research use only
Cytarabine DNA Synthesis Inhibitor
Cat.No.S1648
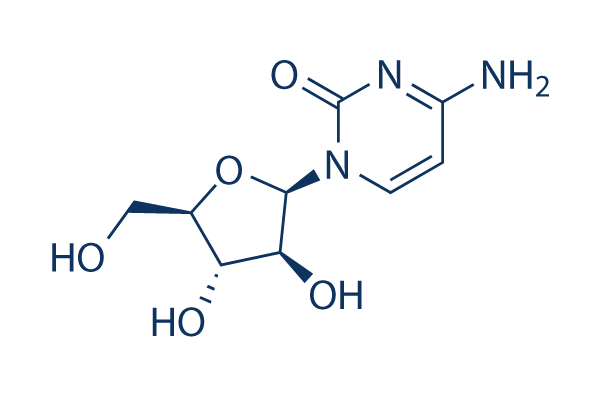
Chemical Structure
Molecular Weight: 243.22
Quality Control
Products Often Used Together with Cytarabine
| Related Targets | HDAC PARP ATM/ATR DNA-PK WRN Topoisomerase PPAR Sirtuin Casein Kinase eIF |
|---|---|
| Other DNA/RNA Synthesis Inhibitors | CX-5461 (Pidnarulex) SCR7 Favipiravir (T-705) EED226 RK-33 BMH-21 Triapine (3-AP) Carmofur Thymidine YK-4-279 |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| murine leukemia P388 | Function assay | Anticancer activity carried out in vitro against murine leukemia P388, IC50=0.14 μM | 11356110 | |||
| L1210 cell lines | Function assay | Concentration required to inhibit replication of L1210 cell lines by 50% after 72 hr incubation, ED50=0.05 μM. | 1652024 | |||
| CCRF-CEM cell lines | Function assay | 72 h | Concentration required to inhibit replication of CCRF-CEM cell lines by 50% after 72 hr incubation, ED50=6 nM. | 1652024 | ||
| CCRF-CEM cell line | Function assay | Inhibit the replication of CCRF-CEM cell line in vitro, ED50=5 nM. | 1995894 | |||
| HeLa human cell lines | Function assay | 144 h | The compound was tested in vitro for antitumor activity(50% inhibition of tumor cell growth) against HeLa human cell lines after 144 hr, ID50=0.1 μM | 3625714 | ||
| L1210 murine cell lines | Function assay | 72 h | The compound was tested in vitro for antitumor activity(50% inhibition of tumor cell growth) against the L1210 murine cell lines after 72 hours, ID50=20 nM. | 3625714 | ||
| P388 murine cell lines | Function assay | 72 h | The compound was tested in vitro for antitumor activity(50% inhibition of tumor cell growth) against the P388 murine cell lines after 72 hours, ID50=20 nM. | 3625714 | ||
| RPMI-6410 human leukemic lymphoblastoid cell lines | Cytotoxic assay | Cytotoxicity against RPMI-6410 human leukemic lymphoblastoid cell lines, IC50=0.06 μM | 6827546 | |||
| CCRF-CEM human leukemic lymphoblastoid cell lines | Cytotoxic assay | Cytotoxicity against CCRF-CEM human leukemic lymphoblastoid cell lines, IC50=20 nM. | 6827546 | |||
| MOLT-4 human leukemic lymphoblastoid cell lines | Cytotoxic assay | Cytotoxicity against MOLT-4 human leukemic lymphoblastoid cell lines, IC50=10 nM. | 6827546 | |||
| human leukemia cell line(CCRF-CEM) | Cytotoxic assay | In vitro cytotoxicity of compounds were determined on Inhibition of human leukemia cell line(CCRF-CEM), IC50=0.09 μM. | 8917645 | |||
| HL60 | Function assay | In vitro concentration required to induce apoptosis in HL60 cells, AC50 = 0.15 μM. | 12877593 | |||
| HL60R | Function assay | In vitro concentration required to induce apoptosis in HL60R cells, AC50 = 8 μM. | 12877593 | |||
| CCRF-CEM | Cytotoxicity assay | 48 hrs | Cytotoxicity against human CCRF-CEM cells after 48 hrs by cell titer-blue assay, EC50 = 0.005614 μM. | 19743858 | ||
| CCRF-CEM/C2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human CCRF-CEM/C2 cells after 48 hrs by cell titer-blue assay, EC50 = 0.0228 μM. | 19743858 | ||
| HL60/MX2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60/MX2 cells after 48 hrs by cell titer-blue assay, EC50 = 0.2612 μM. | 19743858 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells after 48 hrs by cell titer-blue assay, EC50 = 0.6572 μM. | 19743858 | ||
| L1210 | Cytotoxicity assay | 72 hrs | Cytotoxicity against mouse L1210 cells assessed as growth inhibition measured at 72 hrs by trypan blue exclusion assay, ED50 = 0.1 μM. | 118258 | ||
| BGM | Antiviral assay | 5 days | Antiviral activity against Vaccinia virus IHD infected in African green monkey BGM cells assessed as plaque reduction measured on day 5 by disk diffusion assay, ED50 = 0.07 μM. | 178873 | ||
| BGM | Antiviral assay | 5 days | Antiviral activity against Herpes Simplex virus HF infected in African green monkey BGM cells assessed as plaque reduction measured on day 5 by disk diffusion assay, ED50 = 0.16 μM. | 178873 | ||
| L1210 | Function assay | Inhibition of thymidylate synthase in L1210 cells, ED50 = 0.03 μM. | 3027329 | |||
| BALL-1 | Growth inhibition assay | 24 hrs | Growth inhibition of human BALL-1 cells after 24 hrs by WST-1 assay, GI50 = 0.08 μM. | 22264170 | ||
| HCT116 | Growth inhibition assay | 24 hrs | Growth inhibition of against human HCT116 cells after 24 hrs by WST-1 assay, GI50 = 0.12 μM. | 22264170 | ||
| HeLa | Growth inhibition assay | 24 hrs | Growth inhibition of human HeLa cells after 24 hrs by WST-1 assay, GI50 = 0.18 μM. | 22264170 | ||
| A549 | Growth inhibition assay | 24 hrs | Growth inhibition of human A549 cells after 24 hrs by WST-1 assay, GI50 = 0.2 μM. | 22264170 | ||
| HUVEC | Growth inhibition assay | 72 hrs | Growth inhibition of HUVEC cells after 72 hrs by WST-1 assay, GI50 = 0.23 μM. | 22264170 | ||
| NUGC3 | Growth inhibition assay | 24 hrs | Growth inhibition of human NUGC3 cells after 24 hrs by WST-1 assay, GI50 = 0.25 μM. | 22264170 | ||
| HDF | Growth inhibition assay | 72 hrs | Growth inhibition of HDF cells after 72 hrs by WST-1 assay, GI50 = 0.25 μM. | 22264170 | ||
| leukemia L1210 | Function assay | Inhibition of L1210 lymphoid leukemia cells 48 hr after administration (length of preincubation=0 hr), IC50 = 0.01 μM. | 1527783 | |||
| leukemia L1210 | Function assay | Inhibition of L1210 lymphoid leukemia cells 72 hr after administration (length of preincubation=0 hr), IC50 = 0.01 μM. | 1527783 | |||
| leukemia L1210 | Function assay | Inhibition of L1210 lymphoid leukemia cells 24 hr after administration (length of preincubation=0 hr), IC50 = 0.1 μM. | 1527783 | |||
| leukemia L1210 | Function assay | Inhibitory concentration on multidrug-resistant L1210 leukemia cells., IC50 = 0.036 μM. | 1906107 | |||
| leukemia L1210 | Function assay | Inhibitory concentration on parentral (sensitive) L1210 leukemia cells., IC50 = 0.036 μM. | 1906107 | |||
| leukemia L1210 | Function assay | Inhibitory activity against murine leukemia L1210 cells, IC50 = 0.08 μM. | 1992143 | |||
| HT-29 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human HT-29 cells after 96 hrs by MTT assay, IC50 = 0.066 μM. | 2778449 | ||
| P388 | Antiproliferative assay | 48 hrs | Antiproliferative activity against mouse P388 cells after 48 hrs by MTT assay, IC50 = 1.233 μM. | 2778449 | ||
| KB | Function assay | Tested in vitro inhibitory activity against human oral epidermoid carcinoma KB cells, IC50 = 2.3 μM. | 8277499 | |||
| K562(S) | Growth inhibition assay | Inhibition of cell growth against human myeloid leukemia K562(S) cells, IC50 = 0.5 μM. | 10560743 | |||
| NCI-H23 | Cytotoxicity assay | In vitro cytotoxicity was evaluated against lung NCI-H23 cells; 1 x 10E1, IC50 = 1 μM. | 10780904 | |||
| SNB-7 | Cytotoxicity assay | In vitro cytotoxicity was evaluated againstCNS SNB-7 cells; 1 x 10E1, IC50 = 1 μM. | 10780904 | |||
| SK-MEL-28 | Cytotoxicity assay | In vitro cytotoxicity was evaluated against melanoma SK-MEL-28 cells, IC50 = 1 μM. | 10780904 | |||
| HL60 | Function assay | In vitro inhibitory concentration against proliferation of HL60 cells, IC50 = 0.08 μM. | 12877593 | |||
| HL60R | Function assay | In vitro concentration required to induce apoptosis in HL60R cells, IC50 = 0.35 μM. | 12877593 | |||
| CCRF-HSB-2 | Growth inhibition assay | Growth inhibition of human CCRF-HSB-2 cells by MTT assay, IC50 = 0.15 μM. | 17049252 | |||
| KB | Growth inhibition assay | Growth inhibition of human KB cells by MTT assay, IC50 = 0.7 μM. | 17049252 | |||
| L1210/0 | Cytotoxicity assay | 48 hrs | Cytotoxicity against mouse L1210/0 cells after 48 hrs, IC50 = 0.7 μM. | 17341060 | ||
| 2209-23 | Antiviral assay | Antiviral activity against Hepatitis C virus infected human 2209-23 cells by genotype 1b replicon assay, IC50 = 0.02 μM. | 19055482 | |||
| 2209-23 | Antiproliferative assay | Antiproliferative activity against human 2209-23 cells by [3H]thymidine incorporation scintillation proximity assay, IC50 = 0.029 μM. | 19055482 | |||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells after 48 hrs by MTT assay, IC50 = 0.118 μM. | 19321234 | ||
| SF268 | Cytotoxicity assay | Cytotoxicity against human SF268 cells by MTT assay, IC50 = 0.00768 μM. | 19345581 | |||
| L1210 | Cytotoxicity assay | 3 days | Cytotoxicity against deoxynucleoside analogue-sensitive mouse L1210 cells after 3 days by MTT assay, IC50 = 0.0004 μM. | 19674903 | ||
| CCRF-CEM | Cytotoxicity assay | 3 days | Cytotoxicity against deoxynucleoside analogue-sensitive human CCRF-CEM cells after 3 days by MTT assay, IC50 = 0.001 μM. | 19674903 | ||
| MCF7 | Cytotoxicity assay | 3 days | Cytotoxicity against deoxynucleoside analogue-sensitive human MCF7 cells after 3 days by MTT assay, IC50 = 0.004 μM. | 19674903 | ||
| MCF7 1K | Cytotoxicity assay | 3 days | Cytotoxicity against deoxynucleoside analogue-resistant human MCF7 1K cells overexpressing ribonucleotide reductase after 3 days by MTT assay, IC50 = 0.04 μM. | 19674903 | ||
| MESSA | Cytotoxicity assay | 3 days | Cytotoxicity against deoxynucleoside analogue-sensitive human MESSA cells after 3 days by MTT assay, IC50 = 1.7 μM. | 19674903 | ||
| K562 | Cytotoxicity assay | Cytotoxicity against human K562 cells by sulforhodamine B method, IC50 = 0.05 μM. | 19691349 | |||
| SNU638 | Cytotoxicity assay | Cytotoxicity against human SNU638 cells by sulforhodamine B method, IC50 = 0.15 μM. | 19691349 | |||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells by sulforhodamine B method, IC50 = 1.9 μM. | 19691349 | |||
| T47D | Cytotoxicity assay | Cytotoxicity against human T47D cells by sulforhodamine B method, IC50 = 2.7 μM. | 19691349 | |||
| HCT116 | Cytotoxicity assay | Cytotoxicity against human HCT116 cells by sulforhodamine B method, IC50 = 5.3 μM. | 19691349 | |||
| L1210 | Cytotoxicity assay | 2 days | Cytotoxicity against mouse L1210 cells after 2 days, IC50 = 0.024 μM. | 19910080 | ||
| CEM | Cytotoxicity assay | 2 days | Cytotoxicity against human CEM cells after 2 days, IC50 = 0.024 μM. | 19910080 | ||
| CEM | Cytotoxicity assay | 48 hrs | Cytotoxicity against human CEM cells assessed as inhibition of cell proliferation after 48 hrs, IC50 = 0.024 μM. | 20000418 | ||
| L1210 | Cytotoxicity assay | 48 hrs | Cytotoxicity against mouse L1210 cells assessed as inhibition of cell proliferation after 48 hrs, IC50 = 0.024 μM. | 20000418 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells assessed as inhibition of cell proliferation after 48 hrs, IC50 = 0.26 μM. | 20000418 | ||
| RS4:11 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human RS4:11 cells having FLT3-ITD mutation after 72 hrs by WST8 assay, IC50 = 0.26 μM. | 20153646 | ||
| MV411 | Antiproliferative assay | 72 hrs | Antiproliferative activity against wild type FLT3 expressing human MV411 cells after 72 hrs by WST8 assay, IC50 = 3.36 μM. | 20153646 | ||
