research use only
Vandetanib (ZD6474) VEGFR inhibitor
Cat.No.S1046
Chemical Structure
Molecular Weight: 475.35
Quality Control
| Related Targets | EGFR PDGFR FGFR c-Met Src MEK CSF-1R FLT3 HER2 c-Kit |
|---|---|
| Other VEGFR Inhibitors | SAR131675 SU 5402 Cediranib (AZD2171) Vatalanib (PTK787) 2HCl Anlotinib (AL3818) Dihydrochloride Linifanib (ABT-869) Apatinib (YN968D1) Apatinib (YN968D1) mesylate Ki8751 Semaxanib (SU5416) |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| SN179 | Function Assay | 500 nM | 16 h | increases CXCR4 expression significantly | 25676691 | |
| SN186 | Function Assay | 500 nM | 16 h | increases CXCR4 expression significantly | 25676691 | |
| SN179 | Function Assay | 500 nM | 16 h | enhances the CXCL12 directed migration | 25676691 | |
| SN179 | Function Assay | 500 nM | 16 h | increases basal migration | 25676691 | |
| Jurkat | Growth Inhibition Assay | 72 h | GI50=1.5 ± 0.2 μM | 24681205 | ||
| K-562 | Growth Inhibition Assay | 72 h | GI50=1.8 ± 0.1 μM | 24681205 | ||
| NCTC-2544 | Growth Inhibition Assay | 72 h | GI50=4.6 ± 0.3 μM | 24681205 | ||
| A-431 | Growth Inhibition Assay | 72 h | GI50=2.4 ± 0.3 μM | 24681205 | ||
| SK-N-SH | Growth Inhibition Assay | 0.625-20 μM | 48 h | DMSO | inhibits cell growth in a dose dependent manner | 24399074 |
| SH-SY5Y | Growth Inhibition Assay | 0.625-20 μM | 48 h | DMSO | inhibits cell growth in a dose dependent manner | 24399074 |
| SK-N-SH | Apoptosisi Assay | 5/10/20 μM | 48 h | DMSO | induces apoptosis dose dependently | 24399074 |
| SH-SY5Y | Apoptosisi Assay | 5/10/20 μM | 48 h | DMSO | induces apoptosis dose dependently | 24399074 |
| SK-N-SH | Function Assay | 5/10/20 μM | 48 h | DMSO | induces G1 phase cell cycle arrest | 24399074 |
| SH-SY5Y | Function Assay | 5/10/20 μM | 48 h | DMSO | induces G1 phase cell cycle arrest | 24399074 |
| SK-N-SH | Function Assay | 1/5/10 μM | 48 h | DMSO | inhibits RET phosphorylation | 24399074 |
| SH-SY5Y | Function Assay | 1/5/10 μM | 48 h | DMSO | inhibits RET phosphorylation | 24399074 |
| SK-N-SH | Function Assay | 5/10 μM | 48 h | DMSO | inhibits human NB cell migration | 24399074 |
| SH-SY5Y | Function Assay | 5/10 μM | 48 h | DMSO | inhibits human NB cell migration | 24399074 |
| SK-N-SH | Function Assay | 5/10 μM | 48 h | DMSO | inhibits human NB cell invasion | 24399074 |
| SH-SY5Y | Function Assay | 5/10 μM | 48 h | DMSO | inhibits human NB cell invasion | 24399074 |
| SK-N-SH | Function Assay | 5 μM | 24/48/72 h | DMSO | suppresses the expression of CXCR4 and MMP14 mRNA | 24399074 |
| SH-SY5Y | Function Assay | 5 μM | 24/48/72 h | DMSO | suppresses the expression of CXCR4 and MMP14 mRNA | 24399074 |
| SK-N-SH | Function Assay | 5 μM | 48/72 h | DMSO | suppresses expression of the CXCR4 and MMP14 protein | 24399074 |
| SH-SY5Y | Function Assay | 5 μM | 48/72 h | DMSO | suppresses expression of the CXCR4 and MMP14 protein | 24399074 |
| HMEpC | Growth Inhibition Assay | 1 nM-100 μM | 48 h | DMSO | inhibits cell growth in a dose dependent manner | 24138843 |
| MCF-7 | Growth Inhibition Assay | 1 nM-100 μM | 48 h | DMSO | inhibits cell growth in a dose dependent manner | 24138843 |
| ZR-75-1 | Growth Inhibition Assay | 1 nM-100 μM | 48 h | DMSO | inhibits cell growth in a dose dependent manner | 24138843 |
| MDA-MB-231 | Growth Inhibition Assay | 1 nM-100 μM | 48 h | DMSO | inhibits cell growth in a dose dependent manner | 24138843 |
| MDA-MB-468 | Growth Inhibition Assay | 1 nM-100 μM | 48 h | DMSO | inhibits cell growth in a dose dependent manner | 24138843 |
| T-47-D | Growth Inhibition Assay | 1 nM-100 μM | 48 h | DMSO | inhibits cell growth in a dose dependent manner | 24138843 |
