research use only
Alvimopan Opioid Receptor antagonist
Cat.No.S5935
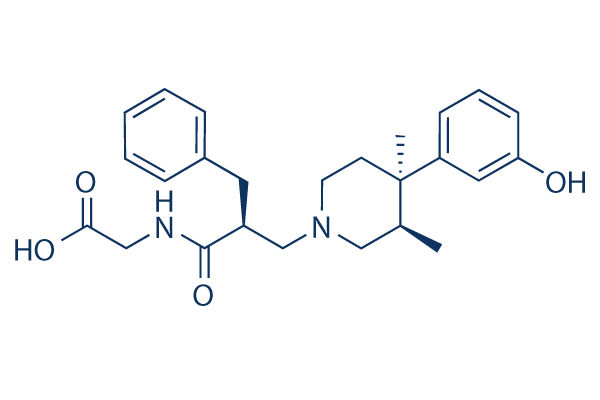
Chemical Structure
Molecular Weight: 424.53
Quality Control
| Related Targets | Adrenergic Receptor AChR 5-HT Receptor COX Calcium Channel Histamine Receptor Dopamine Receptor GABA Receptor TRP Channel Cholinesterase (ChE) |
|---|---|
| Other Opioid Receptor Inhibitors | JTC-801 (+)-Matrine Trimebutine Asimadoline hydrochloride BMS-986122 Racecadotril Aticaprant Trimebutine maleate Naloxegol Oxalate Docusate Sodium |
Solubility
|
In vitro |
DMSO
: 85 mg/mL
(200.22 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 424.53 | Formula | C25H32N2O4 |
Storage (From the date of receipt) | 3 years -20°C powder |
|---|---|---|---|---|---|
| CAS No. | 156053-89-3 | -- | Storage of Stock Solutions |
|
|
| Synonyms | LY 246736, ADL 8-2698 | Smiles | CC1CN(CCC1(C)C2=CC(=CC=C2)O)CC(CC3=CC=CC=C3)C(=O)NCC(=O)O | ||
Mechanism of Action
| Targets/IC50/Ki |
μ-opioid receptor
(Cell-free assay) 0.77 nM(Ki)
δ-opioid receptor
(Cell-free assay) 4.4 nM
δ-opioid receptor
(Cell-free assay) 4.4 nM(Ki)
κ-opioid receptor
(Cell-free assay) 40 nM(Ki)
|
|---|---|
| In vitro |
Alvimopan is highly selective (by ≥227-fold) for the human μ receptor over the κ subtype, but has a more modest (≥6-fold) μ/δ receptor selectivity. In the guinea pig isolated ileum, this compound is a potent antagonist of morphine, DAMGO or endomorphin-1-induced, and μ opioid receptor-mediated, inhibition of electrically-evoked contractions (pA2 values of 9.6 or 9.7). The δ and κ antagonist potencies of this chemical are lower in the guinea pig ileum (pA2 values of 8.7 and 7.8, respectively). This compound (1 or 10 μM) has no significant affinity for a broad range of non-opioid receptors, ion channels and enzymes at which it has been tested. |
| In vivo |
In animals, alvimopan antagonizes centrally mediated, morphine-induced analgesia only at relatively high doses, with very high plasma concentrations needed to cross the blood-brain barrier. After intravenous administration, this compound is approximately 200-times more potent at blocking peripheral verses central μ-receptors. After oral administration, it is also highly active. In dogs, intravenous administration of this chemical provided dose-dependent increases in peak plasma concentrations and plasma area under the concentration-time curve. However, as a result of poor systemic absorption, oral doses up to 100 mg/kg produced low plasma concentrations (mean Cmax =92.9 ng/ml), which resulted in an oral bioavailability of approximately 0.03%. The half-life of this agent is estimated to be approximately 10 min after intravenous administration in dogs and rabbits. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT03068975 | Terminated | Post Operative Ileus |
University of Puerto Rico|Merck Sharp & Dohme LLC |
September 1 2017 | Phase 4 |
| NCT00259922 | Completed | Bowel Dysfunction|Constipation |
Cubist Pharmaceuticals LLC a subsidiary of Merck & Co. Inc. (Rahway New Jersey USA)|GlaxoSmithKline |
August 2005 | Phase 3 |
| NCT00256932 | Completed | Bowel Dysfunction|Constipation |
Cubist Pharmaceuticals LLC a subsidiary of Merck & Co. Inc. (Rahway New Jersey USA)|GlaxoSmithKline |
August 2005 | Phase 3 |
| NCT00241722 | Completed | Bowel Dysfunction|Constipation |
Cubist Pharmaceuticals LLC a subsidiary of Merck & Co. Inc. (Rahway New Jersey USA)|GlaxoSmithKline |
August 1 2005 | Phase 3 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































