research use only
Pemetrexed Disodium Hydrate DHFR inhibitor
Cat.No.S7785
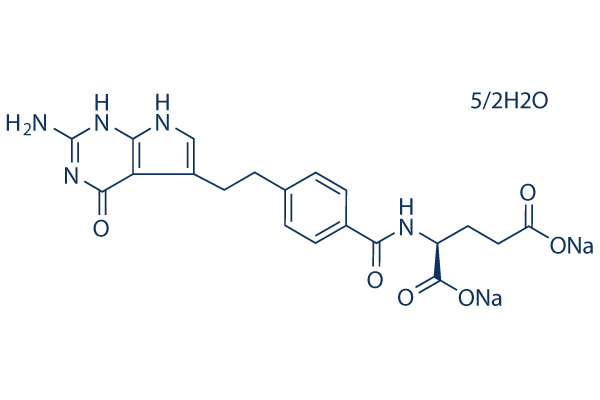
Chemical Structure
Molecular Weight: 516.42
Quality Control
| Related Targets | Dehydrogenase HSP Transferase P450 (e.g. CYP17) PDE phosphatase PPAR Vitamin Carbohydrate Metabolism Mitochondrial Metabolism |
|---|---|
| Other DHFR Inhibitors | Calcium Folinate Aminopterin Diaveridine |
Solubility
|
In vitro |
Water : 100 mg/mL
DMSO
: Insoluble
Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 516.42 | Formula | C20H21N5O6.5/2H2O.2Na |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 357166-30-4 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | LY-231514 Disodium Hydrate | Smiles | C1=CC(=CC=C1CCC2=CNC3=C2C(=O)NC(=N3)N)C(=O)NC(CCC(=O)[O-])C(=O)[O-].C1=CC(=CC=C1CCC2=CNC3=C2C(=O)NC(=N3)N)C(=O)NC(CCC(=O)[O-])C(=O)[O-].O.O.O.O.O.[Na+].[Na+].[Na+].[Na+] | ||
Mechanism of Action
| Targets/IC50/Ki |
TS
1.3 nM(Ki)
DHFR
7.2 nM(Ki)
GARFT
65 nM(Ki)
|
|---|---|
| In vitro |
Pemetrexed disodium hydrate shows the antiproliferative activity in CCRF-CEM leukemia, GC3/C1 colon carcinoma, and HCT-8 ileocecal carcinoma cells with IC50 of 25 nM, 34 nM and 220 nM, respectively. A recent study shows that cisplatin plus this compound combined with SOCS-1 gene delivery shows the antitumor effect by inhibition of cell proliferation, invasiveness, and induction of apoptosis in MPM cells infected with adenovirus-expressing SOCS-1 vector.
|
| Kinase Assay |
Enzyme Assays and Methods.
|
|
TS activity is assayed using a spectrophotometric method, which involved monitoring the increase in absorbance at 340 nm resulting from formation of the product, 7,8-dihydrofolate. The assay buffer contains 50 mM N-tris[hydroxymethyljmethyl-2-aminoethanesulfonic acid, 25 mM MgC12, 6.5 mM formaldehyde, 1 mM EDTA, and 75 mM 2-mercaptoethanol, pH 7.4. The concentrations of deoxyuridylate monophosphate, 6R-MTHF, and hIS are 100 μM, 30μM and 30 nM (1.7 milliunits/mL), respectively. At the 6R-MTHF concentration, an uninhibited reaction and six concentrations of inhibitor are assayed. Ki app values are determined by fitting the data to the Morrison equation using nonlinear regression analysis with the aid of the program ENZFITTER. Ki values are calculated using the equation: Ki app= Ki(1 + [S]/Km), where [S] is equal to 30 μM and Km is equal to 3 μM. DHFR activity is assayed spectrophotometrically by monitoring the dis appearance of the substrates NADPH and 7,8-dihydrofolate at 340 nm. The reaction takes place at 25°C in 0.5 mL of 50 mM potassium phosphate buffer, which contains 150 mM KC1 and 10 nM 2-mercaptoethanol, pH 7.5, and 14 nM (0.34 milliunitlmL) DHFR. The NADPH concentration is 10 μM and 7,8-dihydrofolate is varied at 5, 10, or 15 μM. At each 7,8-dihydrofolate concentration, an uninhibited reaction and seven concentrations of inhibitor are assayed. The ENZFITI'ER microcomputer program is used to obtain Ki app values by fitting the data to the Morrison equation by nonlinear regression analysis. Ki app= Ki(1 + [S]/Km), where [S] is equal to the concentration of 7,8-dihydrofolate used and Km of 7,8-dihydrofolate is equal to 0.15 μM. GARFT activity is assayed spectrophotometrically by monitoring the increase of absorbance resulting from formation of the product 5,8-dideazafolate at 295 nm. The reaction solvent contains 75 mM HEPES, 20% glycerol, and 50 mM a-thioglygerol, pH 7.5, at 25°C. The concentrations of substrates and enzyme used are 10 μM α,β-glycinamide ribonucleotide, 0-10 μM 10-formyl-5,8-dideazafolic acid, and 10 nM (1.9 milliunits/mL) GARFT. Ki values are calculated using the Enzyme Mechanism program of the Beckman DU640 spectrophotometer, which uses nonlinear regression analysis to fit data to the Michaelis-Menten equation for competitive inhibition.