| U251 | Function Assay | 2/4/8 μℳ | 6/12/24 h | DMSO | increases the LC3-II level in a time-dependent and dose-dependent manner | 23799852 |
| U87MG | Function Assay | 2/4/8 μℳ | 6/12/24 h | DMSO | increases the LC3-II level in a time-dependent and dose-dependent manner | 23799852 |
| U251 | Function Assay | 4 μℳ | 2/6/12 h | DMSO | suppresses basal levels of phosphorylation of S6 (S235/236), 4E-BP1 (T37/46), and Akt (S473) in a time-dependent manner | 23799852 |
| U87MG | Function Assay | 4 μℳ | 2/6/12 h | DMSO | suppresses basal levels of phosphorylation of S6 (S235/236), 4E-BP1 (T37/46), and Akt (S473) in a time-dependent manner | 23799852 |
| H1650 | Growth Inhibition Assay | IC50=3.5±1.2 μM | 23274758 | |||
| HUVECs | Growth Inhibition Assay | 72 h | IC50 = 7.1 μmol/L | 22611027 | ||
| KYN-2 | Growth Inhibition Assay | 72 h | IC50 = 8.1 μmol/L | 22611027 | ||
| HuH-7 | Growth Inhibition Assay | 72 h | IC50 = 9.4 μmol/L | 22611027 | ||
| HUVECs | Function Assay | 1/5/10 μM | 1 h | significantly inhibits VEGFR-2 phosphorylation | 22611027 | |
| HAK1-B | Function Assay | 1/5/10 μM | 1 h | suppresses EGFR phosphorylation | 22611027 | |
| UM-22A | Growth Inhibition Assay | 0-6 μM | 72 h | DMSO | inhibits cell growth in a dose dependent manner | 22307735 |
| UM-22B | Growth Inhibition Assay | 0-6 μM | 72 h | DMSO | inhibits cell growth in a dose dependent manner | 22307735 |
| PCI-37A | Growth Inhibition Assay | 0-6 μM | 72 h | DMSO | inhibits cell growth in a dose dependent manner | 22307735 |
| PCI-37B | Growth Inhibition Assay | 0-6 μM | 72 h | DMSO | inhibits cell growth in a dose dependent manner | 22307735 |
| PCI-15B | Growth Inhibition Assay | 0-6 μM | 72 h | DMSO | inhibits cell growth in a dose dependent manner | 22307735 |
| SCC-25 | Growth Inhibition Assay | 0-6 μM | 72 h | DMSO | inhibits cell growth in a dose dependent manner | 22307735 |
| UM-22A | Function Assay | 0-10 μM | 24 h | DMSO | inhibits the activation of the EGFR tyrosine kinase and also decreases the expression of phosphorylated forms of the downstream signaling elements, STAT3 and MAPK | 22307735 |
| UM-22B | Function Assay | 0-10 μM | 24 h | DMSO | inhibits the activation of the EGFR tyrosine kinase and also decreases the expression of phosphorylated forms of the downstream signaling elements, STAT3 and MAPK | 22307735 |
| PCI-15B | Function Assay | 0-10 μM | 24 h | DMSO | inhibits the activation of the EGFR tyrosine kinase and also decreases the expression of phosphorylated forms of the downstream signaling elements, STAT3 and MAPK | 22307735 |
| PCI-37A | Function Assay | 1 μM | 24 h | DMSO | downregulates VEGF production | 22307735 |
| UM-22A | Function Assay | 1 μM | 24 h | DMSO | downregulates VEGF production | 22307735 |
| PCI-15B | Function Assay | 1 μM | 24 h | DMSO | downregulates VEGF production | 22307735 |
| PCI-15B | Invasion Assay | 24 h | DMSO | EC50=558 nM | 22307735 | |
| PCI-37A | Invasion Assay | 24 h | DMSO | EC50=1695 nM | 22307735 | |
| UM-22A | Invasion Assay | 24 h | DMSO | EC50=0.3 nM | 22307735 | |
| SCC-25 | Invasion Assay | 24 h | DMSO | EC50=10 nM | 22307735 | |
| UM-22B | Invasion Assay | 24 h | DMSO | EC50=2424 nM | 22307735 | |
| PCI-37B | Invasion Assay | 24 h | DMSO | EC50=1726 nM | 22307735 | |
| 201T | Function Assay | 2.5 μM | 48 h | DMSO | inhibits phospho-MAPK following EGF | 22258476 |
| 273T | Function Assay | 2.5 μM | 48 h | DMSO | inhibits phospho-MAPK following EGF | 22258476 |
| A549 | Function Assay | 2.5 μM | 48 h | DMSO | inhibits phospho-MAPK following EGF | 22258476 |
| 201T | Function Assay | 1/5/10 μM | 48 h | DMSO | blocks the phosphorylation of Akt induced by VEGFC | 22258476 |