| HCT116 | Antitumor assay | 24 hrs | Antitumor activity against human HCT116 cells after 24 hrs by MTT assay, IC50 = 0.1 μM. | 20211564 | ||
| H460 | Antitumor assay | 24 hrs | Antitumor activity against human H460 cells after 24 hrs by MTT assay, IC50 = 0.1 μM. | 20211564 | ||
| MOLT4 | Antitumor assay | 24 hrs | Antitumor activity against human MOLT4 cells after 24 hrs by MTT assay, IC50 = 1 μM. | 20211564 | ||
| SW620 | Antitumor assay | 24 hrs | Antitumor activity against human SW620 cells after 24 hrs by MTT assay, IC50 = 30 μM. | 20211564 | ||
| SF268 | Cytotoxicity assay | 3 days | Cytotoxicity against human SF268 cells after 3 days by MTT assay, IC50 = 7.68 μM. | 20356655 | ||
| SFME | Antiproliferative assay | 24 hrs | Antiproliferative activity against mouse SFME cells after 24 hrs by MTT assay, IC50 = 0.8 μM. | 20398972 | ||
| HM-SFME-1 | Antiproliferative assay | 24 hrs | Antiproliferative activity against mouse r/m HM-SFME-1 cells after 24 hrs by MTT assay, IC50 = 33.3 μM. | 20398972 | ||
| HepG2 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HepG2 cells after 24 hrs by MTT assay, IC50 = 0.2 μM. | 20692841 | ||
| K562 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human K562 cells after 24 hrs by MTT assay, IC50 = 33.5 μM. | 20692841 | ||
| Bel7402 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human Bel7402 cells after 24 hrs by MTT assay, IC50 = 34.2 μM. | 20692841 | ||
| K562 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human K562 cells after 48 hrs by MTT assay, IC50 = 24.29 μM. | 21620529 | ||
| S180 | Antiproliferative assay | 48 hrs | Antiproliferative activity against mouse S180 cells after 48 hrs by MTT assay, IC50 = 25.43 μM. | 21620529 | ||
| H22 | Antiproliferative assay | 48 hrs | Antiproliferative activity against mouse H22 cells after 48 hrs by MTT assay, IC50 = 25.98 μM. | 21620529 | ||
| HepG2 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 26.23 μM. | 21620529 | ||
| B16 | Antiproliferative assay | 48 hrs | Antiproliferative activity against mouse B16 cells after 48 hrs by MTT assay, IC50 = 30.62 μM. | 21620529 | ||
| CCRF-CEM | Cytotoxicity assay | 48 hrs | Cytotoxicity against human CCRF-CEM cells assessed as cell viability after 48 hrs by celltiter-blue assay, IC50 = 0.006 μM. | 22582991 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells assessed as cell viability after 48 hrs by celltiter-blue assay, IC50 = 0.049 μM. | 22582991 | ||
| K562 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human K562 cells assessed as cell viability after 48 hrs by celltiter-blue assay, IC50 = 14 μM. | 22582991 | ||
| blood stem/progenitor cells | Cytotoxicity assay | 48 hrs | Cytotoxicity against human CD34-positive blood stem/progenitor cells after 48 hrs by MTT assay, IC50 = 0.59 μM. | 23578690 | ||
| MA9.3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MA9.3 cells after 48 hrs by MTT assay, IC50 = 0.92 μM. | 23578690 | ||
| HeLa | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HeLa cells after 72 hrs by SRB assay, IC50 = 3.54 μM. | 23867603 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells after 72 hrs by SRB assay, IC50 = 3.82 μM. | 23867603 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by SRB assay, IC50 = 4.07 μM. | 23867603 | ||
| K562 | Cytotoxicity assay | Cytotoxicity against human K562 cells by SRB assay, IC50 = 0.05 μM. | 24956556 | |||
| SNU638 | Cytotoxicity assay | Cytotoxicity against human SNU638 cells by SRB assay, IC50 = 0.15 μM. | 24956556 | |||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells by SRB assay, IC50 = 1.9 μM. | 24956556 | |||
| T47D | Cytotoxicity assay | Cytotoxicity against human T47D cells by SRB assay, IC50 = 2.72 μM. | 24956556 | |||
| HCT116 | Cytotoxicity assay | Cytotoxicity against human HCT116 cells by SRB assay, IC50 = 5.3 μM. | 24956556 | |||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells assessed as inhibition of cell proliferation after 72 hrs by sulforhodamine B assay, IC50 = 1.2 μM. | 26010585 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-231 cells assessed as inhibition of cell proliferation after 72 hrs by sulforhodamine B assay, IC50 = 1.37 μM. | 26010585 | ||
| SKHEP1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SKHEP1 cells assessed as inhibition of cell proliferation after 72 hrs by sulforhodamine B assay, IC50 = 2.12 μM. | 26010585 | ||
| HCT116 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT116 cells assessed as inhibition of cell proliferation after 72 hrs by sulforhodamine B assay, IC50 = 2.42 μM. | 26010585 | ||
| SNU638 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SNU638 cells assessed as inhibition of cell proliferation after 72 hrs by sulforhodamine B assay, IC50 = 3.98 μM. | 26010585 | ||
| 143B | Cytotoxicity assay | 72 hrs | Cytotoxicity against human 143B cells after 72 hrs by SRB assay, IC50 = 2.06 μM. | 27073055 | ||
| HepG2 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HepG2 cells after 72 hrs by SRB assay, IC50 = 2.86 μM. | 27073055 | ||
| HeLa | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HeLa cells after 72 hrs by SRB assay, IC50 = 3.54 μM. | 27073055 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7cells after 72 hrs by SRB assay, IC50 = 3.82 μM. | 27073055 | ||
| KB | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KB cells after 72 hrs by SRB assay, IC50 = 4.07 μM. | 27073055 | ||
| HDF | Cytotoxicity assay | 72 hrs | Cytotoxicity against HDF cells after 72 hrs by SRB assay, IC50 = 4.99 μM. | 27073055 | ||
| thymidine kinase deficient 143B | Cytotoxicity assay | 72 hrs | Cytotoxicity against human thymidine kinase deficient 143B cells after 72 hrs by SRB assay, IC50 = 16.88 μM. | 27073055 | ||
| SMMC7721 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SMMC7721 cells assessed as inhibition of cell proliferation after 72 hrs by MTT assay, IC50 = 0.013 μM. | 29886320 | ||
| LO2 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human LO2 cells assessed as inhibition of cell proliferation after 72 hrs by MTT assay, IC50 = 0.17 μM. | 29886320 | ||
| HeLa | Cytotoxicity assay | Cytotoxicity against human HeLa cells, IC50 = 0.95 μM. | 29886320 | |||
| SNU638 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SNU638 cells assessed as decrease in cell viability after 72 hrs by SRB assay, IC50 = 1.08 μM. | 29906687 | ||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells assessed as decrease in cell viability after 72 hrs by SRB assay, IC50 = 1.75 μM. | 29906687 | ||
| SKHEP1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SKHEP1 cells assessed as decrease in cell viability after 72 hrs by SRB assay, IC50 = 1.8 μM. | 29906687 | ||
| HCT116 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT116 cells assessed as decrease in cell viability after 72 hrs by SRB assay, IC50 = 4.4 μM. | 29906687 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-231 cells assessed as decrease in cell viability after 72 hrs by SRB assay, IC50 = 10.49 μM. | 29906687 | ||
| PC3 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human PC3 cells assessed as decrease in cell viability after 72 hrs by SRB assay, IC50 = 48.4 μM. | 29906687 | ||
| Jurkat E6-1 | Antiproliferative assay | Antiproliferative activity against human Jurkat E6-1 cells, IC50 = 0.018 μM. | 30114661 | |||
| HL60 | Antiproliferative assay | Antiproliferative activity against human HL60 cells, IC50 = 0.185 μM. | 30114661 | |||
| BL21(DE3) | Function assay | 3 mins | Activity of human recombinant N-terminal His-tagged DCK Pro122Ser mutant expressed in Escherichia coli BL21(DE3) cells assessed as cytosine-beta-D-arabinofuranoside-5'-MP formation incubated for 3 mins by liquid chromatography-tandem mass spectrometry, Km = 1.56 μM. | 23230131 | ||
| BL21(DE3) | Function assay | 3 mins | Activity of human recombinant N-terminal His-tagged DCK Ile24Val mutant expressed in Escherichia coli BL21(DE3) cells assessed as cytosine-beta-D-arabinofuranoside-5'-MP formation incubated for 3 mins by liquid chromatography-tandem mass spectrometry, Km = 2.45 μM. | 23230131 | ||
| BL21(DE3) | Function assay | 3 mins | Activity of human recombinant N-terminal His-tagged DCK Ala119Gly mutant expressed in Escherichia coli BL21(DE3) cells assessed as cytosine-beta-D-arabinofuranoside-5'-MP formation incubated for 3 mins by liquid chromatography-tandem mass spectrometry, Km = 2.54 μM. | 23230131 | ||
| BL21(DE3) | Function assay | 3 mins | Activity of human recombinant N-terminal His-tagged wild type DCK expressed in Escherichia coli BL21(DE3) cells assessed as cytosine-beta-D-arabinofuranoside-5'-MP formation incubated for 3 mins by liquid chromatography-tandem mass spectrometry, Km = 3.04 μM. | 23230131 | ||
| KB | Antiviral assay | 72 hrs | Antiviral activity against Pseudorabies virus infected in human KB cells assessed as inhibition of virus-induced cytopathic effect after 72 hrs by microscopically | 458802 | ||
| KB | Antiviral assay | 72 hrs | Antiviral activity against Herpes simplex virus type 1 infected in human KB cells assessed as inhibition of virus-induced cytopathic effect after 72 hrs by microscopically | 458802 | ||
| KB | Antiviral assay | 72 hrs | Antiviral activity against Vaccinia virus infected in human KB cells assessed as inhibition of virus-induced cytopathic effect after 72 hrs by microscopically | 458802 | ||
| KB | Antiviral assay | 72 hrs | Antiviral activity against Myxoma virus infected in human KB cells assessed as inhibition of virus-induced cytopathic effect after 72 hrs by microscopically | 458802 | ||
| H441 | Cytotoxicity assay | Cytotoxicity against human H441 cells by serum lactate dehydrogenase assay | 19574048 | |||
| H441 | Cytotoxicity assay | Cytotoxicity against human H441 cells by glucosaminidase assay | 19574048 | |||
| KG1 | Antitumor assay | 100 mg/kg | 11 days | Antitumor activity against human KG1 cells xenografted in mouse assessed as tumor regression at 100 mg/kg, po qd measured after 11 days post tumor implantation | 26505898 | |
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for A673 cells) | 29435139 | |||
| P12-ICHIKAWA | Growth Inhibition Assay | Inhibition of human P12-ICHIKAWA cell growth in a cell viability assay, IC50 = 0.005537 μM. | SANGER | |||
| 697 | Growth Inhibition Assay | Inhibition of human 697 cell growth in a cell viability assay, IC50 = 0.0106 μM. | SANGER | |||
| ES1 | Growth Inhibition Assay | Inhibition of human ES1 cell growth in a cell viability assay, IC50 = 0.01297 μM. | SANGER | |||
| HEL | Growth Inhibition Assay | Inhibition of human HEL cell growth in a cell viability assay, IC50 = 0.02052 μM. | SANGER | |||
| CHP-212 | Growth Inhibition Assay | Inhibition of human CHP-212 cell growth in a cell viability assay, IC50 = 0.03347 μM. | SANGER | |||