|
|
| In vivo |
In the human H460 non-small cell lung carcinoma xenograft, Pemetrexed disodium hydrate produces a duration-dependent tumor growth delay (TGD).
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | EGFR / p-EGFR AKT / p-AKT / GSK3β / p-GSK3β Topo IIα / Topo I / γH2AX / Cleaved PARP / Survivin p-Chk1 / Chk1 / Cyclin D / Cyclin E / p-Histone H3 / Histone H3 / Cyclin B1 / p-Cdc2 / Cdc2 |
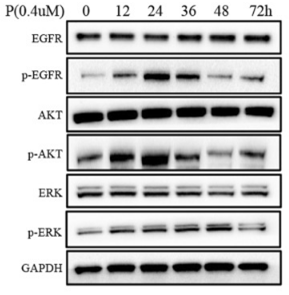
|
30953548 |
| Growth inhibition assay | Cell viability |
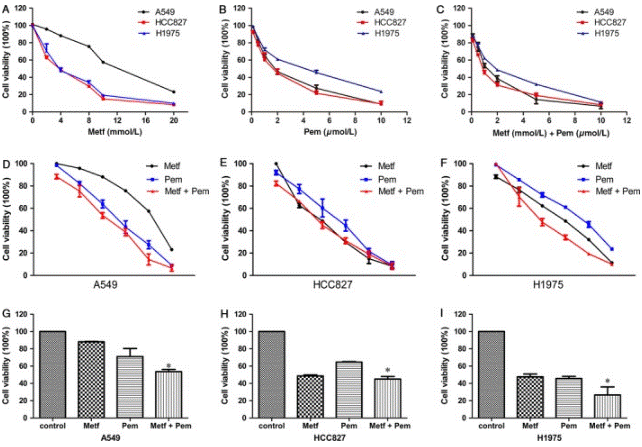
|
28719077 |
| Immunofluorescence | p-AKT |
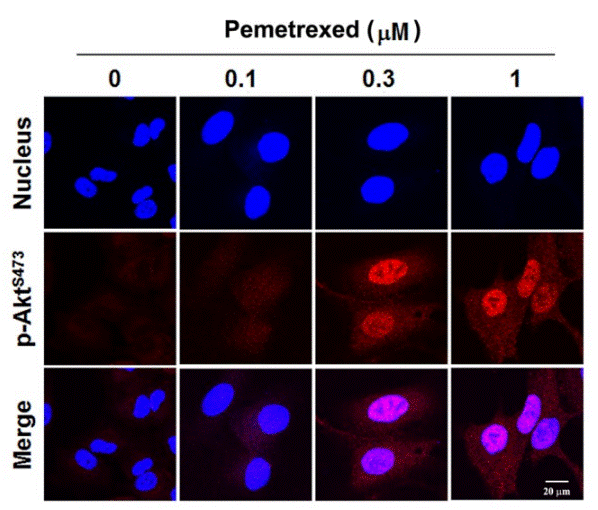
|
24847863 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06378892 | Recruiting | Non Small Cell Lung Cancer Metastatic|ALK Gene Mutation |
Centro di Riferimento Oncologico - Aviano |
March 15 2024 | Phase 2 |
| NCT06010277 | Recruiting | NSCLC|Mesothelioma|Thymoma |
Amphia Hospital|Albert Schweitzer Hospital |
February 6 2023 | Phase 4 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































