research use only
Tropifexor (LJN452) FXR agonist
Cat.No.S8733
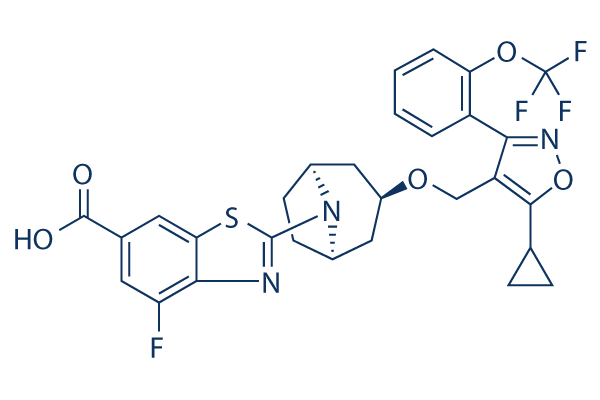
Chemical Structure
Molecular Weight: 603.58
Quality Control
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(165.67 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 603.58 | Formula | C29H25F4N3O5S |
Storage (From the date of receipt) | 3 years -20°C powder |
|---|---|---|---|---|---|
| CAS No. | 1383816-29-2 | -- | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | C1CC1C2=C(C(=NO2)C3=CC=CC=C3OC(F)(F)F)COC4CC5CCC(C4)N5C6=NC7=C(C=C(C=C7S6)C(=O)O)F | ||
Mechanism of Action
| Targets/IC50/Ki |
FXR
(HTRF assay) 0.2 nM(EC50)
|
|---|---|
| In vitro |
In in vitro pharmacological studies, Tropifexor (LJN452) has shown to be a potent human FXR agonist with > 30,000-fold selectivity over other nuclear receptors. It could increase BSEP and SHP expression in primary human hepatocyte. |
| In vivo |
In rat pharmacokinetics, Tropifexor (LJN452) has low clearance (CL = 9 mL/min/kg) and a significantly longer terminal half-life (T1/2 = 3.7 h). Oral bioavailability of this compound in the rat is 20% from an aqueous microemulsion formulation. In the mouse, following IV administration, it exhibits low clearance and small volume of distribution with a half-life of 2.6 h. In the dog, following IV injection, the clearance was low and T1/2 was 7.4 h, also with a small volume of distribution (0.46 L/kg). Preclinical data demonstrate a dose-dependent increase in fibroblast growth factor 19 (FGF-19) levels with it, in single-dose or multiple-dose studies, with its target engagement in enterocyte FXRs. It improves liver transaminases and fibrosis in the ANIT model. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT04408937 | Completed | Liver Disease |
Novartis Pharmaceuticals|Novartis |
May 29 2020 | Phase 1 |
| NCT02855164 | Terminated | Non-alcoholic Steatohepatitis (NASH) |
Novartis Pharmaceuticals|Novartis |
August 1 2016 | Phase 2 |
| NCT02713243 | Completed | Primary Bile Acid Diarrhea |
Novartis Pharmaceuticals|Novartis |
January 16 2016 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































