research use only
Evacetrapib (LY2484595) CETP inhibitor
Cat.No.S2925
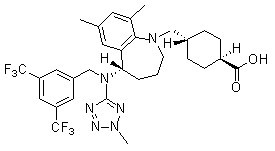
Chemical Structure
Molecular Weight: 638.65
Quality Control
| Related Targets | Dehydrogenase HSP Transferase P450 (e.g. CYP17) PDE phosphatase PPAR Vitamin Carbohydrate Metabolism Mitochondrial Metabolism |
|---|---|
| Other CETP Inhibitors | Anacetrapib (MK-0859) Dalcetrapib (JTT-705) Torcetrapib Obicetrapib |
Solubility
|
In vitro |
DMSO
: 12.8 mg/mL
(20.04 mM)
Ethanol : 12.8 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 638.65 | Formula | C31H36F6N6O2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1186486-62-3 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CC1=CC(=C2C(=C1)C(CCCN2CC3CCC(CC3)C(=O)O)N(CC4=CC(=CC(=C4)C(F)(F)F)C(F)(F)F)C5=NN(N=N5)C)C | ||
Mechanism of Action
| Targets/IC50/Ki |
CETP
5.5 nM
|
|---|---|
| In vitro |
Evacetrapib (LY2484595) inhibits human plasma CETP protein with IC50 of 26 nM. This compound (< 10 μM) does not induce aldosterone or cortisol synthesis in H295R cells.
|
| In vivo |
Evacetrapib (LY2484595) (30 mg/kg, orally) results in 98.4%, 98.6%, and 18.4% inhibition of CETP activity at 4 hours, 8 hours and 24 hours post dose respectively in human ApoAI and CETP double transgenic mice. This compound (30 mg/kg) results in 129.7% increase in HDL-C 8 hours after oral administration. The ED50 values of CETP inhibitory activity 8 hours post oral dosing for this chemical in two dose-response studies are calculated to be 3.5 mg/kg and 4.1 mg/kg respectively. It (< 200 mg/kg) does not increase blood pressure in Zucker diabetic fatty rats.
|
References |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02497391 | Completed | Healthy |
Eli Lilly and Company |
July 2015 | Phase 1 |
| NCT02365558 | Completed | Healthy |
Eli Lilly and Company |
January 2015 | Phase 1 |
| NCT02271425 | Completed | Healthy Volunteers |
Eli Lilly and Company |
October 2014 | Phase 1 |
| NCT02226653 | Completed | Healthy Volunteers |
Eli Lilly and Company |
September 2014 | Phase 1 |
| NCT02156492 | Completed | Healthy Volunteers |
Eli Lilly and Company |
June 2014 | Phase 1 |
| NCT02168803 | Completed | Dyslipidemia |
Eli Lilly and Company |
May 2014 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