| CTB-1 | Growth Inhibition Assay | Inhibition of human CTB-1 cell growth in a cell viability assay, IC50 = 0.0344 μM. | SANGER | |||
| A427 | Growth Inhibition Assay | Inhibition of human A427 cell growth in a cell viability assay, IC50 = 0.03449 μM. | SANGER | |||
| MOLT-16 | Growth Inhibition Assay | Inhibition of human MOLT-16 cell growth in a cell viability assay, IC50 = 0.03607 μM. | SANGER | |||
| MC-IXC | Growth Inhibition Assay | Inhibition of human MC-IXC cell growth in a cell viability assay, IC50 = 0.04141 μM. | SANGER | |||
| BHT-101 | Growth Inhibition Assay | Inhibition of human BHT-101 cell growth in a cell viability assay, IC50 = 0.04176 μM. | SANGER | |||
| K5 | Growth Inhibition Assay | Inhibition of human K5 cell growth in a cell viability assay, IC50 = 0.0432 μM. | SANGER | |||
| A549 | Growth Inhibition Assay | Inhibition of human A549 cell growth in a cell viability assay, IC50 = 0.04966 μM. | SANGER | |||
| NCI-H1703 | Growth Inhibition Assay | Inhibition of human NCI-H1703 cell growth in a cell viability assay, IC50 = 0.04981 μM. | SANGER | |||
| LB2241-RCC | Growth Inhibition Assay | Inhibition of human LB2241-RCC cell growth in a cell viability assay, IC50 = 0.05534 μM. | SANGER | |||
| MLMA | Growth Inhibition Assay | Inhibition of human MLMA cell growth in a cell viability assay, IC50 = 0.05551 μM. | SANGER | |||
| P30-OHK | Growth Inhibition Assay | Inhibition of human P30-OHK cell growth in a cell viability assay, IC50 = 0.0565 μM. | SANGER | |||
| NCI-H510A | Growth Inhibition Assay | Inhibition of human NCI-H510A cell growth in a cell viability assay, IC50 = 0.06826 μM. | SANGER | |||
| J-RT3-T3-5 | Growth Inhibition Assay | Inhibition of human J-RT3-T3-5 cell growth in a cell viability assay, IC50 = 0.06842 μM. | SANGER | |||
| VA-ES-BJ | Growth Inhibition Assay | Inhibition of human VA-ES-BJ cell growth in a cell viability assay, IC50 = 0.07165 μM. | SANGER | |||
| T-24 | Growth Inhibition Assay | Inhibition of human T-24 cell growth in a cell viability assay, IC50 = 0.07856 μM. | SANGER | |||
| NB69 | Growth Inhibition Assay | Inhibition of human NB69 cell growth in a cell viability assay, IC50 = 0.07948 μM. | SANGER | |||
| CTV-1 | Growth Inhibition Assay | Inhibition of human CTV-1 cell growth in a cell viability assay, IC50 = 0.08474 μM. | SANGER | |||
| MEL-JUSO | Growth Inhibition Assay | Inhibition of human MEL-JUSO cell growth in a cell viability assay, IC50 = 0.08576 μM. | SANGER | |||
| MOLT-4 | Growth Inhibition Assay | Inhibition of human MOLT-4 cell growth in a cell viability assay, IC50 = 0.08967 μM. | SANGER | |||
| SW982 | Growth Inhibition Assay | Inhibition of human SW982 cell growth in a cell viability assay, IC50 = 0.09123 μM. | SANGER | |||
| NCI-SNU-1 | Growth Inhibition Assay | Inhibition of human NCI-SNU-1 cell growth in a cell viability assay, IC50 = 0.09677 μM. | SANGER | |||
| GP5d | Growth Inhibition Assay | Inhibition of human GP5d cell growth in a cell viability assay, IC50 = 0.10205 μM. | SANGER | |||
| SAS | Growth Inhibition Assay | Inhibition of human SAS cell growth in a cell viability assay, IC50 = 0.10757 μM. | SANGER | |||
| BFTC-905 | Growth Inhibition Assay | Inhibition of human BFTC-905 cell growth in a cell viability assay, IC50 = 0.10831 μM. | SANGER | |||
| LB1047-RCC | Growth Inhibition Assay | Inhibition of human LB1047-RCC cell growth in a cell viability assay, IC50 = 0.10842 μM. | SANGER | |||
| BB65-RCC | Growth Inhibition Assay | Inhibition of human BB65-RCC cell growth in a cell viability assay, IC50 = 0.11019 μM. | SANGER | |||
| KE-37 | Growth Inhibition Assay | Inhibition of human KE-37 cell growth in a cell viability assay, IC50 = 0.11085 μM. | SANGER | |||
| NCI-H292 | Growth Inhibition Assay | Inhibition of human NCI-H292 cell growth in a cell viability assay, IC50 = 0.1142 μM. | SANGER | |||
| HT-1080 | Growth Inhibition Assay | Inhibition of human HT-1080 cell growth in a cell viability assay, IC50 = 0.11447 μM. | SANGER | |||
| CAKI-1 | Growth Inhibition Assay | Inhibition of human CAKI-1 cell growth in a cell viability assay, IC50 = 0.12001 μM. | SANGER | |||
| HMV-II | Growth Inhibition Assay | Inhibition of human HMV-II cell growth in a cell viability assay, IC50 = 0.12363 μM. | SANGER | |||
| ES8 | Growth Inhibition Assay | Inhibition of human ES8 cell growth in a cell viability assay, IC50 = 0.12393 μM. | SANGER | |||
| NEC8 | Growth Inhibition Assay | Inhibition of human NEC8 cell growth in a cell viability assay, IC50 = 0.13421 μM. | SANGER | |||
| EW-1 | Growth Inhibition Assay | Inhibition of human EW-1 cell growth in a cell viability assay, IC50 = 0.13862 μM. | SANGER | |||
| H-EMC-SS | Growth Inhibition Assay | Inhibition of human H-EMC-SS cell growth in a cell viability assay, IC50 = 0.14528 μM. | SANGER | |||
| NTERA-S-cl-D1 | Growth Inhibition Assay | Inhibition of human NTERA-S-cl-D1 cell growth in a cell viability assay, IC50 = 0.1591 μM. | SANGER | |||
| COR-L105 | Growth Inhibition Assay | Inhibition of human COR-L105 cell growth in a cell viability assay, IC50 = 0.16039 μM. | SANGER | |||
| A2780 | Growth Inhibition Assay | Inhibition of human A2780 cell growth in a cell viability assay, IC50 = 0.16238 μM. | SANGER | |||
| C2BBe1 | Growth Inhibition Assay | Inhibition of human C2BBe1 cell growth in a cell viability assay, IC50 = 0.16596 μM. | SANGER | |||
| SK-UT-1 | Growth Inhibition Assay | Inhibition of human SK-UT-1 cell growth in a cell viability assay, IC50 = 0.18142 μM. | SANGER | |||
| CGTH-W-1 | Growth Inhibition Assay | Inhibition of human CGTH-W-1 cell growth in a cell viability assay, IC50 = 0.1818 μM. | SANGER | |||
| PA-1 | Growth Inhibition Assay | Inhibition of human PA-1 cell growth in a cell viability assay, IC50 = 0.18665 μM. | SANGER | |||
| NB10 | Growth Inhibition Assay | Inhibition of human NB10 cell growth in a cell viability assay, IC50 = 0.18814 μM. | SANGER | |||
| HCC1806 | Growth Inhibition Assay | Inhibition of human HCC1806 cell growth in a cell viability assay, IC50 = 0.19102 μM. | SANGER | |||
| PSN1 | Growth Inhibition Assay | Inhibition of human PSN1 cell growth in a cell viability assay, IC50 = 0.19281 μM. | SANGER | |||
| ES3 | Growth Inhibition Assay | Inhibition of human ES3 cell growth in a cell viability assay, IC50 = 0.1987 μM. | SANGER | |||
| OVCAR-8 | Growth Inhibition Assay | Inhibition of human OVCAR-8 cell growth in a cell viability assay, IC50 = 0.2108 μM. | SANGER | |||
| EM-2 | Growth Inhibition Assay | Inhibition of human EM-2 cell growth in a cell viability assay, IC50 = 0.21139 μM. | SANGER | |||
| ES4 | Growth Inhibition Assay | Inhibition of human ES4 cell growth in a cell viability assay, IC50 = 0.21401 μM. | SANGER | |||
| TE-8 | Growth Inhibition Assay | Inhibition of human TE-8 cell growth in a cell viability assay, IC50 = 0.2191 μM. | SANGER | |||
| OVCAR-5 | Growth Inhibition Assay | Inhibition of human OVCAR-5 cell growth in a cell viability assay, IC50 = 0.21963 μM. | SANGER | |||
| HN | Growth Inhibition Assay | Inhibition of human HN cell growth in a cell viability assay, IC50 = 0.2207 μM. | SANGER | |||
| A673 | Growth Inhibition Assay | Inhibition of human A673 cell growth in a cell viability assay, IC50 = 0.22382 μM. | SANGER | |||
| 769-P | Growth Inhibition Assay | Inhibition of human 769-P cell growth in a cell viability assay, IC50 = 0.22786 μM. | SANGER | |||
| LC-2-ad | Growth Inhibition Assay | Inhibition of human LC-2-ad cell growth in a cell viability assay, IC50 = 0.22961 μM. | SANGER | |||
| SJSA-1 | Growth Inhibition Assay | Inhibition of human SJSA-1 cell growth in a cell viability assay, IC50 = 0.23516 μM. | SANGER | |||
| HuP-T4 | Growth Inhibition Assay | Inhibition of human HuP-T4 cell growth in a cell viability assay, IC50 = 0.23681 μM. | SANGER | |||
| ES5 | Growth Inhibition Assay | Inhibition of human ES5 cell growth in a cell viability assay, IC50 = 0.2429 μM. | SANGER | |||
| HCT-116 | Growth Inhibition Assay | Inhibition of human HCT-116 cell growth in a cell viability assay, IC50 = 0.24477 μM. | SANGER | |||
| DB | Growth Inhibition Assay | Inhibition of human DB cell growth in a cell viability assay, IC50 = 0.24595 μM. | SANGER | |||
| LXF-289 | Growth Inhibition Assay | Inhibition of human LXF-289 cell growth in a cell viability assay, IC50 = 0.25141 μM. | SANGER | |||
| ES7 | Growth Inhibition Assay | Inhibition of human ES7 cell growth in a cell viability assay, IC50 = 0.25716 μM. | SANGER | |||
| EW-16 | Growth Inhibition Assay | Inhibition of human EW-16 cell growth in a cell viability assay, IC50 = 0.26588 μM. | SANGER | |||
| LU-99A | Growth Inhibition Assay | Inhibition of human LU-99A cell growth in a cell viability assay, IC50 = 0.26643 μM. | SANGER | |||
| D-566MG | Growth Inhibition Assay | Inhibition of human D-566MG cell growth in a cell viability assay, IC50 = 0.26904 μM. | SANGER | |||
| ML-2 | Growth Inhibition Assay | Inhibition of human ML-2 cell growth in a cell viability assay, IC50 = 0.26965 μM. | SANGER | |||
| H4 | Growth Inhibition Assay | Inhibition of human H4 cell growth in a cell viability assay, IC50 = 0.27445 μM. | SANGER | |||
| HCE-4 | Growth Inhibition Assay | Inhibition of human HCE-4 cell growth in a cell viability assay, IC50 = 0.27713 μM. | SANGER | |||
| ALL-PO | Growth Inhibition Assay | Inhibition of human ALL-PO cell growth in a cell viability assay, IC50 = 0.28709 μM. | SANGER | |||
| SNU-387 | Growth Inhibition Assay | Inhibition of human SNU-387 cell growth in a cell viability assay, IC50 = 0.28877 μM. | SANGER | |||
| LOXIMVI | Growth Inhibition Assay | Inhibition of human LOXIMVI cell growth in a cell viability assay, IC50 = 0.29031 μM. | SANGER | |||
| YKG-1 | Growth Inhibition Assay | Inhibition of human YKG-1 cell growth in a cell viability assay, IC50 = 0.29831 μM. | SANGER | |||
| D-423MG | Growth Inhibition Assay | Inhibition of human D-423MG cell growth in a cell viability assay, IC50 = 0.29851 μM. | SANGER | |||
| KYSE-510 | Growth Inhibition Assay | Inhibition of human KYSE-510 cell growth in a cell viability assay, IC50 = 0.30633 μM. | SANGER | |||
| NB14 | Growth Inhibition Assay | Inhibition of human NB14 cell growth in a cell viability assay, IC50 = 0.30956 μM. | SANGER | |||
| A204 | Growth Inhibition Assay | Inhibition of human A204 cell growth in a cell viability assay, IC50 = 0.31068 μM. | SANGER | |||
| 23132-87 | Growth Inhibition Assay | Inhibition of human 23132-87 cell growth in a cell viability assay, IC50 = 0.31872 μM. | SANGER | |||
| Ca9-22 | Growth Inhibition Assay | Inhibition of human Ca9-22 cell growth in a cell viability assay, IC50 = 0.32077 μM. | SANGER | |||
| MHH-ES-1 | Growth Inhibition Assay | Inhibition of human MHH-ES-1 cell growth in a cell viability assay, IC50 = 0.32338 μM. | SANGER | |||