| H2052 | Growth Inhibition Assay | IC50=1.07±0.04 μM | 21970874 | |||
| H2452 | Growth Inhibition Assay | IC50=3.52±1.13 μM | 21970874 | |||
| H28 | Growth Inhibition Assay | IC50=0.32±0.07 μM | 21970874 | |||
| MSTO-211H | Growth Inhibition Assay | IC50=1.42±0.03 μM | 21970874 | |||
| Hth83 | Growth Inhibition Assay | 72 h | DMSO | IC50=3.30 ± 0.66 μM | 21220477 | |
| C643 | Growth Inhibition Assay | 72 h | DMSO | IC50=3.65 ± 1.22 μM | 21220477 | |
| 8505C | Growth Inhibition Assay | 72 h | DMSO | IC50=7.56 ± 1.13 μM | 21220477 | |
| Hth74 | Growth Inhibition Assay | 72 h | DMSO | IC50=8.56 ± 1.01 μM | 21220477 | |
| SW1736 | Growth Inhibition Assay | 72 h | DMSO | IC50=9.05 ± 0.55 μM | 21220477 | |
| Hth7 | Growth Inhibition Assay | 72 h | DMSO | IC50=9.66 ± 0.38 μM | 21220477 | |
| Hth104 | Growth Inhibition Assay | 72 h | DMSO | IC50=±16.98 ± NA μM | 21220477 | |
| HTB3 | Growth Inhibition Assay | 0-20 μM | 24 h | inhibits cell growth in a dose dependent manner | 19220256 | |
| HT1376 | Growth Inhibition Assay | 0-20 μM | 24 h | inhibits cell growth in a dose dependent manner | 19220256 | |
| RT4 | Growth Inhibition Assay | 0-20 μM | 24 h | inhibits cell growth in a dose dependent manner | 19220256 | |
| J82 | Growth Inhibition Assay | 0-20 μM | 24 h | inhibits cell growth in a dose dependent manner | 19220256 | |
| CRL1749 | Growth Inhibition Assay | 0-20 μM | 24 h | inhibits cell growth in a dose dependent manner | 19220256 | |
| T24 | Growth Inhibition Assay | 0-20 μM | 24 h | inhibits cell growth in a dose dependent manner | 19220256 | |
| SUP | Growth Inhibition Assay | 0-20 μM | 24 h | inhibits cell growth in a dose dependent manner | 19220256 | |
| HTB9 | Growth Inhibition Assay | 0-20 μM | 24 h | inhibits cell growth in a dose dependent manner | 19220256 | |
| ACC3 | Growth Inhibition Assay | 0-10 μM | 72 h | inhibits cell growth in a dose dependent manner | 18698025 | |
| ACC2 | Growth Inhibition Assay | 0-10 μM | 72 h | inhibits cell growth in a dose dependent manner | 18698025 | |
| ACCM | Growth Inhibition Assay | 0-10 μM | 72 h | inhibits cell growth in a dose dependent manner | 18698025 | |
| ACC3 | Apoptosisi Assay | 0-10 μM | 72 h | induces apoptosis dose dependently | 18698025 | |
| ACC2 | Apoptosisi Assay | 0-10 μM | 72 h | induces apoptosis dose dependently | 18698025 | |
| ACCM | Apoptosisi Assay | 0-10 μM | 72 h | induces apoptosis dose dependently | 18698025 | |
| EHMES-1 | Growth Inhibition Assay | 72 h | DMSO | IC50=10.6 μM | 18364248 | |
| EHMES-10 | Growth Inhibition Assay | 72 h | DMSO | IC50=0.3 μM | 18364248 | |
| 211H | Growth Inhibition Assay | 72 h | DMSO | IC50=2.2 μM | 18364248 | |
| H28 | Growth Inhibition Assay | 72 h | DMSO | IC50=1.8 μM | 18364248 | |
| H2052 | Growth Inhibition Assay | 72 h | DMSO | IC50=8.0 μM | 18364248 | |
| H2452 | Growth Inhibition Assay | 72 h | DMSO | IC50=5.5 μM | 18364248 | |
| CNE-1 | Growth Inhibition Assay | 0.1-25.6 μM | 48 h | IC50=3.6 μM | 17631646 | |
| CNE-2 | Growth Inhibition Assay | 0.1-25.6 μM | 48 h | IC50=6.2 μM | 17631646 | |
| C666-1 | Growth Inhibition Assay | 0.1-25.6 μM | 48 h | IC50=23.4 μM | 17631646 | |
| CNE-1 | Growth Inhibition Assay | 0.1-25.6 μM | 72 h | IC50=2.3 μM | 17631646 | |
| CNE-2 | Growth Inhibition Assay | 0.1-25.6 μM | 72 h | IC50=3.6 μM | 17631646 | |
| C666-1 | Growth Inhibition Assay | 0.1-25.6 μM | 72 h | IC50=4.86 μM | 17631646 | |
| CNE-1 | Function Assay | 6 μM | 24 h | delays G0/G1 cell cycle progression | 17631646 | |
| CNE-2 | Function Assay | 6 μM | 24 h | delays G0/G1 cell cycle progression | 17631646 | |
