | ||
| sf9 cells | Function assay | IC50 = 0.0003 μM | 27994736 | |||
| Sf9 insect cells | Function assay | 60 mins | IC50 = 0.00034 μM | 27912175 | ||
| Sf9 insect cells | Function assay | 60 mins | IC50 = 0.00034 μM | 28432946 | ||
| Sf9 insect cells | Function assay | 60 mins | IC50 = 0.00034 μM | 27956037 | ||
| Sf9 cells | Function assay | 60 mins | IC50 = 0.0004 μM | 30006143 | ||
| Sf9 cells | Function assay | 60 mins | IC50 = 0.0005 μM | 21958547 | ||
| Ramos cells | Function assay | 1 h | IC50 = 0.0005 μM | 28280261 | ||
| Sf9 cells | Function assay | 60 mins | IC50 = 0.001 μM | 30077608 | ||
| Pfeiffer cells | Cytotoxicity assay | 72 h | GI50 = 0.002 μM | 24915291 | ||
| B cells | Function assay | 1 h | IC50 = 0.0046 μM | 30290988 | ||
| Sf9 insect cells | Function assay | 2 to 60 mins | Ki = 0.0048 μM | 28315597 | ||
| Ramos cells | Function assay | 1 h | IC50 = 0.0075 μM | 24915291 | ||
| Sf9 insect cells | Function assay | 60 mins | IC50 = 0.008 μM | 28315597 | ||
| TMD8 cells | Antiproliferative activity assay | 72 h | IC50 = 0.01 μM | 29715023 | ||
| CD19+ B cells | Function assay | 1 h | IC50 = 0.012 μM | 29457982 | ||
| Sf9 insect cells | Function assay | 60 mins | IC50 = 0.012 μM | 28315597 | ||
| Ramos cells | Function assay | 1 h | IC50 = 0.014 μM | 24915291 | ||
| Sf9 insect cells | Function assay | 1 h | IC50 = 0.0144 μM | 29715023 | ||
| Sf9 insect cells | Function assay | 60 mins | IC50 = 0.0161 μM | 29146136 | ||
| HCC827 cells | Antiproliferative activity assay | 72 h | IC50 = 0.039 μM | 28734581 | ||
| PC9 cells | Function assay | 72 h | GI50 = 0.05 μM | 28282122 | ||
| Sf9 cells | Function assay | 60 mins | IC50 = 0.1 μM | 30006143 | ||
| H3255 cells | Function assay | 72 h | GI50 = 0.11 μM | 28282122 | ||
| BaF3 cells | Function assay | 72 h | GI50 = 0.12 μM | 26630553 | ||
| BAF3 cells | Antiproliferative activity assay | 72 h | GI50 = 0.12 μM | 28956923 | ||
| Sf9 insect cells | Function assay | 60 mins | IC50 = 0.123 μM | 28315597 | ||
| Sf9 insect cells | Function assay | 60 mins | IC50 = 0.146 μM | 28315597 | ||
| BAF3 cells | Function assay | 72 h | GI50 = 0.16 μM | 28282122 | ||
| Sf9 cells | Function assay | 60 mins | IC50 = 0.2 μM | 21958547 | ||
| MV411 cells | Growth inhibition assay | GI50 = 0.25 μM | 28315597 | |||
| MV4-11 cells | Antiproliferative activity assay | 72 h | GI50 = 0.33 μM | 26630553 | ||
| MV4-11 cells | Antiproliferative activity assay | 72 h | GI50 = 0.33 μM | 28956923 | ||
| DOHH2 cells | Cytotoxicity assay | 72 h | GI50 = 0.41 μM | 24915291 | ||
| HCC827 cells | Antiproliferative activity assay | 96 h | EC50 = 0.45 μM | 28853575 | ||
| SU-DHL6 cells | Cytotoxicity assay | 72 h | GI50 = 0.58 μM | 24915291 | ||
| M07e cells | Growth inhibition assay | GI50 = 0.59 μM | 28315597 | |||
| NCI-H1975 cells | Antiproliferative activity assay | 96 h | EC50 = 0.64 μM | 28853575 | ||