| MSTO-211H | Growth Inhibition Assay | Inhibition of human MSTO-211H cell growth in a cell viability assay, IC50 = 0.32388 μM. | SANGER | |||
| YH-13 | Growth Inhibition Assay | Inhibition of human YH-13 cell growth in a cell viability assay, IC50 = 0.32755 μM. | SANGER | |||
| HAL-01 | Growth Inhibition Assay | Inhibition of human HAL-01 cell growth in a cell viability assay, IC50 = 0.32786 μM. | SANGER | |||
| RH-1 | Growth Inhibition Assay | Inhibition of human RH-1 cell growth in a cell viability assay, IC50 = 0.32816 μM. | SANGER | |||
| LAMA-84 | Growth Inhibition Assay | Inhibition of human LAMA-84 cell growth in a cell viability assay, IC50 = 0.33454 μM. | SANGER | |||
| NBsusSR | Growth Inhibition Assay | Inhibition of human NBsusSR cell growth in a cell viability assay, IC50 = 0.3354 μM. | SANGER | |||
| JAR | Growth Inhibition Assay | Inhibition of human JAR cell growth in a cell viability assay, IC50 = 0.33788 μM. | SANGER | |||
| GI-1 | Growth Inhibition Assay | Inhibition of human GI-1 cell growth in a cell viability assay, IC50 = 0.33883 μM. | SANGER | |||
| RPMI-7951 | Growth Inhibition Assay | Inhibition of human RPMI-7951 cell growth in a cell viability assay, IC50 = 0.33952 μM. | SANGER | |||
| 8-MG-BA | Growth Inhibition Assay | Inhibition of human 8-MG-BA cell growth in a cell viability assay, IC50 = 0.3407 μM. | SANGER | |||
| GT3TKB | Growth Inhibition Assay | Inhibition of human GT3TKB cell growth in a cell viability assay, IC50 = 0.34071 μM. | SANGER | |||
| UM-UC-3 | Growth Inhibition Assay | Inhibition of human UM-UC-3 cell growth in a cell viability assay, IC50 = 0.34227 μM. | SANGER | |||
| U-2-OS | Growth Inhibition Assay | Inhibition of human U-2-OS cell growth in a cell viability assay, IC50 = 0.34387 μM. | SANGER | |||
| SW620 | Growth Inhibition Assay | Inhibition of human SW620 cell growth in a cell viability assay, IC50 = 0.3492 μM. | SANGER | |||
| HOP-92 | Growth Inhibition Assay | Inhibition of human HOP-92 cell growth in a cell viability assay, IC50 = 0.3544 μM. | SANGER | |||
| BFTC-909 | Growth Inhibition Assay | Inhibition of human BFTC-909 cell growth in a cell viability assay, IC50 = 0.35736 μM. | SANGER | |||
| D-283MED | Growth Inhibition Assay | Inhibition of human D-283MED cell growth in a cell viability assay, IC50 = 0.36027 μM. | SANGER | |||
| SW48 | Growth Inhibition Assay | Inhibition of human SW48 cell growth in a cell viability assay, IC50 = 0.36566 μM. | SANGER | |||
| ATN-1 | Growth Inhibition Assay | Inhibition of human ATN-1 cell growth in a cell viability assay, IC50 = 0.36571 μM. | SANGER | |||
| ACHN | Growth Inhibition Assay | Inhibition of human ACHN cell growth in a cell viability assay, IC50 = 0.37018 μM. | SANGER | |||
| LS-513 | Growth Inhibition Assay | Inhibition of human LS-513 cell growth in a cell viability assay, IC50 = 0.3795 μM. | SANGER | |||
| TE-5 | Growth Inhibition Assay | Inhibition of human TE-5 cell growth in a cell viability assay, IC50 = 0.3806 μM. | SANGER | |||
| IST-MEL1 | Growth Inhibition Assay | Inhibition of human IST-MEL1 cell growth in a cell viability assay, IC50 = 0.3808 μM. | SANGER | |||
| RS4-11 | Growth Inhibition Assay | Inhibition of human RS4-11 cell growth in a cell viability assay, IC50 = 0.381 μM. | SANGER | |||
| GR-ST | Growth Inhibition Assay | Inhibition of human GR-ST cell growth in a cell viability assay, IC50 = 0.38472 μM. | SANGER | |||
| NCI-H1048 | Growth Inhibition Assay | Inhibition of human NCI-H1048 cell growth in a cell viability assay, IC50 = 0.38876 μM. | SANGER | |||
| ONS-76 | Growth Inhibition Assay | Inhibition of human ONS-76 cell growth in a cell viability assay, IC50 = 0.39003 μM. | SANGER | |||
| H9 | Growth Inhibition Assay | Inhibition of human H9 cell growth in a cell viability assay, IC50 = 0.39142 μM. | SANGER | |||
| GAMG | Growth Inhibition Assay | Inhibition of human GAMG cell growth in a cell viability assay, IC50 = 0.39364 μM. | SANGER | |||
| GMS-10 | Growth Inhibition Assay | Inhibition of human GMS-10 cell growth in a cell viability assay, IC50 = 0.39556 μM. | SANGER | |||
| A375 | Growth Inhibition Assay | Inhibition of human A375 cell growth in a cell viability assay, IC50 = 0.39567 μM. | SANGER | |||
| NCI-H1155 | Growth Inhibition Assay | Inhibition of human NCI-H1155 cell growth in a cell viability assay, IC50 = 0.39685 μM. | SANGER | |||
| OC-314 | Growth Inhibition Assay | Inhibition of human OC-314 cell growth in a cell viability assay, IC50 = 0.39891 μM. | SANGER | |||
| HSC-3 | Growth Inhibition Assay | Inhibition of human HSC-3 cell growth in a cell viability assay, IC50 = 0.39994 μM. | SANGER | |||
| RPMI-2650 | Growth Inhibition Assay | Inhibition of human RPMI-2650 cell growth in a cell viability assay, IC50 = 0.40146 μM. | SANGER | |||
| SK-PN-DW | Growth Inhibition Assay | Inhibition of human SK-PN-DW cell growth in a cell viability assay, IC50 = 0.41557 μM. | SANGER | |||
| MZ7-mel | Growth Inhibition Assay | Inhibition of human MZ7-mel cell growth in a cell viability assay, IC50 = 0.41695 μM. | SANGER | |||
| GOTO | Growth Inhibition Assay | Inhibition of human GOTO cell growth in a cell viability assay, IC50 = 0.42403 μM. | SANGER | |||
| COLO-800 | Growth Inhibition Assay | Inhibition of human COLO-800 cell growth in a cell viability assay, IC50 = 0.42456 μM. | SANGER | |||
| NCI-H2122 | Growth Inhibition Assay | Inhibition of human NCI-H2122 cell growth in a cell viability assay, IC50 = 0.42962 μM. | SANGER | |||
| JEG-3 | Growth Inhibition Assay | Inhibition of human JEG-3 cell growth in a cell viability assay, IC50 = 0.43108 μM. | SANGER | |||
| SK-N-AS | Growth Inhibition Assay | Inhibition of human SK-N-AS cell growth in a cell viability assay, IC50 = 0.45046 μM. | SANGER | |||
| NB5 | Growth Inhibition Assay | Inhibition of human NB5 cell growth in a cell viability assay, IC50 = 0.45227 μM. | SANGER | |||
| HGC-27 | Growth Inhibition Assay | Inhibition of human HGC-27 cell growth in a cell viability assay, IC50 = 0.45523 μM. | SANGER | |||
| HTC-C3 | Growth Inhibition Assay | Inhibition of human HTC-C3 cell growth in a cell viability assay, IC50 = 0.45932 μM. | SANGER | |||
| LK-2 | Growth Inhibition Assay | Inhibition of human LK-2 cell growth in a cell viability assay, IC50 = 0.46056 μM. | SANGER | |||
| HSC-4 | Growth Inhibition Assay | Inhibition of human HSC-4 cell growth in a cell viability assay, IC50 = 0.46741 μM. | SANGER | |||
| SW13 | Growth Inhibition Assay | Inhibition of human SW13 cell growth in a cell viability assay, IC50 = 0.47618 μM. | SANGER | |||
| LCLC-97TM1 | Growth Inhibition Assay | Inhibition of human LCLC-97TM1 cell growth in a cell viability assay, IC50 = 0.47741 μM. | SANGER | |||
| BCPAP | Growth Inhibition Assay | Inhibition of human BCPAP cell growth in a cell viability assay, IC50 = 0.50118 μM. | SANGER | |||
| 5637 | Growth Inhibition Assay | Inhibition of human 5637 cell growth in a cell viability assay, IC50 = 0.50148 μM. | SANGER | |||
| RT-112 | Growth Inhibition Assay | Inhibition of human RT-112 cell growth in a cell viability assay, IC50 = 0.50193 μM. | SANGER | |||
| BV-173 | Growth Inhibition Assay | Inhibition of human BV-173 cell growth in a cell viability assay, IC50 = 0.51551 μM. | SANGER | |||
| HOS | Growth Inhibition Assay | Inhibition of human HOS cell growth in a cell viability assay, IC50 = 0.51822 μM. | SANGER | |||
| BT-549 | Growth Inhibition Assay | Inhibition of human BT-549 cell growth in a cell viability assay, IC50 = 0.53102 μM. | SANGER | |||
| Daoy | Growth Inhibition Assay | Inhibition of human Daoy cell growth in a cell viability assay, IC50 = 0.53222 μM. | SANGER | |||
| MHH-PREB-1 | Growth Inhibition Assay | Inhibition of human MHH-PREB-1 cell growth in a cell viability assay, IC50 = 0.53311 μM. | SANGER | |||
| VM-CUB-1 | Growth Inhibition Assay | Inhibition of human VM-CUB-1 cell growth in a cell viability assay, IC50 = 0.53346 μM. | SANGER | |||
| KP-4 | Growth Inhibition Assay | Inhibition of human KP-4 cell growth in a cell viability assay, IC50 = 0.53574 μM. | SANGER | |||
| NCI-H1623 | Growth Inhibition Assay | Inhibition of human NCI-H1623 cell growth in a cell viability assay, IC50 = 0.536 μM. | SANGER | |||
| OCI-AML2 | Growth Inhibition Assay | Inhibition of human OCI-AML2 cell growth in a cell viability assay, IC50 = 0.54502 μM. | SANGER | |||
| C-33-A | Growth Inhibition Assay | Inhibition of human C-33-A cell growth in a cell viability assay, IC50 = 0.55083 μM. | SANGER | |||
| NCI-H1355 | Growth Inhibition Assay | Inhibition of human NCI-H1355 cell growth in a cell viability assay, IC50 = 0.55554 μM. | SANGER | |||
| SK-HEP-1 | Growth Inhibition Assay | Inhibition of human SK-HEP-1 cell growth in a cell viability assay, IC50 = 0.55641 μM. | SANGER | |||
| EC-GI-10 | Growth Inhibition Assay | Inhibition of human EC-GI-10 cell growth in a cell viability assay, IC50 = 0.55698 μM. | SANGER | |||
| HUTU-80 | Growth Inhibition Assay | Inhibition of human HUTU-80 cell growth in a cell viability assay, IC50 = 0.56122 μM. | SANGER | |||
| KYSE-180 | Growth Inhibition Assay | Inhibition of human KYSE-180 cell growth in a cell viability assay, IC50 = 0.56471 μM. | SANGER | |||
| CAL-51 | Growth Inhibition Assay | Inhibition of human CAL-51 cell growth in a cell viability assay, IC50 = 0.57064 μM. | SANGER | |||
| DU-4475 | Growth Inhibition Assay | Inhibition of human DU-4475 cell growth in a cell viability assay, IC50 = 0.5754 μM. | SANGER | |||
| NUGC-3 | Growth Inhibition Assay | Inhibition of human NUGC-3 cell growth in a cell viability assay, IC50 = 0.58891 μM. | SANGER | |||
| NCI-H2228 | Growth Inhibition Assay | Inhibition of human NCI-H2228 cell growth in a cell viability assay, IC50 = 0.59651 μM. | SANGER | |||
| RKO | Growth Inhibition Assay | Inhibition of human RKO cell growth in a cell viability assay, IC50 = 0.59858 μM. | SANGER | |||
| PC-14 | Growth Inhibition Assay | Inhibition of human PC-14 cell growth in a cell viability assay, IC50 = 0.6095 μM. | SANGER | |||
| TI-73 | Growth Inhibition Assay | Inhibition of human TI-73 cell growth in a cell viability assay, IC50 = 0.61266 μM. | SANGER | |||
| CW-2 | Growth Inhibition Assay | Inhibition of human CW-2 cell growth in a cell viability assay, IC50 = 0.61279 μM. | SANGER | |||
| SN12C | Growth Inhibition Assay | Inhibition of human SN12C cell growth in a cell viability assay, IC50 = 0.62292 μM. | SANGER | |||
| M14 | Growth Inhibition Assay | Inhibition of human M14 cell growth in a cell viability assay, IC50 = 0.63224 μM. | SANGER | |||
| KOSC-2 | Growth Inhibition Assay | Inhibition of human KOSC-2 cell growth in a cell viability assay, IC50 = 0.64309 μM. | SANGER | |||
| G-402 | Growth Inhibition Assay | Inhibition of human G-402 cell growth in a cell viability assay, IC50 = 0.64977 μM. | SANGER | |||