| C666-1 | Function Assay | 6 μM | 24 h | delays G0/G1 cell cycle progression | 17631646 | |
| KDR15 | Function assay | Inhibitory activity against VEGF stimulated autophosphorylation of VEGFR2 expressed in KDR15 cells, IC50 = 0.015 μM. | 16302797 | |||
| Sf9 | Function assay | Inhibition of human recombinant histidine-tagged RET (700-1020) expressed in Sf9 cells by ELISA, IC50 = 0.097 μM. | 20409618 | |||
| TPC1 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human TPC1 cells expressing RET/PCT1 after 72 hrs by [3H]thymidine incorporation assay, IC50 = 0.116 μM. | 20409618 | ||
| HEK293 | Function assay | Inhibition of FGFR1/VEGFR2 chimeric construct expressed in HEK293 cells by ELISA, ED50 = 0.15 μM. | 19101155 | |||
| Sf21 | Function assay | 15 mins | Inhibition of recombinant His-tagged human KDR expressed in insect Sf21 cells preincubated for 15 mins followed by substrate addition measured after 20 mins by HTRF assay, IC50 = 0.175 μM. | 26874741 | ||
| umbilical vein endothelial cells | Function assay | Inhibition of VEGF-induced proliferation of human umbilical vein endothelial cells, IC50 = 0.4 μM. | 15743202 | |||
| BA/F3 | Function assay | 48 hrs | Inhibition of KIF5B/RET (unknown origin) expressed in mouse BA/F3 cells assessed as reduction in cell viability after 48 hrs by Cell titre glo-based luminescence assay, IC50 = 0.4 μM. | 26874741 | ||
| BA/F3 | Function assay | 48 hrs | Inhibition of KDR (unknown origin) expressed in mouse BA/F3 cells assessed as reduction in cell viability after 48 hrs by Cell titre glo-based luminescence assay, IC50 = 0.63 μM. | 26874741 | ||
| umbilical vein endothelial cells | Function assay | Inhibition of basic FGF-induced proliferation of human umbilical vein endothelial cells, IC50 = 1.2 μM. | 15743202 | |||
| HL60 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HL60 cells after 72 hrs by MTT assay, IC50 = 1.492 μM. | 26995527 | ||
| 293 | Function assay | Inhibitory activity against VEGFR2 transiently transfected in 293 adenovirus transfected kidney cells by ELISA, IC50 = 1.66 μM. | 16275072 | |||
| 293 | Function assay | Inhibition of VEGFR2 in 293 adenovirus transfected kidney cells by cell-based ELISA assay, IC50 = 1.66 μM. | 16321531 | |||
| HEK293 | Function assay | Inhibition of VEGFR2 phosphorylation in HEK293 cells by cell-based ELISA, IC50 = 1.66 μM. | 16460936 | |||
| HT-29 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HT-29 cells after 72 hrs by MTT assay, IC50 = 1.925 μM. | 26995527 | ||
| DU145 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human DU145 cells after 72 hrs by MTT assay, IC50 = 1.974 μM. | 26995527 | ||
| MGHU3 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MGHU3 cells after 72 hrs by CellTiter-Glo assay, IC50 = 2.5 μM. | 30309671 | ||
| A549 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human A549 cells after 72 hrs by CellTiter-Glo assay, IC50 = 2.5 μM. | 30309671 | ||
| RT112 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human RT112 cells after 72 hrs by CellTiter-Glo assay, IC50 = 2.5 μM. | 30309671 | ||
| A549 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human A549 cells after 72 hrs by MTT assay, IC50 = 2.63 μM. | 26995527 | ||
| CHO | Function assay | Inhibition of VEGFR induced autophosphorylation of human Vascular endothelial growth factor receptor 2 (VEGFR2) transfected in CHO cells, IC50 = 2.673 μM. | 12477352 | |||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MCF7 cells after 72 hrs by MTT assay, IC50 = 3.536 μM. | 26995527 | ||
| PANC1 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human PANC1 cells after 72 hrs by MTT assay, IC50 = 4.107 μM. | 26995527 | ||
