research use only
Anastrozole Aromatase inhibitor
Cat.No.S1188
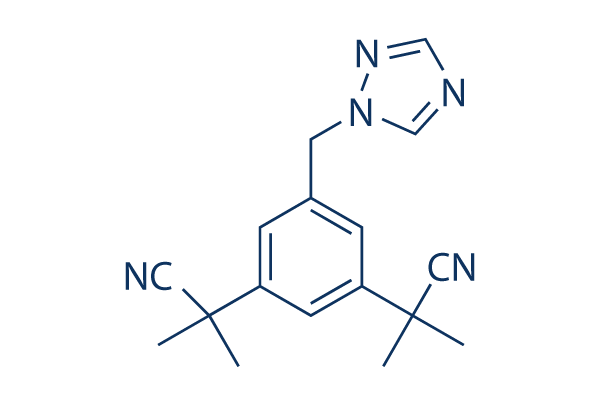
Chemical Structure
Molecular Weight: 293.37
Quality Control
| Related Targets | Adrenergic Receptor Estrogen/progestogen Receptor GPR Androgen Receptor Glucocorticoid Receptor ACE RAAS Progesterone Receptor Opioid Receptor PGES |
|---|---|
| Other Aromatase Inhibitors | Obacunone (AI3-37934) Fadrozole (CGS16949A) alpha-Naphthoflavone |
Solubility
|
In vitro |
DMSO
: 59 mg/mL
(201.11 mM)
Ethanol : 59 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 293.37 | Formula | C17H19N5 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 120511-73-1 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | ZD-1033 | Smiles | CC(C)(C#N)C1=CC(=CC(=C1)CN2C=NC=N2)C(C)(C)C#N | ||
Mechanism of Action
| Targets/IC50/Ki |
Aromatase
(human placental microsomes) 15 nM
|
|---|---|
| In vitro |
Anastrozole has high intrinsic potency in vitro and inhibits human placental aromatase with an IC50 of 15 nM. This compound is 200 times as potent as AG, twice as potent as 4-OHA and one third as potent as fadrozole. . This chemical inhibits aromatase by binding competitively to the hem group of the CYP unit of the enzyme thereby reducing estrogen biosynthesis in the periphery and the breast. It has little or no effect on other steroid hormones.
|
| In vivo |
Anastrozole (0.1 mg/kg) given orally to mature rats on Day 2 or 3 of the estrous cycle, blocked ovulation in mature females; in pubertal rats, the same dose given for 3 days completely blocks androstenedione-stimulated uterine development. These effects may be attributes to inhibition of the normal preovulatory rise in ovarian follicular estrogen synthesis in mature females and to inhibition of metabolism of the exogenous androstenedione by the immature ovary in prepubertal females. Twice-daily administration of >9.1 mg/kg of this compound given orally to male pigtailed monkeys inhibits peripheral aromatase, thereby reducing circulating estradiol concentrations by 50% to 60%.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06154590 | Not yet recruiting | Metastatic Breast Cancer|Tumor|Muscle Neoplasms|Chronic Pain |
DR. DIANE CHISESI NFS. MD. PHD.|IRB|Paradyne Networks A Foundation |
July 2024 | -- |
| NCT04714619 | Terminated | Advanced Breast Cancer |
MedSIR|Cellestia Biotech AG |
May 6 2021 | Phase 2 |
| NCT04227327 | Active not recruiting | Advanced Breast Cancer |
University of Milano Bicocca |
January 7 2020 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