| TYK-nu | Growth Inhibition Assay | Inhibition of human TYK-nu cell growth in a cell viability assay, IC50 = 0.66085 μM. | SANGER | |||
| UACC-62 | Growth Inhibition Assay | Inhibition of human UACC-62 cell growth in a cell viability assay, IC50 = 0.66553 μM. | SANGER | |||
| EW-22 | Growth Inhibition Assay | Inhibition of human EW-22 cell growth in a cell viability assay, IC50 = 0.66762 μM. | SANGER | |||
| 786-0 | Growth Inhibition Assay | Inhibition of human 786-0 cell growth in a cell viability assay, IC50 = 0.66871 μM. | SANGER | |||
| LoVo | Growth Inhibition Assay | Inhibition of human LoVo cell growth in a cell viability assay, IC50 = 0.67124 μM. | SANGER | |||
| NCI-H1299 | Growth Inhibition Assay | Inhibition of human NCI-H1299 cell growth in a cell viability assay, IC50 = 0.67177 μM. | SANGER | |||
| Saos-2 | Growth Inhibition Assay | Inhibition of human Saos-2 cell growth in a cell viability assay, IC50 = 0.6817 μM. | SANGER | |||
| SJRH30 | Growth Inhibition Assay | Inhibition of human SJRH30 cell growth in a cell viability assay, IC50 = 0.68338 μM. | SANGER | |||
| A2058 | Growth Inhibition Assay | Inhibition of human A2058 cell growth in a cell viability assay, IC50 = 0.68678 μM. | SANGER | |||
| HO-1-N-1 | Growth Inhibition Assay | Inhibition of human HO-1-N-1 cell growth in a cell viability assay, IC50 = 0.69055 μM. | SANGER | |||
| SF126 | Growth Inhibition Assay | Inhibition of human SF126 cell growth in a cell viability assay, IC50 = 0.69317 μM. | SANGER | |||
| LS-411N | Growth Inhibition Assay | Inhibition of human LS-411N cell growth in a cell viability assay, IC50 = 0.69527 μM. | SANGER | |||
| RXF393 | Growth Inhibition Assay | Inhibition of human RXF393 cell growth in a cell viability assay, IC50 = 0.70032 μM. | SANGER | |||
| CAL-62 | Growth Inhibition Assay | Inhibition of human CAL-62 cell growth in a cell viability assay, IC50 = 0.70983 μM. | SANGER | |||
| SF539 | Growth Inhibition Assay | Inhibition of human SF539 cell growth in a cell viability assay, IC50 = 0.71677 μM. | SANGER | |||
| SW756 | Growth Inhibition Assay | Inhibition of human SW756 cell growth in a cell viability assay, IC50 = 0.71942 μM. | SANGER | |||
| ES6 | Growth Inhibition Assay | Inhibition of human ES6 cell growth in a cell viability assay, IC50 = 0.72144 μM. | SANGER | |||
| MHH-NB-11 | Growth Inhibition Assay | Inhibition of human MHH-NB-11 cell growth in a cell viability assay, IC50 = 0.72787 μM. | SANGER | |||
| A388 | Growth Inhibition Assay | Inhibition of human A388 cell growth in a cell viability assay, IC50 = 0.73992 μM. | SANGER | |||
| RPMI-8866 | Growth Inhibition Assay | Inhibition of human RPMI-8866 cell growth in a cell viability assay, IC50 = 0.74014 μM. | SANGER | |||
| HD-MY-Z | Growth Inhibition Assay | Inhibition of human HD-MY-Z cell growth in a cell viability assay, IC50 = 0.74471 μM. | SANGER | |||
| G-401 | Growth Inhibition Assay | Inhibition of human G-401 cell growth in a cell viability assay, IC50 = 0.75823 μM. | SANGER | |||
| EW-24 | Growth Inhibition Assay | Inhibition of human EW-24 cell growth in a cell viability assay, IC50 = 0.79393 μM. | SANGER | |||
| COR-L23 | Growth Inhibition Assay | Inhibition of human COR-L23 cell growth in a cell viability assay, IC50 = 0.79673 μM. | SANGER | |||
| NCI-H1437 | Growth Inhibition Assay | Inhibition of human NCI-H1437 cell growth in a cell viability assay, IC50 = 0.79806 μM. | SANGER | |||
| SK-MEL-28 | Growth Inhibition Assay | Inhibition of human SK-MEL-28 cell growth in a cell viability assay, IC50 = 0.79873 μM. | SANGER | |||
| PANC-10-05 | Growth Inhibition Assay | Inhibition of human PANC-10-05 cell growth in a cell viability assay, IC50 = 0.80176 μM. | SANGER | |||
| EoL-1-cell | Growth Inhibition Assay | Inhibition of human EoL-1-cell cell growth in a cell viability assay, IC50 = 0.8031 μM. | SANGER | |||
| D-336MG | Growth Inhibition Assay | Inhibition of human D-336MG cell growth in a cell viability assay, IC50 = 0.80494 μM. | SANGER | |||
| SK-MES-1 | Growth Inhibition Assay | Inhibition of human SK-MES-1 cell growth in a cell viability assay, IC50 = 0.80682 μM. | SANGER | |||
| COLO-668 | Growth Inhibition Assay | Inhibition of human COLO-668 cell growth in a cell viability assay, IC50 = 0.80916 μM. | SANGER | |||
| BE-13 | Growth Inhibition Assay | Inhibition of human BE-13 cell growth in a cell viability assay, IC50 = 0.8245 μM. | SANGER | |||
| EW-3 | Growth Inhibition Assay | Inhibition of human EW-3 cell growth in a cell viability assay, IC50 = 0.82999 μM. | SANGER | |||
| OMC-1 | Growth Inhibition Assay | Inhibition of human OMC-1 cell growth in a cell viability assay, IC50 = 0.83236 μM. | SANGER | |||
| Hs-578-T | Growth Inhibition Assay | Inhibition of human Hs-578-T cell growth in a cell viability assay, IC50 = 0.83886 μM. | SANGER | |||
| A3-KAW | Growth Inhibition Assay | Inhibition of human A3-KAW cell growth in a cell viability assay, IC50 = 0.84424 μM. | SANGER | |||
| KYSE-270 | Growth Inhibition Assay | Inhibition of human KYSE-270 cell growth in a cell viability assay, IC50 = 0.84868 μM. | SANGER | |||
| HuP-T3 | Growth Inhibition Assay | Inhibition of human HuP-T3 cell growth in a cell viability assay, IC50 = 0.85567 μM. | SANGER | |||
| GCT | Growth Inhibition Assay | Inhibition of human GCT cell growth in a cell viability assay, IC50 = 0.85568 μM. | SANGER | |||
| SiHa | Growth Inhibition Assay | Inhibition of human SiHa cell growth in a cell viability assay, IC50 = 0.85606 μM. | SANGER | |||
| Ramos-2G6-4C10 | Growth Inhibition Assay | Inhibition of human Ramos-2G6-4C10 cell growth in a cell viability assay, IC50 = 0.86757 μM. | SANGER | |||
| DEL | Growth Inhibition Assay | Inhibition of human DEL cell growth in a cell viability assay, IC50 = 0.87485 μM. | SANGER | |||
| SNU-423 | Growth Inhibition Assay | Inhibition of human SNU-423 cell growth in a cell viability assay, IC50 = 0.8808 μM. | SANGER | |||
| PANC-03-27 | Growth Inhibition Assay | Inhibition of human PANC-03-27 cell growth in a cell viability assay, IC50 = 0.89662 μM. | SANGER | |||
| BHY | Growth Inhibition Assay | Inhibition of human BHY cell growth in a cell viability assay, IC50 = 0.89762 μM. | SANGER | |||
| DOK | Growth Inhibition Assay | Inhibition of human DOK cell growth in a cell viability assay, IC50 = 0.90446 μM. | SANGER | |||
| SKG-IIIa | Growth Inhibition Assay | Inhibition of human SKG-IIIa cell growth in a cell viability assay, IC50 = 0.91503 μM. | SANGER | |||
| AGS | Growth Inhibition Assay | Inhibition of human AGS cell growth in a cell viability assay, IC50 = 0.91755 μM. | SANGER | |||
| MN-60 | Growth Inhibition Assay | Inhibition of human MN-60 cell growth in a cell viability assay, IC50 = 0.93484 μM. | SANGER | |||
| CAL-54 | Growth Inhibition Assay | Inhibition of human CAL-54 cell growth in a cell viability assay, IC50 = 0.94033 μM. | SANGER | |||
| KMOE-2 | Growth Inhibition Assay | Inhibition of human KMOE-2 cell growth in a cell viability assay, IC50 = 0.94389 μM. | SANGER | |||
| SCC-4 | Growth Inhibition Assay | Inhibition of human SCC-4 cell growth in a cell viability assay, IC50 = 0.96513 μM. | SANGER | |||
| CAL-39 | Growth Inhibition Assay | Inhibition of human CAL-39 cell growth in a cell viability assay, IC50 = 0.99829 μM. | SANGER | |||
| CHL-1 | Growth Inhibition Assay | Inhibition of human CHL-1 cell growth in a cell viability assay, IC50 = 1.04316 μM. | SANGER | |||
| NCI-H2030 | Growth Inhibition Assay | Inhibition of human NCI-H2030 cell growth in a cell viability assay, IC50 = 1.04946 μM. | SANGER | |||
| COLO-792 | Growth Inhibition Assay | Inhibition of human COLO-792 cell growth in a cell viability assay, IC50 = 1.05355 μM. | SANGER | |||
| CFPAC-1 | Growth Inhibition Assay | Inhibition of human CFPAC-1 cell growth in a cell viability assay, IC50 = 1.05515 μM. | SANGER | |||
| NKM-1 | Growth Inhibition Assay | Inhibition of human NKM-1 cell growth in a cell viability assay, IC50 = 1.06403 μM. | SANGER | |||
| SK-N-DZ | Growth Inhibition Assay | Inhibition of human SK-N-DZ cell growth in a cell viability assay, IC50 = 1.07213 μM. | SANGER | |||
| HC-1 | Growth Inhibition Assay | Inhibition of human HC-1 cell growth in a cell viability assay, IC50 = 1.07779 μM. | SANGER | |||
| SCC-15 | Growth Inhibition Assay | Inhibition of human SCC-15 cell growth in a cell viability assay, IC50 = 1.08069 μM. | SANGER | |||
| D-247MG | Growth Inhibition Assay | Inhibition of human D-247MG cell growth in a cell viability assay, IC50 = 1.08967 μM. | SANGER | |||
| KYSE-450 | Growth Inhibition Assay | Inhibition of human KYSE-450 cell growth in a cell viability assay, IC50 = 1.09059 μM. | SANGER | |||
| Becker | Growth Inhibition Assay | Inhibition of human Becker cell growth in a cell viability assay, IC50 = 1.09665 μM. | SANGER | |||
| EGI-1 | Growth Inhibition Assay | Inhibition of human EGI-1 cell growth in a cell viability assay, IC50 = 1.10918 μM. | SANGER | |||
| U251 | Growth Inhibition Assay | Inhibition of human U251 cell growth in a cell viability assay, IC50 = 1.12999 μM. | SANGER | |||
| KG-1 | Growth Inhibition Assay | Inhibition of human KG-1 cell growth in a cell viability assay, IC50 = 1.15685 μM. | SANGER | |||
| NH-12 | Growth Inhibition Assay | Inhibition of human NH-12 cell growth in a cell viability assay, IC50 = 1.16227 μM. | SANGER | |||
| D-263MG | Growth Inhibition Assay | Inhibition of human D-263MG cell growth in a cell viability assay, IC50 = 1.1717 μM. | SANGER | |||
| A4-Fuk | Growth Inhibition Assay | Inhibition of human A4-Fuk cell growth in a cell viability assay, IC50 = 1.17349 μM. | SANGER | |||
| CAL-33 | Growth Inhibition Assay | Inhibition of human CAL-33 cell growth in a cell viability assay, IC50 = 1.17622 μM. | SANGER | |||
| NCI-H23 | Growth Inhibition Assay | Inhibition of human NCI-H23 cell growth in a cell viability assay, IC50 = 1.19572 μM. | SANGER | |||
| EW-11 | Growth Inhibition Assay | Inhibition of human EW-11 cell growth in a cell viability assay, IC50 = 1.21388 μM. | SANGER | |||
| HT-144 | Growth Inhibition Assay | Inhibition of human HT-144 cell growth in a cell viability assay, IC50 = 1.23101 μM. | SANGER | |||
| 647-V | Growth Inhibition Assay | Inhibition of human 647-V cell growth in a cell viability assay, IC50 = 1.23274 μM. | SANGER | |||
| Mewo | Growth Inhibition Assay | Inhibition of human Mewo cell growth in a cell viability assay, IC50 = 1.23969 μM. | SANGER | |||
| OAW-42 | Growth Inhibition Assay | Inhibition of human OAW-42 cell growth in a cell viability assay, IC50 = 1.24927 μM. | SANGER | |||
| U031 | Growth Inhibition Assay | Inhibition of human U031 cell growth in a cell viability assay, IC50 = 1.25079 μM. | SANGER | |||
| RD | Growth Inhibition Assay | Inhibition of human RD cell growth in a cell viability assay, IC50 = 1.27311 μM. | SANGER | |||