| HT-29 | Antiproliferative assay | 10 uM | 72 hrs | Antiproliferative activity against human HT-29 cells at 10 uM after 72 hrs by MTS assay, IC50 = 4.2 μM. | 21353546 | |
| EAhy926 | Antiproliferative assay | 10 uM | 72 hrs | Antiproliferative activity against human EAhy926 cells at 10 uM after 72 hrs by MTS assay, IC50 = 5.1 μM. | 21353546 | |
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells measured after 48 hrs by MTT assay, IC50 = 11.83 μM. | 27688180 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity in human MCF7 cells assessed as inhibition of cell growth incubated for 48 hrs by MTT assay, IC50 = 16.52 μM. | 28942113 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 18.5 μM. | 26741358 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 18.5 μM. | 26475519 | ||
| HT-29 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HT-29 cells measured after 48 hrs by MTT assay, IC50 = 18.95 μM. | 27688180 | ||
| H460 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human H460 cells measured after 48 hrs by MTT assay, IC50 = 37.1 μM. | 27688180 | ||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | |||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for MG 63 (6-TG R) cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for SK-N-SH cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Rh41 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 60 mg/mL
(126.22 mM)
Warmed with 50°C water bath;
Ultrasonicated;
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 475.35 | Formula | C22H24BrFN4O2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 443913-73-3 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | ZD6474 | Smiles | CN1CCC(CC1)COC2=C(C=C3C(=C2)N=CN=C3NC4=C(C=C(C=C4)Br)F)OC | ||
Mechanism of Action
| Targets/IC50/Ki |
VEGFR2
(Cell-free assay) 40 nM
VEGFR3
(Cell-free assay) 110 nM
EGFR
(Cell-free assay) 500 nM
|
|---|---|
| In vitro |
Vandetanib also inhibits VEGFR3 and EGFR with IC50 of 110 nM and 500 nM, respectively. This compound is not sensitive to PDGFRβ, Flt1, Tie-2 and FGFR1 with IC50 of 1.1-3.6 μM, while almost has no activity against MEK, CDK2, c-Kit, erbB2, FAK, PDK1, Akt and IGF-1R with IC50 above 10 μM. It inhibits VEGF-, EGF- and bFGF-stimulated HUVEC proliferation with IC50 of 60 nM, 170 nM and 800 nM, with no effect on basal endothelial cell growth. This chemical inhibits tumor cell growth with IC50 of 2.7 μM (A549) to 13.5 μM (Calu-6). It displays an inhibitory effect on the basal ABCG2-ATPase. Parental and ABCG2-expressing A431 cells showed similar sensitivities toward this compound. Exposure to EGFR inhibitors decreases pEGFR levels in A431 cells, with this compound displaying only a moderate effect. It displays a slight but measurable effect, whereas gefitinib, pelitinib and neratinib completely inhibit ABCG2-mediated efflux of mitoxantrone from A431/ABCG2 cells, similarly to the specific ABCG2 inhibitor Ko143. It inhibits both PC3wt and PC3R cell lines with similar IC50 of 13.3 μM and 11.5 μM, respectively. This chemical suppresses phosphorylation of VEGFR2 in HUVEC and EGFR in hepatoma cells and inhibits cell proliferation. It causes an accumulation of cells in the G0-G1 phases in GEO and OVCAR-3 cells and increases apoptosis in OVCAR-3, ZR-75-1, MCF-10A ras, and GEO cells. This compound causes a dose-dependent inhibition of EGFR phosphorylation in mouse NIH-EGFR fibroblasts and human MCF-10A ras breast cancer cells, two cell lines that overexpress the human EGFR. It treatment results in a dose-dependent inhibition of soft agar growth in seven human cell lines (breast, colon, gastric, and ovarian) with functional EGFR but lacking VEGFR2.