| SU-DHL-2 cells | Growth inhibition assay | GI50 = 0.64 μM | 28315597 | |||
| HEK293T cells | Function assay | 1 h | IC50 = 0.9 μM | 28280261 | ||
| Ramos cells | Antiproliferative activity assay | 72 h | IC50 = 0.92 μM | 29715023 | ||
| BA/F3 cells | Antiproliferative activity assay | 72 h | IC50 = 1 μM | 26258521 | ||
| WSU-NHL cells | Cytotoxicity assay | 72 h | GI50 = 1.09 μM | 24915291 | ||
| NCI-H1975 cells | Function assay | 72 h | GI50 = 1.2 μM | 28282122 | ||
| NCI-H1975 cells | Antiproliferative activity assay | 72 h | IC50 = 1.27 μM | 28734581 | ||
| Raji cells | Antiproliferative activity assay | 48 h | IC50 = 1.49 μM | 29567295 | ||
| Pfeiffer cells | Growth inhibition assay | GI50 = 1.6 μM | 28315597 | |||
| A431 cells | Antiproliferative activity assay | 96 h | EC50 = 2.38 μM | 28853575 | ||
| BAF3 cells | Antiproliferative activity assay | 72 h | GI50 = 2.5 μM | 28956923 | ||
| U937 cells | Growth inhibition assay | GI50 = 2.9 μM | 28315597 | |||
| NB4 cells | Growth inhibition assay | GI50 = 3 μM | 28315597 | |||
| Ramos cells | Growth inhibition assay | GI50 = 3.4 μM | 28315597 | |||
| SKM1 cells | Growth inhibition assay | GI50 = 3.6 μM | 28315597 | |||
| U2932 cells | Growth inhibition assay | GI50 = 4.4 μM | 28315597 | |||
| Ramos cells | Antiproliferative activity assay | 72 h | IC50 = 5.14 μM | 30006143 | ||
| Ramos cells | Antiproliferative activity assay | 48 h | IC50 = 5.14 μM | 27956037 | ||
| Ramos cells | Antiproliferative activity assay | 48 h | IC50 = 5.88 μM | 28432946 | ||
| Ramos cells | Antiproliferative activity assay | 72 h | IC50 = 6.62 μM | 29146136 | ||
| K562 cells | Antiproliferative activity assay | 48 h | IC50 = 7.5 μM | 30077608 | ||
| HL60 cells | Antiproliferative activity assay | 48 h | IC50 = 8 μM | 30077608 | ||
| Ramos cells | Antiproliferative activity assay | 48 h | IC50 = 8.11 μM | 27994736 | ||
| Ramos cells | Antiproliferative activity assay | 48 h | IC50 = 8.26 μM | 29567295 | ||
| OCI-AML3 cells | Growth inhibition assay | GI50 = 9.2 μM | 28315597 | |||
| BAF3 cells | Cytotoxicity assay | 72 h | GI50 = 10 μM | 26630553 | ||
| BAF3 cells | Antiproliferative activity assay | 72 h | GI50 = 10 μM | 28956923 | ||
| BAF3 cells | Growth inhibition assay | 72 h | GI50 = 10 μM | 28282122 | ||
| NAMALWA cells | Antiproliferative activity assay | 72 h | IC50 = 10.45 μM | 29146136 | ||
| Ramos cells | Antiproliferative activity assay | 48 h | IC50 = 12.6 μM | 27912175 | ||
| Raji cells | Antiproliferative activity assay | 48 h | IC50 = 14.2 μM | 30077608 | ||
| Raji cells | Antiproliferative activity assay | 48 h | IC50 = 15.2 μM | 27994736 | ||
| MIAPaCa2 cells | Cytotoxicity assay | 3 days | IC50 = 16.6 μM | 27077228 | ||
| HeLa cells | Cytotoxicity assay | 3 days | IC50 = 16.8 μM | 27077228 | ||