| PC-3 | Growth Inhibition Assay | Inhibition of human PC-3 cell growth in a cell viability assay, IC50 = 1.27636 μM. | SANGER | |||
| MC116 | Growth Inhibition Assay | Inhibition of human MC116 cell growth in a cell viability assay, IC50 = 1.28349 μM. | SANGER | |||
| LNCaP-Clone-FGC | Growth Inhibition Assay | Inhibition of human LNCaP-Clone-FGC cell growth in a cell viability assay, IC50 = 1.31198 μM. | SANGER | |||
| SK-LU-1 | Growth Inhibition Assay | Inhibition of human SK-LU-1 cell growth in a cell viability assay, IC50 = 1.31214 μM. | SANGER | |||
| MPP-89 | Growth Inhibition Assay | Inhibition of human MPP-89 cell growth in a cell viability assay, IC50 = 1.31316 μM. | SANGER | |||
| SW780 | Growth Inhibition Assay | Inhibition of human SW780 cell growth in a cell viability assay, IC50 = 1.32041 μM. | SANGER | |||
| HuO-3N1 | Growth Inhibition Assay | Inhibition of human HuO-3N1 cell growth in a cell viability assay, IC50 = 1.32628 μM. | SANGER | |||
| L-363 | Growth Inhibition Assay | Inhibition of human L-363 cell growth in a cell viability assay, IC50 = 1.33929 μM. | SANGER | |||
| VMRC-RCZ | Growth Inhibition Assay | Inhibition of human VMRC-RCZ cell growth in a cell viability assay, IC50 = 1.35399 μM. | SANGER | |||
| SF268 | Growth Inhibition Assay | Inhibition of human SF268 cell growth in a cell viability assay, IC50 = 1.36068 μM. | SANGER | |||
| DOHH-2 | Growth Inhibition Assay | Inhibition of human DOHH-2 cell growth in a cell viability assay, IC50 = 1.3786 μM. | SANGER | |||
| HT-3 | Growth Inhibition Assay | Inhibition of human HT-3 cell growth in a cell viability assay, IC50 = 1.38152 μM. | SANGER | |||
| FTC-133 | Growth Inhibition Assay | Inhibition of human FTC-133 cell growth in a cell viability assay, IC50 = 1.38559 μM. | SANGER | |||
| MDA-MB-157 | Growth Inhibition Assay | Inhibition of human MDA-MB-157 cell growth in a cell viability assay, IC50 = 1.38917 μM. | SANGER | |||
| AsPC-1 | Growth Inhibition Assay | Inhibition of human AsPC-1 cell growth in a cell viability assay, IC50 = 1.39216 μM. | SANGER | |||
| NCI-H1792 | Growth Inhibition Assay | Inhibition of human NCI-H1792 cell growth in a cell viability assay, IC50 = 1.42426 μM. | SANGER | |||
| MZ2-MEL | Growth Inhibition Assay | Inhibition of human MZ2-MEL cell growth in a cell viability assay, IC50 = 1.43438 μM. | SANGER | |||
| EPLC-272H | Growth Inhibition Assay | Inhibition of human EPLC-272H cell growth in a cell viability assay, IC50 = 1.44992 μM. | SANGER | |||
| JVM-3 | Growth Inhibition Assay | Inhibition of human JVM-3 cell growth in a cell viability assay, IC50 = 1.4541 μM. | SANGER | |||
| HSC-2 | Growth Inhibition Assay | Inhibition of human HSC-2 cell growth in a cell viability assay, IC50 = 1.45612 μM. | SANGER | |||
| DMS-273 | Growth Inhibition Assay | Inhibition of human DMS-273 cell growth in a cell viability assay, IC50 = 1.45632 μM. | SANGER | |||
| AM-38 | Growth Inhibition Assay | Inhibition of human AM-38 cell growth in a cell viability assay, IC50 = 1.46755 μM. | SANGER | |||
| MIA-PaCa-2 | Growth Inhibition Assay | Inhibition of human MIA-PaCa-2 cell growth in a cell viability assay, IC50 = 1.48862 μM. | SANGER | |||
| NB7 | Growth Inhibition Assay | Inhibition of human NB7 cell growth in a cell viability assay, IC50 = 1.51552 μM. | SANGER | |||
| ABC-1 | Growth Inhibition Assay | Inhibition of human ABC-1 cell growth in a cell viability assay, IC50 = 1.52053 μM. | SANGER | |||
| EFO-21 | Growth Inhibition Assay | Inhibition of human EFO-21 cell growth in a cell viability assay, IC50 = 1.54986 μM. | SANGER | |||
| NCI-H1666 | Growth Inhibition Assay | Inhibition of human NCI-H1666 cell growth in a cell viability assay, IC50 = 1.5762 μM. | SANGER | |||
| OS-RC-2 | Growth Inhibition Assay | Inhibition of human OS-RC-2 cell growth in a cell viability assay, IC50 = 1.58228 μM. | SANGER | |||
| HLE | Growth Inhibition Assay | Inhibition of human HLE cell growth in a cell viability assay, IC50 = 1.59175 μM. | SANGER | |||
| DoTc2-4510 | Growth Inhibition Assay | Inhibition of human DoTc2-4510 cell growth in a cell viability assay, IC50 = 1.59856 μM. | SANGER | |||
| J82 | Growth Inhibition Assay | Inhibition of human J82 cell growth in a cell viability assay, IC50 = 1.61802 μM. | SANGER | |||
| SW900 | Growth Inhibition Assay | Inhibition of human SW900 cell growth in a cell viability assay, IC50 = 1.62161 μM. | SANGER | |||
| MKN45 | Growth Inhibition Assay | Inhibition of human MKN45 cell growth in a cell viability assay, IC50 = 1.64811 μM. | SANGER | |||
| GI-ME-N | Growth Inhibition Assay | Inhibition of human GI-ME-N cell growth in a cell viability assay, IC50 = 1.65967 μM. | SANGER | |||
| DMS-114 | Growth Inhibition Assay | Inhibition of human DMS-114 cell growth in a cell viability assay, IC50 = 1.68124 μM. | SANGER | |||
| HT-1376 | Growth Inhibition Assay | Inhibition of human HT-1376 cell growth in a cell viability assay, IC50 = 1.68168 μM. | SANGER | |||
| T98G | Growth Inhibition Assay | Inhibition of human T98G cell growth in a cell viability assay, IC50 = 1.71811 μM. | SANGER | |||
| SW1710 | Growth Inhibition Assay | Inhibition of human SW1710 cell growth in a cell viability assay, IC50 = 1.72915 μM. | SANGER | |||
| TGBC11TKB | Growth Inhibition Assay | Inhibition of human TGBC11TKB cell growth in a cell viability assay, IC50 = 1.73145 μM. | SANGER | |||
| TK10 | Growth Inhibition Assay | Inhibition of human TK10 cell growth in a cell viability assay, IC50 = 1.73331 μM. | SANGER | |||
| NCI-H661 | Growth Inhibition Assay | Inhibition of human NCI-H661 cell growth in a cell viability assay, IC50 = 1.73446 μM. | SANGER | |||
| MKN1 | Growth Inhibition Assay | Inhibition of human MKN1 cell growth in a cell viability assay, IC50 = 1.73948 μM. | SANGER | |||
| U-87-MG | Growth Inhibition Assay | Inhibition of human U-87-MG cell growth in a cell viability assay, IC50 = 1.74257 μM. | SANGER | |||
| RERF-LC-MS | Growth Inhibition Assay | Inhibition of human RERF-LC-MS cell growth in a cell viability assay, IC50 = 1.76445 μM. | SANGER | |||
| NCI-H1581 | Growth Inhibition Assay | Inhibition of human NCI-H1581 cell growth in a cell viability assay, IC50 = 1.76955 μM. | SANGER | |||
| IGR-1 | Growth Inhibition Assay | Inhibition of human IGR-1 cell growth in a cell viability assay, IC50 = 1.78279 μM. | SANGER | |||
| TE-9 | Growth Inhibition Assay | Inhibition of human TE-9 cell growth in a cell viability assay, IC50 = 1.78754 μM. | SANGER | |||
| HEC-1 | Growth Inhibition Assay | Inhibition of human HEC-1 cell growth in a cell viability assay, IC50 = 1.78799 μM. | SANGER | |||
| IGROV-1 | Growth Inhibition Assay | Inhibition of human IGROV-1 cell growth in a cell viability assay, IC50 = 1.78989 μM. | SANGER | |||
| OE33 | Growth Inhibition Assay | Inhibition of human OE33 cell growth in a cell viability assay, IC50 = 1.80243 μM. | SANGER | |||
| MZ1-PC | Growth Inhibition Assay | Inhibition of human MZ1-PC cell growth in a cell viability assay, IC50 = 1.80338 μM. | SANGER | |||
| NCI-H28 | Growth Inhibition Assay | Inhibition of human NCI-H28 cell growth in a cell viability assay, IC50 = 1.82221 μM. | SANGER | |||
| M059J | Growth Inhibition Assay | Inhibition of human M059J cell growth in a cell viability assay, IC50 = 1.82297 μM. | SANGER | |||
| AN3-CA | Growth Inhibition Assay | Inhibition of human AN3-CA cell growth in a cell viability assay, IC50 = 1.84009 μM. | SANGER | |||
| MV-4-11 | Growth Inhibition Assay | Inhibition of human MV-4-11 cell growth in a cell viability assay, IC50 = 1.8467 μM. | SANGER | |||
| TGBC24TKB | Growth Inhibition Assay | Inhibition of human TGBC24TKB cell growth in a cell viability assay, IC50 = 1.8517 μM. | SANGER | |||
| KYSE-70 | Growth Inhibition Assay | Inhibition of human KYSE-70 cell growth in a cell viability assay, IC50 = 1.85716 μM. | SANGER | |||
| COLO-829 | Growth Inhibition Assay | Inhibition of human COLO-829 cell growth in a cell viability assay, IC50 = 1.86471 μM. | SANGER | |||
| HCC38 | Growth Inhibition Assay | Inhibition of human HCC38 cell growth in a cell viability assay, IC50 = 1.87965 μM. | SANGER | |||
| NOS-1 | Growth Inhibition Assay | Inhibition of human NOS-1 cell growth in a cell viability assay, IC50 = 1.90764 μM. | SANGER | |||
| CAL-27 | Growth Inhibition Assay | Inhibition of human CAL-27 cell growth in a cell viability assay, IC50 = 1.92856 μM. | SANGER | |||
| EFO-27 | Growth Inhibition Assay | Inhibition of human EFO-27 cell growth in a cell viability assay, IC50 = 1.93966 μM. | SANGER | |||
| NMC-G1 | Growth Inhibition Assay | Inhibition of human NMC-G1 cell growth in a cell viability assay, IC50 = 1.95708 μM. | SANGER | |||
| EW-13 | Growth Inhibition Assay | Inhibition of human EW-13 cell growth in a cell viability assay, IC50 = 1.9815 μM. | SANGER | |||
| MDA-MB-175-VII | Growth Inhibition Assay | Inhibition of human MDA-MB-175-VII cell growth in a cell viability assay, IC50 = 1.99141 μM. | SANGER | |||
| MCF7 | Growth Inhibition Assay | Inhibition of human MCF7 cell growth in a cell viability assay, IC50 = 2.00248 μM. | SANGER | |||
| Detroit562 | Growth Inhibition Assay | Inhibition of human Detroit562 cell growth in a cell viability assay, IC50 = 2.0621 μM. | SANGER | |||
| KNS-62 | Growth Inhibition Assay | Inhibition of human KNS-62 cell growth in a cell viability assay, IC50 = 2.07553 μM. | SANGER | |||
| MDA-MB-453 | Growth Inhibition Assay | Inhibition of human MDA-MB-453 cell growth in a cell viability assay, IC50 = 2.10739 μM. | SANGER | |||
| KASUMI-1 | Growth Inhibition Assay | Inhibition of human KASUMI-1 cell growth in a cell viability assay, IC50 = 2.16268 μM. | SANGER | |||
| FADU | Growth Inhibition Assay | Inhibition of human FADU cell growth in a cell viability assay, IC50 = 2.16998 μM. | SANGER | |||
| KYSE-410 | Growth Inhibition Assay | Inhibition of human KYSE-410 cell growth in a cell viability assay, IC50 = 2.19194 μM. | SANGER | |||
| IST-SL1 | Growth Inhibition Assay | Inhibition of human IST-SL1 cell growth in a cell viability assay, IC50 = 2.20154 μM. | SANGER | |||
| NCI-H2009 | Growth Inhibition Assay | Inhibition of human NCI-H2009 cell growth in a cell viability assay, IC50 = 2.25295 μM. | SANGER | |||
| ETK-1 | Growth Inhibition Assay | Inhibition of human ETK-1 cell growth in a cell viability assay, IC50 = 2.3042 μM. | SANGER | |||
| COLO-741 | Growth Inhibition Assay | Inhibition of human COLO-741 cell growth in a cell viability assay, IC50 = 2.32544 μM. | SANGER | |||
| NOMO-1 | Growth Inhibition Assay | Inhibition of human NOMO-1 cell growth in a cell viability assay, IC50 = 2.33232 μM. | SANGER | |||
| KU812 | Growth Inhibition Assay | Inhibition of human KU812 cell growth in a cell viability assay, IC50 = 2.3578 μM. | SANGER | |||
| MEG-01 | Growth Inhibition Assay | Inhibition of human MEG-01 cell growth in a cell viability assay, IC50 = 2.38965 μM. | SANGER | |||