|
| Kinase Assay |
Kinase inhibition
|
|
Vandetanib is incubated with enzyme, 10 mM MnCl2, and 2 μM ATP in 96-well plates coated with a poly(Glu, Ala, Tyr) 6:3:1 random copolymer substrate. Phosphorylated tyrosine is then detected by sequential incubation with a mouse IgG anti-phosphotyrosine 4G10 antibody, a horseradish peroxidase-linked sheep antimouse immunoglobulin antibody, and 2,2′-azino-bis(3-ethylbenzthiazoline-6-sulfonic acid). This methodology is adapted to examine selectivity versus tyrosine kinases associated with EGFR, PDGFRβ, Tie-2, FGFR1, c-kit, erbB2, IGF-1R, and FAK. All enzyme assays (tyrosine or serine-threonine) used appropriate ATP concentrations at or just below the respective Km (0.2–14 μM). Selectivity versus serine-threonine kinases (CDK2, AKT, and PDK1) is examined using a relevant scintillation proximity-assay (SPA) in 96-well plates. CDK2 assays contained 10 mM MnCl2, 4.5 μM ATP, 0.15 μCi of [γ-33 P]ATP/reaction, 50 mM HEPES (pH 7.5), 1 mM DTT, 0.1 mM sodium orthovanadate, 0.1 mM sodium fluoride, 10 mM sodium glycerophosphate, 1 mg/mL BSA fraction V, and a retinoblastoma substrate (part of the retinoblastoma gene, 792–928, expressed in a glutathione S-transferase expression system; 0.22 μM final concentration). Reactions are allowed to proceed at room temperature for 60 minutes before quenching for 2 hours with 150 μL of a solution containing EDTA (62 mM final concentration), 3 μg of a rabbit immunoglobulin anti-glutathione S-transferase antibody and protein A SPA-polyvinyltoluene beads (0.8 mg/reaction). Plates are then sealed, centrifuged (1200× g for 5 minutes), and counted on a Microplate scintillation counter for 30 seconds.