| Raji cells | Antiproliferative activity assay | 48 h | IC50 = 19.3 μM | 27912175 | ||
| Raji cells | Antiproliferative activity assay | 48 h | IC50 = 19.3 μM | 28432946 | ||
| Raji cells | Antiproliferative activity assay | 72 h | IC50 = 19.5 μM | 30006143 | ||
| Raji cells | Antiproliferative activity assay | 48 h | IC50 = 19.5 μM | 27956037 | ||
| NAMALWA cells | Antiproliferative activity assay | 72 h | IC50 = 19.6 μM | 30006143 | ||
| A2780 cells | Cytotoxicity assay | 3 days | EC50 = 20.1 μM | 27077228 | ||
| Raji cells | Antiproliferative activity assay | 72 h | IC50 = 20.88 μM | 29146136 | ||
| A549 cells | Antiproliferative activity assay | 72 h | IC50 = 21.79 μM | 28734581 | ||
| SW480 cells | Cytotoxicity assay | 3 days | IC50 = 25.6 μM | 27077228 | ||
| Ramos cells | Cytotoxicity assay | 24 h | IC50 = 28.7 μM | 28274675 | ||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 88 mg/mL
(199.77 mM)
Ethanol : 8 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 440.5 | Formula | C25H24N6O2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 936563-96-1 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | PCI-32765 | Smiles | C=CC(=O)N1CCCC(C1)N2C3=NC=NC(=C3C(=N2)C4=CC=C(C=C4)OC5=CC=CC=C5)N | ||
Mechanism of Action
| Targets/IC50/Ki |
BTK
(Cell-free assay) 0.5 nM
BLK
(Cell-free assay) 0.5 nM
Bmx
(Cell-free assay) 0.8 nM
CSK
(Cell-free assay) 2.3 nM
FGR
(Cell-free assay) 2.3 nM
BRK
(Cell-free assay) 3.3 nM
Hck
(Cell-free assay) 3.7 nM
EGFR
(Cell-free assay) 5.6 nM
YES
(Cell-free assay) 6.5 nM
ErbB2
(Cell-free assay) 9.4 nM
ITK
(Cell-free assay) 10.7 nM
JAK3
(Cell-free assay) 16.1 nM
FRK
(Cell-free assay) 29.2 nM
LCK
(Cell-free assay) 33.2 nM
RET
(Cell-free assay) 36.5 nM
|
|---|---|
| In vitro |
Ibrutinib shows the potent and irreversible inhibitory effect and selectivity for Btk enzymatic activity. In BCR pathway-activated DOHH2 cell line, this compound inhibits autophosphorylation of Btk, phosphorylation of Btk's physiological substrate PLCγ, and phosphorylation of further downstream kinase, ERK with IC50 of 11 nM, 29 nM and 13 nM, respectively. It exhibits a significant dose-dependent and time-dependent induction of cytotoxicity in chronic lymphocytic leukemia (CLL) cells. In addition, this compound induces cell death depending on caspase pathway activation and antagonizes the ability of CLL cells to proliferate after TLR signaling. A recent study shows that this chemical inhibits BCR-activated primary B cell proliferation with IC50 of 8 nM and results in inhibition of TNFα, IL-1β and IL-6 production in primary monocytes with IC50 of 2.6 nM, 0.5 nM and 3.9 nM, respectively. |
| Kinase Assay |
Kinase Assays
|
|
In vitro kinase IC50 values are measured using 33P filtration binding assay after 1 hour incubation of kinase, 33P-ATP, Ibrutinib, and substrate [0.2 mg/mL poly(EY)(4:1]. Assays are performed at Reaction Biology.
