research use only
Minocycline Hydrochloride Antibiotic
Cat.No.S4226
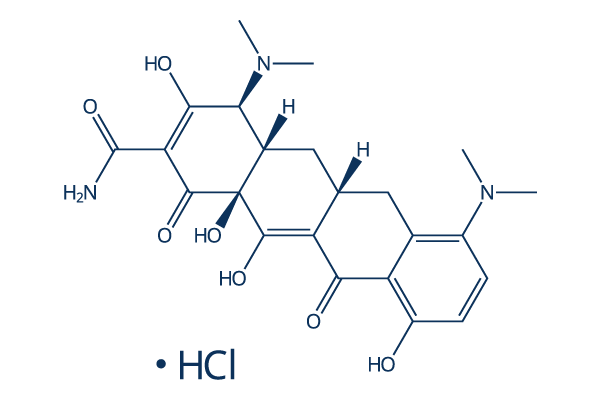
Chemical Structure
Molecular Weight: 493.94
Quality Control
| Related Targets | Integrase Bacterial Anti-infection Fungal Antiviral COVID-19 Parasite Reverse Transcriptase HIV HCV Protease |
|---|---|
| Other Antibiotics Inhibitors | G418 Sulfate (Geneticin) Puromycin Nanchangmycin Sitafloxacin Hydrate Trovafloxacin Mesylate Spiramycin Nadifloxacin G-418 Gamithromycin Thiamphenicol |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| Jurkat cells | Cell viability assay | 10-200 µM | 24 h | reduces cell viability and induces DNA fragmentation and dissipation of the mitochondrial membrane potential in Jurkat cells but is harmless to human peripheral blood lymphocyte cells (hPBLCs) | ||
| hPBLCs | Cell viability assay | 10-200 µM | 24 h | reduces cell viability and induces DNA fragmentation and dissipation of the mitochondrial membrane potential in Jurkat cells but is harmless to human peripheral blood lymphocyte cells (hPBLCs) | ||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
Water : 6 mg/mL
DMSO
: Insoluble
Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 493.94 | Formula | C23H27N3O7.HCl |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 13614-98-7 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | CL 59806 | Smiles | CN(C)C1C2CC3CC4=C(C=CC(=C4C(=C3C(=O)C2(C(=C(C1=O)C(=O)N)O)O)O)O)N(C)C.Cl | ||
Mechanism of Action
| In vitro |
Minocycline is a second-generation tetracycline used in humans, which effectively crosses the blood−brain barrier. It inhibits the activity of caspase-1, caspase-3, inducible form of nitric oxide synthetase (iNOS) and p38 mitogen-activated protein kinase (MAPK). After experimental ischemia, minocycline inhibits caspase-1 and inducible nitric oxide synthetase (iNOS) upregulation, and decreases infarct size. Minocycline inhibits mitochondrial permeability-transition-mediated cytochrome c release. Minocycline-mediated inhibition of cytochrome c release is demonstrated in vivo, in cells, and in isolated mitochondria. |
|---|---|
| In vivo |
Minocycline is a semi-synthetic tetracycline derivative which is well absorbed and distributed in body tissues and is suitable for twice daily administration. Minocycline's effects are related to the inhibition of protein synthesis. Although minocycline's broader spectrum of activity, compared to other members of the group, includes activity against Neisseria meningitidis, its use as a prophylaxis is no longer recomended because of side effects (dizziness and vertigo). Minocycline is remarkable neuroprotective qualities in models of cerebral ischaemia, traumatic brain injury, and Huntington's and Parkinson's disease. The neuroprotective action of minocycline may include its inhibitory effect on 5-lipoxygenase, an inflammatory enzyme associated with brain aging. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
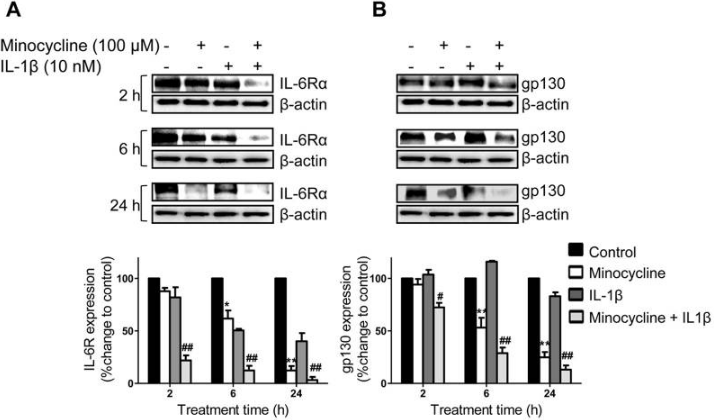
| Western blot | IL-6Rα / gp130 p-STAT3 / STAT3 / Mcl-1 / p-ERK / ERK |
|
23593315 |
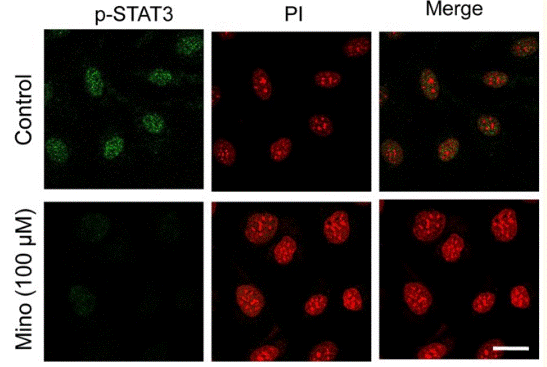
| Immunofluorescence | p-STAT3 STAT3 |
|
23593315 |
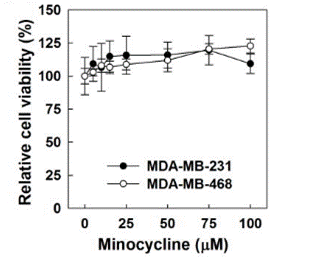
| Growth inhibition assay | Cell viability |
|
27555377 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05605366 | Not yet recruiting | Sickle Cell Disease|Cognitive Impairment|Cognitive Decline|Cognitive Change|Cognitive Dysfunction|Cognitive Deficit|Neuroinflammatory Response |
University of Cincinnati |
June 1 2024 | Phase 1 |
| NCT05861258 | Recruiting | Mycobacterium Avium Complex Pulmonary Disease |
Radboud University Medical Center |
May 8 2023 | Phase 2 |
| NCT05630534 | Not yet recruiting | Intracerebral Hemorrhage |
Second Affiliated Hospital School of Medicine Zhejiang University|The Fourth Affiliated Hospital of Zhejiang University School of Medicine|The Second Affiliated Hospital of Jiaxing University |
January 1 2023 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