| NCI-H1755 | Growth Inhibition Assay | Inhibition of human NCI-H1755 cell growth in a cell viability assay, IC50 = 2.41912 μM. | SANGER | |||
| MKN28 | Growth Inhibition Assay | Inhibition of human MKN28 cell growth in a cell viability assay, IC50 = 2.41927 μM. | SANGER | |||
| HL-60 | Growth Inhibition Assay | Inhibition of human HL-60 cell growth in a cell viability assay, IC50 = 2.44486 μM. | SANGER | |||
| SCC-9 | Growth Inhibition Assay | Inhibition of human SCC-9 cell growth in a cell viability assay, IC50 = 2.44796 μM. | SANGER | |||
| NCI-H226 | Growth Inhibition Assay | Inhibition of human NCI-H226 cell growth in a cell viability assay, IC50 = 2.52099 μM. | SANGER | |||
| KYSE-520 | Growth Inhibition Assay | Inhibition of human KYSE-520 cell growth in a cell viability assay, IC50 = 2.54159 μM. | SANGER | |||
| MG-63 | Growth Inhibition Assay | Inhibition of human MG-63 cell growth in a cell viability assay, IC50 = 2.55986 μM. | SANGER | |||
| HCC2998 | Growth Inhibition Assay | Inhibition of human HCC2998 cell growth in a cell viability assay, IC50 = 2.57816 μM. | SANGER | |||
| DBTRG-05MG | Growth Inhibition Assay | Inhibition of human DBTRG-05MG cell growth in a cell viability assay, IC50 = 2.61987 μM. | SANGER | |||
| HH | Growth Inhibition Assay | Inhibition of human HH cell growth in a cell viability assay, IC50 = 2.64005 μM. | SANGER | |||
| CAS-1 | Growth Inhibition Assay | Inhibition of human CAS-1 cell growth in a cell viability assay, IC50 = 2.64247 μM. | SANGER | |||
| NCI-N87 | Growth Inhibition Assay | Inhibition of human NCI-N87 cell growth in a cell viability assay, IC50 = 2.67652 μM. | SANGER | |||
| SW1573 | Growth Inhibition Assay | Inhibition of human SW1573 cell growth in a cell viability assay, IC50 = 2.67671 μM. | SANGER | |||
| LU-134-A | Growth Inhibition Assay | Inhibition of human LU-134-A cell growth in a cell viability assay, IC50 = 2.72472 μM. | SANGER | |||
| KYSE-140 | Growth Inhibition Assay | Inhibition of human KYSE-140 cell growth in a cell viability assay, IC50 = 2.7526 μM. | SANGER | |||
| 22RV1 | Growth Inhibition Assay | Inhibition of human 22RV1 cell growth in a cell viability assay, IC50 = 2.7581 μM. | SANGER | |||
| SW626 | Growth Inhibition Assay | Inhibition of human SW626 cell growth in a cell viability assay, IC50 = 2.7603 μM. | SANGER | |||
| MS-1 | Growth Inhibition Assay | Inhibition of human MS-1 cell growth in a cell viability assay, IC50 = 2.76845 μM. | SANGER | |||
| ACN | Growth Inhibition Assay | Inhibition of human ACN cell growth in a cell viability assay, IC50 = 2.79438 μM. | SANGER | |||
| GCIY | Growth Inhibition Assay | Inhibition of human GCIY cell growth in a cell viability assay, IC50 = 2.80234 μM. | SANGER | |||
| Ca-Ski | Growth Inhibition Assay | Inhibition of human Ca-Ski cell growth in a cell viability assay, IC50 = 2.86167 μM. | SANGER | |||
| SK-MEL-1 | Growth Inhibition Assay | Inhibition of human SK-MEL-1 cell growth in a cell viability assay, IC50 = 2.90249 μM. | SANGER | |||
| S-117 | Growth Inhibition Assay | Inhibition of human S-117 cell growth in a cell viability assay, IC50 = 2.92422 μM. | SANGER | |||
| HuCCT1 | Growth Inhibition Assay | Inhibition of human HuCCT1 cell growth in a cell viability assay, IC50 = 2.93645 μM. | SANGER | |||
| TE-11 | Growth Inhibition Assay | Inhibition of human TE-11 cell growth in a cell viability assay, IC50 = 2.95427 μM. | SANGER | |||
| MFH-ino | Growth Inhibition Assay | Inhibition of human MFH-ino cell growth in a cell viability assay, IC50 = 2.956 μM. | SANGER | |||
| MDA-MB-415 | Growth Inhibition Assay | Inhibition of human MDA-MB-415 cell growth in a cell viability assay, IC50 = 2.9585 μM. | SANGER | |||
| EFM-19 | Growth Inhibition Assay | Inhibition of human EFM-19 cell growth in a cell viability assay, IC50 = 2.98059 μM. | SANGER | |||
| NCI-H2405 | Growth Inhibition Assay | Inhibition of human NCI-H2405 cell growth in a cell viability assay, IC50 = 3.00449 μM. | SANGER | |||
| HCC1395 | Growth Inhibition Assay | Inhibition of human HCC1395 cell growth in a cell viability assay, IC50 = 3.0071 μM. | SANGER | |||
| NCI-H2452 | Growth Inhibition Assay | Inhibition of human NCI-H2452 cell growth in a cell viability assay, IC50 = 3.12218 μM. | SANGER | |||
| CAL-72 | Growth Inhibition Assay | Inhibition of human CAL-72 cell growth in a cell viability assay, IC50 = 3.1454 μM. | SANGER | |||
| LB2518-MEL | Growth Inhibition Assay | Inhibition of human LB2518-MEL cell growth in a cell viability assay, IC50 = 3.20734 μM. | SANGER | |||
| NCI-H1395 | Growth Inhibition Assay | Inhibition of human NCI-H1395 cell growth in a cell viability assay, IC50 = 3.24713 μM. | SANGER | |||
| SCH | Growth Inhibition Assay | Inhibition of human SCH cell growth in a cell viability assay, IC50 = 3.26893 μM. | SANGER | |||
| CAPAN-1 | Growth Inhibition Assay | Inhibition of human CAPAN-1 cell growth in a cell viability assay, IC50 = 3.27528 μM. | SANGER | |||
| HT55 | Growth Inhibition Assay | Inhibition of human HT55 cell growth in a cell viability assay, IC50 = 3.28944 μM. | SANGER | |||
| NCI-H747 | Growth Inhibition Assay | Inhibition of human NCI-H747 cell growth in a cell viability assay, IC50 = 3.29752 μM. | SANGER | |||
| TCCSUP | Growth Inhibition Assay | Inhibition of human TCCSUP cell growth in a cell viability assay, IC50 = 3.30446 μM. | SANGER | |||
| SW1088 | Growth Inhibition Assay | Inhibition of human SW1088 cell growth in a cell viability assay, IC50 = 3.31361 μM. | SANGER | |||
| NCI-H2170 | Growth Inhibition Assay | Inhibition of human NCI-H2170 cell growth in a cell viability assay, IC50 = 3.35641 μM. | SANGER | |||
| D-392MG | Growth Inhibition Assay | Inhibition of human D-392MG cell growth in a cell viability assay, IC50 = 3.40336 μM. | SANGER | |||
| DSH1 | Growth Inhibition Assay | Inhibition of human DSH1 cell growth in a cell viability assay, IC50 = 3.40884 μM. | SANGER | |||
| AU565 | Growth Inhibition Assay | Inhibition of human AU565 cell growth in a cell viability assay, IC50 = 3.47021 μM. | SANGER | |||
| A172 | Growth Inhibition Assay | Inhibition of human A172 cell growth in a cell viability assay, IC50 = 3.5178 μM. | SANGER | |||
| HT-29 | Growth Inhibition Assay | Inhibition of human HT-29 cell growth in a cell viability assay, IC50 = 3.52424 μM. | SANGER | |||
| SK-NEP-1 | Growth Inhibition Assay | Inhibition of human SK-NEP-1 cell growth in a cell viability assay, IC50 = 3.54517 μM. | SANGER | |||
| IST-MES1 | Growth Inhibition Assay | Inhibition of human IST-MES1 cell growth in a cell viability assay, IC50 = 3.55449 μM. | SANGER | |||
| KARPAS-45 | Growth Inhibition Assay | Inhibition of human KARPAS-45 cell growth in a cell viability assay, IC50 = 3.60977 μM. | SANGER | |||
| HCC70 | Growth Inhibition Assay | Inhibition of human HCC70 cell growth in a cell viability assay, IC50 = 3.62428 μM. | SANGER | |||
| BPH-1 | Growth Inhibition Assay | Inhibition of human BPH-1 cell growth in a cell viability assay, IC50 = 3.64205 μM. | SANGER | |||
| 8505C | Growth Inhibition Assay | Inhibition of human 8505C cell growth in a cell viability assay, IC50 = 3.70201 μM. | SANGER | |||
| A498 | Growth Inhibition Assay | Inhibition of human A498 cell growth in a cell viability assay, IC50 = 3.72949 μM. | SANGER | |||
| SW962 | Growth Inhibition Assay | Inhibition of human SW962 cell growth in a cell viability assay, IC50 = 3.73962 μM. | SANGER | |||
| SW1990 | Growth Inhibition Assay | Inhibition of human SW1990 cell growth in a cell viability assay, IC50 = 3.82795 μM. | SANGER | |||
| HuO9 | Growth Inhibition Assay | Inhibition of human HuO9 cell growth in a cell viability assay, IC50 = 3.85347 μM. | SANGER | |||
| NCI-H446 | Growth Inhibition Assay | Inhibition of human NCI-H446 cell growth in a cell viability assay, IC50 = 3.9054 μM. | SANGER | |||
| TE-10 | Growth Inhibition Assay | Inhibition of human TE-10 cell growth in a cell viability assay, IC50 = 3.90867 μM. | SANGER | |||
| WM-115 | Growth Inhibition Assay | Inhibition of human WM-115 cell growth in a cell viability assay, IC50 = 3.99333 μM. | SANGER | |||
| RCM-1 | Growth Inhibition Assay | Inhibition of human RCM-1 cell growth in a cell viability assay, IC50 = 4.01309 μM. | SANGER | |||
| SK-MEL-2 | Growth Inhibition Assay | Inhibition of human SK-MEL-2 cell growth in a cell viability assay, IC50 = 4.0281 μM. | SANGER | |||
| SF295 | Growth Inhibition Assay | Inhibition of human SF295 cell growth in a cell viability assay, IC50 = 4.03391 μM. | SANGER | |||
| KINGS-1 | Growth Inhibition Assay | Inhibition of human KINGS-1 cell growth in a cell viability assay, IC50 = 4.05297 μM. | SANGER | |||
| NCI-H2052 | Growth Inhibition Assay | Inhibition of human NCI-H2052 cell growth in a cell viability assay, IC50 = 4.05679 μM. | SANGER | |||
| NCI-H1650 | Growth Inhibition Assay | Inhibition of human NCI-H1650 cell growth in a cell viability assay, IC50 = 4.13216 μM. | SANGER | |||
| U-118-MG | Growth Inhibition Assay | Inhibition of human U-118-MG cell growth in a cell viability assay, IC50 = 4.15547 μM. | SANGER | |||
| MDA-MB-361 | Growth Inhibition Assay | Inhibition of human MDA-MB-361 cell growth in a cell viability assay, IC50 = 4.22726 μM. | SANGER | |||
| HCC1419 | Growth Inhibition Assay | Inhibition of human HCC1419 cell growth in a cell viability assay, IC50 = 4.26266 μM. | SANGER | |||
| OCUB-M | Growth Inhibition Assay | Inhibition of human OCUB-M cell growth in a cell viability assay, IC50 = 4.28414 μM. | SANGER | |||
| BxPC-3 | Growth Inhibition Assay | Inhibition of human BxPC-3 cell growth in a cell viability assay, IC50 = 4.29914 μM. | SANGER | |||
| HT-1197 | Growth Inhibition Assay | Inhibition of human HT-1197 cell growth in a cell viability assay, IC50 = 4.31327 μM. | SANGER | |||
| NCI-H526 | Growth Inhibition Assay | Inhibition of human NCI-H526 cell growth in a cell viability assay, IC50 = 4.33328 μM. | SANGER | |||
| KS-1 | Growth Inhibition Assay | Inhibition of human KS-1 cell growth in a cell viability assay, IC50 = 4.34468 μM. | SANGER | |||
| NCI-H650 | Growth Inhibition Assay | Inhibition of human NCI-H650 cell growth in a cell viability assay, IC50 = 4.35377 μM. | SANGER | |||
| KU-19-19 | Growth Inhibition Assay | Inhibition of human KU-19-19 cell growth in a cell viability assay, IC50 = 4.42891 μM. | SANGER | |||
| UACC-257 | Growth Inhibition Assay | Inhibition of human UACC-257 cell growth in a cell viability assay, IC50 = 4.46437 μM. | SANGER | |||
| TGBC1TKB | Growth Inhibition Assay | Inhibition of human TGBC1TKB cell growth in a cell viability assay, IC50 = 4.5212 μM. | SANGER | |||
| LB831-BLC | Growth Inhibition Assay | Inhibition of human LB831-BLC cell growth in a cell viability assay, IC50 = 4.58569 μM. | SANGER | |||