|
|
| In vivo |
Vandetanib (2.5 mg/kg, i.v.), reverses a VEGF-induced hypotension by 63% but does not significantly affect a bFGF-induced hypotension. This compound (100 mg/kg) inhibits the tumor-induced blood vessel formation by 79%. It (12.5-100 mg/kg, orally) shows great tumor growth inhibition in human tumor xenografts including Calu-6, PC-3, MDA-MA-231, SKOV-3, SW620, A549, A431, B16-F10(AP3) and Lewis Lung, with little effects on body weight. In PC3wt xenografts, administration of this compound alone exerts paradoxical tumor growth stimulating effects. In PC3R xenografts, the low dose of this chemical (25 mg/kg) has no significant effect relative to control, whereas the high dose (50 mg/kg) significantly inhibits tumor growth compared with control. In contrast, the high-dose combination reveals a significant negative interaction between this compound 50 mg/kg and docetaxel 30 mg/kg in PC3R cells. In tumor-bearing mice, it suppresses phosphorylation of VEGFR2 and EGFR in tumor tissues, significantly decreases tumor vessel density, enhances tumor cell apoptosis, suppresses tumor growth, improves survival, reduces number of intrahepatic metastases, and up-regulates VEGF, TGF-alpha and EGF in tumor tissues. Treatment with this compound is not associated with serious adverse events, including ALT abnormality, bone marrow suppression or body weight loss. This chemical treatment of nude mice bearing palpable GEO colon cancer xenografts (which are sensitive to inhibition of EGFR signaling) induces dose-dependent tumor growth inhibition.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p-ERK / ERK / p-AKT / AKT p-EGFR / EGFR |
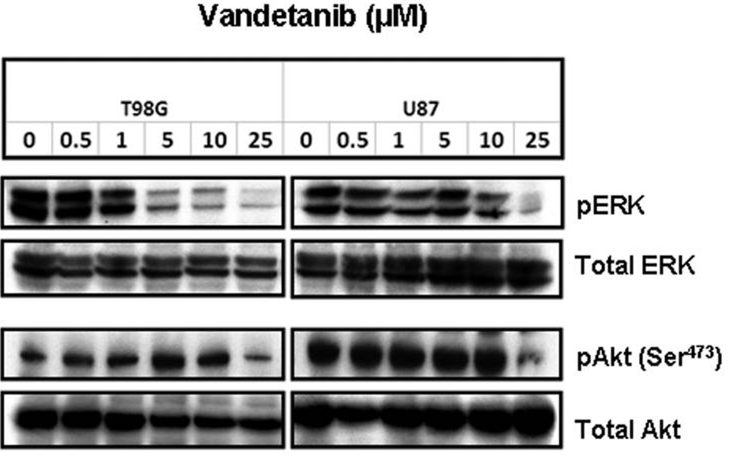
|
19622715 |
| Growth inhibition assay | Cell viability |
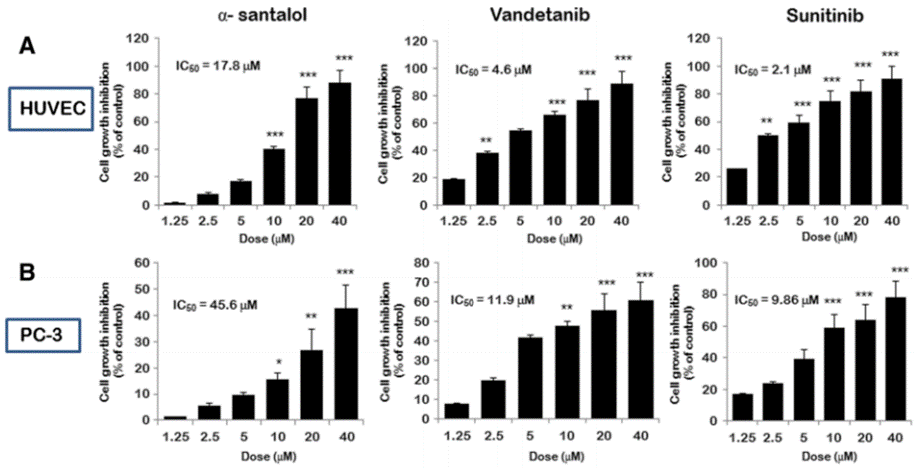
|
24261856 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT03291379 | Completed | Carcinoma Hepatocellular|Metastatic Colorectal Cancer |
Boston Scientific Corporation|Biocompatibles UK Ltd |
May 17 2017 | Early Phase 1 |
| NCT02495103 | Terminated | Renal Cell Carcinoma|Hereditary Leiomyomatosis|Renal Cell Cancer |
National Cancer Institute (NCI)|National Institutes of Health Clinical Center (CC) |
August 26 2015 | Phase 1|Phase 2 |
| NCT02530411 | Unknown status | Neoplasms |
Velindre NHS Trust|Cancer Research UK|AstraZeneca |
April 2015 | Phase 2 |
| NCT02268734 | Completed | Metastatic Sporadic Medullary Thyroid Cancer |
Fondazione IRCCS Istituto Nazionale dei Tumori Milano |
April 2014 | -- |
| NCT01876784 | Completed | Differentiated Thyroid Cancer |
Genzyme a Sanofi Company|Sanofi |
September 17 2013 | Phase 3 |
| NCT01661179 | Completed | Unresectable Locally Advanced or Metastatic Medullary Thyroid Carcinoma |
Genzyme a Sanofi Company|Sanofi |
November 2012 | Phase 1|Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































