research use only
Neomycin sulfate Antineoplastic and Immunosuppressive Antibiotics inhibitor
Cat.No.S2568
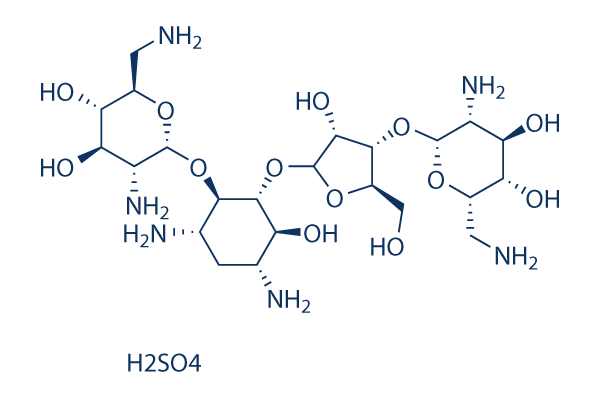
Chemical Structure
Molecular Weight: 712.72
Quality Control
| Related Targets | Integrase Bacterial Antibiotics Anti-infection Fungal Antiviral COVID-19 Parasite Reverse Transcriptase HIV |
|---|---|
| Other Antineoplastic and Immunosuppressive Antibiotics Inhibitors | Staurosporine (STS) Cyclosporin A Oligomycin A (MCH 32) Puromycin Dihydrochloride Nigericin sodium salt Geldanamycin (NSC 122750) Honokiol Streptozotocin (STZ) Sodium Monensin (NSC 343257) Cephalomannine |
Solubility
|
In vitro |
Water : 100 mg/mL
DMSO
: Insoluble
Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 712.72 | Formula | C23H46N6O13.H2SO4 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 28002-70-2 ,1405-10-3 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Framycin sulfate | Smiles | C1C(C(C(C(C1N)OC2C(C(C(C(O2)CN)O)O)N)OC3C(C(C(O3)CO)OC4C(C(C(C(O4)CN)O)O)N)O)O)N.OS(=O)(=O)O | ||
Mechanism of Action
| In vitro |
Neomycin interacts preferentially with the ribozyme-substrate complex and that this interaction leads to a reduction in the cleavage rate by stabilizing the ground state of the complex and destabilizing the transition state of the cleavage step. Neomycin effects a conformational change in the structure of trans-activating region (TAR) that can be detected by circular dichroism spectroscopy. Neomycin acts as a noncompetitive inhibitor that can bind to the Tat-TAR complex and increase the rate constant (koff) for dissociation of the peptide from the RNA. Neomycin is the most effective aminoglycoside (groove binder) in stabilizing a DNA triple helix. Neomycin stabilizes TAT, as well as mixed base DNA triplexes, better than known DNA minor groove binders (which usually destabilize the triplex) and polyamines. Neomycin shows a preference for stabilization of TAT triplets but can also accommodate CGC(+) triplets. Neomycin induces a concentration- and voltage-dependent partial block from both the cytosolic and luminal faces of the channel. Neomycin has a greater affinity for the luminal site of interaction than the cytosolic site: zero-voltage dissociation constants (Kb(0)) are respectively 210.20 nM and 589.70 nM for luminal and cytosolic block. Neomycin also exhibits voltage-dependent relief of block at holding potentials >+60 mV. |
References |
|
|---|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05593601 | Recruiting | Colonization Asymptomatic |
Mahidol University |
November 24 2022 | Phase 4 |
| NCT00795951 | Completed | Dermatitis Contact |
Allerderm |
November 2008 | Phase 4 |
| NCT00058617 | Completed | Epstein-Barr Virus-Related Hodgkin Lymphoma|Epstein-Barr Virus-Related Non-Hodgkin Lymphoma|EBV Positive Plasma Cell Neoplasm |
Baylor College of Medicine|The Methodist Hospital Research Institute|Center for Cell and Gene Therapy Baylor College of Medicine |
January 1996 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