|
|
| In vivo |
In a collagen-induced arthritis model, Ibrutinib significantly reduces clinical arthritis scores reflecting paw swelling and joint inflammation by inhibiting B-cell activation. In a MRL-Fas(lpr) lupus model, this compound reduces renal disease and autoantibody production. In an adoptive transfer TCL1 mouse model of CLL, this compound (25 mg/kg/day) causes a transient early lymphocytosis, and delays CLL disease progression. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | pEGFR(Tyr1068) / EGFR pBTK / pPLCγ2 / pAKT / pERK / pJNK |
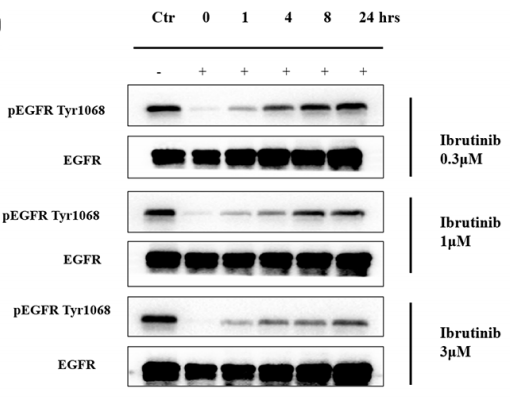
|
28061447 |
| Immunofluorescence | CD11b COX-2 |
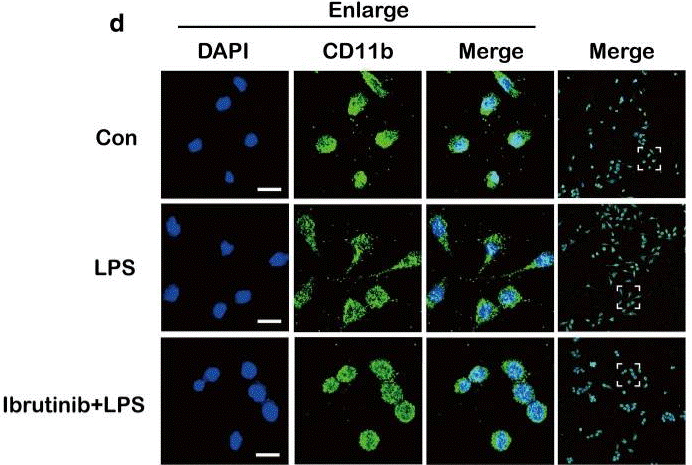
|
30231870 |
| ELISA | hTNFα IL-10 |
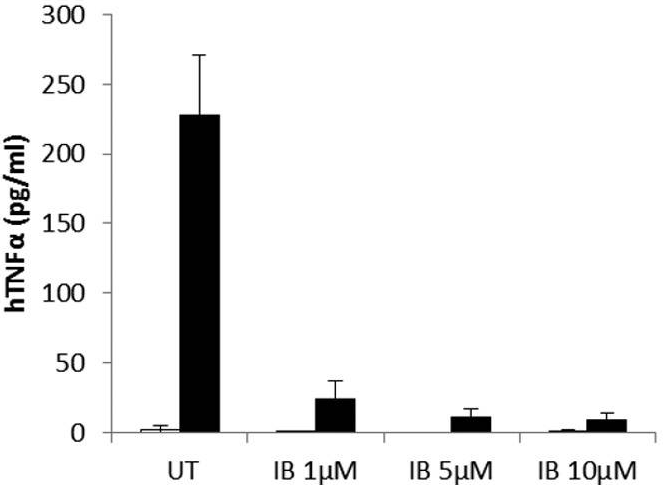
|
26627823 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06084923 | Not yet recruiting | Chronic Lymphocytic Leukemia |
Gruppo Italiano Malattie EMatologiche dell''Adulto |
May 2024 | -- |
| NCT06224452 | Not yet recruiting | Hematological Malignancy|Atrial Fibrillation |
University Hospital Caen |
March 1 2024 | -- |
| NCT05694312 | Recruiting | Autoimmune Hemolytic Anemia|Chronic Lymphocytic Leukemia|Small Lymphocytic Lymphoma|Monoclonal B-Cell Lymphocytosis CLL-Type |
Gruppo Italiano Malattie EMatologiche dell''Adulto |
November 24 2023 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
How to reconstitute it for in vivo studies?
Answer:
For in vivo study, we suggest to use 5% DMSO+30% PEG 300+5% Tween 80+ddH2O up to 10mg/ml for it.






