| SW1783 | Growth Inhibition Assay | Inhibition of human SW1783 cell growth in a cell viability assay, IC50 = 4.61383 μM. | SANGER | |||
| NCI-H2347 | Growth Inhibition Assay | Inhibition of human NCI-H2347 cell growth in a cell viability assay, IC50 = 4.63698 μM. | SANGER | |||
| RCC10RGB | Growth Inhibition Assay | Inhibition of human RCC10RGB cell growth in a cell viability assay, IC50 = 4.68428 μM. | SANGER | |||
| A101D | Growth Inhibition Assay | Inhibition of human A101D cell growth in a cell viability assay, IC50 = 4.76358 μM. | SANGER | |||
| NCI-H2087 | Growth Inhibition Assay | Inhibition of human NCI-H2087 cell growth in a cell viability assay, IC50 = 4.80595 μM. | SANGER | |||
| NCI-H1651 | Growth Inhibition Assay | Inhibition of human NCI-H1651 cell growth in a cell viability assay, IC50 = 4.98474 μM. | SANGER | |||
| SK-N-FI | Growth Inhibition Assay | Inhibition of human SK-N-FI cell growth in a cell viability assay, IC50 = 5.06408 μM. | SANGER | |||
| SW1116 | Growth Inhibition Assay | Inhibition of human SW1116 cell growth in a cell viability assay, IC50 = 5.12 μM. | SANGER | |||
| C3A | Growth Inhibition Assay | Inhibition of human C3A cell growth in a cell viability assay, IC50 = 5.23422 μM. | SANGER | |||
| SNU-449 | Growth Inhibition Assay | Inhibition of human SNU-449 cell growth in a cell viability assay, IC50 = 5.30865 μM. | SANGER | |||
| NCI-H630 | Growth Inhibition Assay | Inhibition of human NCI-H630 cell growth in a cell viability assay, IC50 = 5.34812 μM. | SANGER | |||
| DK-MG | Growth Inhibition Assay | Inhibition of human DK-MG cell growth in a cell viability assay, IC50 = 5.39082 μM. | SANGER | |||
| YAPC | Growth Inhibition Assay | Inhibition of human YAPC cell growth in a cell viability assay, IC50 = 5.44769 μM. | SANGER | |||
| BEN | Growth Inhibition Assay | Inhibition of human BEN cell growth in a cell viability assay, IC50 = 5.44777 μM. | SANGER | |||
| Calu-3 | Growth Inhibition Assay | Inhibition of human Calu-3 cell growth in a cell viability assay, IC50 = 5.45736 μM. | SANGER | |||
| 8305C | Growth Inhibition Assay | Inhibition of human 8305C cell growth in a cell viability assay, IC50 = 5.46669 μM. | SANGER | |||
| ESS-1 | Growth Inhibition Assay | Inhibition of human ESS-1 cell growth in a cell viability assay, IC50 = 5.47851 μM. | SANGER | |||
| ME-180 | Growth Inhibition Assay | Inhibition of human ME-180 cell growth in a cell viability assay, IC50 = 5.48535 μM. | SANGER | |||
| D-502MG | Growth Inhibition Assay | Inhibition of human D-502MG cell growth in a cell viability assay, IC50 = 5.49075 μM. | SANGER | |||
| CP50-MEL-B | Growth Inhibition Assay | Inhibition of human CP50-MEL-B cell growth in a cell viability assay, IC50 = 5.59584 μM. | SANGER | |||
| OAW-28 | Growth Inhibition Assay | Inhibition of human OAW-28 cell growth in a cell viability assay, IC50 = 5.66484 μM. | SANGER | |||
| SCC-25 | Growth Inhibition Assay | Inhibition of human SCC-25 cell growth in a cell viability assay, IC50 = 5.66836 μM. | SANGER | |||
| NCI-H810 | Growth Inhibition Assay | Inhibition of human NCI-H810 cell growth in a cell viability assay, IC50 = 5.67785 μM. | SANGER | |||
| no-11 | Growth Inhibition Assay | Inhibition of human no-11 cell growth in a cell viability assay, IC50 = 5.81096 μM. | SANGER | |||
| OVCAR-4 | Growth Inhibition Assay | Inhibition of human OVCAR-4 cell growth in a cell viability assay, IC50 = 6.07487 μM. | SANGER | |||
| A704 | Growth Inhibition Assay | Inhibition of human A704 cell growth in a cell viability assay, IC50 = 6.13363 μM. | SANGER | |||
| GB-1 | Growth Inhibition Assay | Inhibition of human GB-1 cell growth in a cell viability assay, IC50 = 6.14107 μM. | SANGER | |||
| NCI-H358 | Growth Inhibition Assay | Inhibition of human NCI-H358 cell growth in a cell viability assay, IC50 = 6.19117 μM. | SANGER | |||
| A431 | Growth Inhibition Assay | Inhibition of human A431 cell growth in a cell viability assay, IC50 = 6.26477 μM. | SANGER | |||
| SK-MEL-24 | Growth Inhibition Assay | Inhibition of human SK-MEL-24 cell growth in a cell viability assay, IC50 = 6.2749 μM. | SANGER | |||
| NB17 | Growth Inhibition Assay | Inhibition of human NB17 cell growth in a cell viability assay, IC50 = 6.44011 μM. | SANGER | |||
| BB49-HNC | Growth Inhibition Assay | Inhibition of human BB49-HNC cell growth in a cell viability assay, IC50 = 6.49529 μM. | SANGER | |||
| NCI-H1573 | Growth Inhibition Assay | Inhibition of human NCI-H1573 cell growth in a cell viability assay, IC50 = 6.70803 μM. | SANGER | |||
| GAK | Growth Inhibition Assay | Inhibition of human GAK cell growth in a cell viability assay, IC50 = 6.8404 μM. | SANGER | |||
| CAL-120 | Growth Inhibition Assay | Inhibition of human CAL-120 cell growth in a cell viability assay, IC50 = 7.07643 μM. | SANGER | |||
| PANC-08-13 | Growth Inhibition Assay | Inhibition of human PANC-08-13 cell growth in a cell viability assay, IC50 = 7.78108 μM. | SANGER | |||
| SW1463 | Growth Inhibition Assay | Inhibition of human SW1463 cell growth in a cell viability assay, IC50 = 8.01873 μM. | SANGER | |||
| SW872 | Growth Inhibition Assay | Inhibition of human SW872 cell growth in a cell viability assay, IC50 = 8.07734 μM. | SANGER | |||
| HCT-15 | Growth Inhibition Assay | Inhibition of human HCT-15 cell growth in a cell viability assay, IC50 = 8.12311 μM. | SANGER | |||
| SH-4 | Growth Inhibition Assay | Inhibition of human SH-4 cell growth in a cell viability assay, IC50 = 8.14247 μM. | SANGER | |||
| TE-6 | Growth Inhibition Assay | Inhibition of human TE-6 cell growth in a cell viability assay, IC50 = 8.24962 μM. | SANGER | |||
| Capan-2 | Growth Inhibition Assay | Inhibition of human Capan-2 cell growth in a cell viability assay, IC50 = 8.36036 μM. | SANGER | |||
| RO82-W-1 | Growth Inhibition Assay | Inhibition of human RO82-W-1 cell growth in a cell viability assay, IC50 = 8.4861 μM. | SANGER | |||
| SW948 | Growth Inhibition Assay | Inhibition of human SW948 cell growth in a cell viability assay, IC50 = 8.56121 μM. | SANGER | |||
| NB12 | Growth Inhibition Assay | Inhibition of human NB12 cell growth in a cell viability assay, IC50 = 8.6577 μM. | SANGER | |||
| NCI-H2342 | Growth Inhibition Assay | Inhibition of human NCI-H2342 cell growth in a cell viability assay, IC50 = 8.67749 μM. | SANGER | |||
| PFSK-1 | Growth Inhibition Assay | Inhibition of human PFSK-1 cell growth in a cell viability assay, IC50 = 8.73766 μM. | SANGER | |||
| HPAF-II | Growth Inhibition Assay | Inhibition of human HPAF-II cell growth in a cell viability assay, IC50 = 8.77154 μM. | SANGER | |||
| MDA-MB-231 | Growth Inhibition Assay | Inhibition of human MDA-MB-231 cell growth in a cell viability assay, IC50 = 8.82383 μM. | SANGER | |||
| KNS-81-FD | Growth Inhibition Assay | Inhibition of human KNS-81-FD cell growth in a cell viability assay, IC50 = 9.19714 μM. | SANGER | |||
| NCI-H1563 | Growth Inhibition Assay | Inhibition of human NCI-H1563 cell growth in a cell viability assay, IC50 = 9.21672 μM. | SANGER | |||
| MEL-HO | Growth Inhibition Assay | Inhibition of human MEL-HO cell growth in a cell viability assay, IC50 = 9.31744 μM. | SANGER | |||
| NCI-H2126 | Growth Inhibition Assay | Inhibition of human NCI-H2126 cell growth in a cell viability assay, IC50 = 9.32815 μM. | SANGER | |||
| SNB75 | Growth Inhibition Assay | Inhibition of human SNB75 cell growth in a cell viability assay, IC50 = 9.36079 μM. | SANGER | |||
| NCI-H1693 | Growth Inhibition Assay | Inhibition of human NCI-H1693 cell growth in a cell viability assay, IC50 = 9.57931 μM. | SANGER | |||
| NCI-H460 | Growth Inhibition Assay | Inhibition of human NCI-H460 cell growth in a cell viability assay, IC50 = 9.6401 μM. | SANGER | |||
| COLO-824 | Growth Inhibition Assay | Inhibition of human COLO-824 cell growth in a cell viability assay, IC50 = 9.83259 μM. | SANGER | |||
| CAL-12T | Growth Inhibition Assay | Inhibition of human CAL-12T cell growth in a cell viability assay, IC50 = 10.1844 μM. | SANGER | |||
| KM12 | Growth Inhibition Assay | Inhibition of human KM12 cell growth in a cell viability assay, IC50 = 10.2778 μM. | SANGER | |||
| IA-LM | Growth Inhibition Assay | Inhibition of human IA-LM cell growth in a cell viability assay, IC50 = 10.4447 μM. | SANGER | |||
| SK-MEL-3 | Growth Inhibition Assay | Inhibition of human SK-MEL-3 cell growth in a cell viability assay, IC50 = 11.1627 μM. | SANGER | |||
| LB771-HNC | Growth Inhibition Assay | Inhibition of human LB771-HNC cell growth in a cell viability assay, IC50 = 11.1986 μM. | SANGER | |||
| NCI-H2291 | Growth Inhibition Assay | Inhibition of human NCI-H2291 cell growth in a cell viability assay, IC50 = 11.3553 μM. | SANGER | |||
| NCI-H1092 | Growth Inhibition Assay | Inhibition of human NCI-H1092 cell growth in a cell viability assay, IC50 = 11.7291 μM. | SANGER | |||
| MMAC-SF | Growth Inhibition Assay | Inhibition of human MMAC-SF cell growth in a cell viability assay, IC50 = 11.9527 μM. | SANGER | |||
| CP66-MEL | Growth Inhibition Assay | Inhibition of human CP66-MEL cell growth in a cell viability assay, IC50 = 11.9806 μM. | SANGER | |||
| LAN-6 | Growth Inhibition Assay | Inhibition of human LAN-6 cell growth in a cell viability assay, IC50 = 12.4743 μM. | SANGER | |||
| COLO-684 | Growth Inhibition Assay | Inhibition of human COLO-684 cell growth in a cell viability assay, IC50 = 12.9893 μM. | SANGER | |||
| NCI-H720 | Growth Inhibition Assay | Inhibition of human NCI-H720 cell growth in a cell viability assay, IC50 = 13.1893 μM. | SANGER | |||
| C32 | Growth Inhibition Assay | Inhibition of human C32 cell growth in a cell viability assay, IC50 = 13.2745 μM. | SANGER | |||
| KLE | Growth Inhibition Assay | Inhibition of human KLE cell growth in a cell viability assay, IC50 = 13.3925 μM. | SANGER | |||
| NCI-H1648 | Growth Inhibition Assay | Inhibition of human NCI-H1648 cell growth in a cell viability assay, IC50 = 13.6829 μM. | SANGER | |||
| SBC-1 | Growth Inhibition Assay | Inhibition of human SBC-1 cell growth in a cell viability assay, IC50 = 14.2024 μM. | SANGER | |||
| RVH-421 | Growth Inhibition Assay | Inhibition of human RVH-421 cell growth in a cell viability assay, IC50 = 14.3302 μM. | SANGER | |||
| LB373-MEL-D | Growth Inhibition Assay | Inhibition of human LB373-MEL-D cell growth in a cell viability assay, IC50 = 14.38 μM. | SANGER | |||
| NCI-H441 | Growth Inhibition Assay | Inhibition of human NCI-H441 cell growth in a cell viability assay, IC50 = 14.7391 μM. | SANGER | |||
| ECC10 | Growth Inhibition Assay | Inhibition of human ECC10 cell growth in a cell viability assay, IC50 = 15.1731 μM. | SANGER | |||
| SNU-C2B | Growth Inhibition Assay | Inhibition of human SNU-C2B cell growth in a cell viability assay, IC50 = 15.4643 μM. | SANGER | |||
| U-266 | Growth Inhibition Assay | Inhibition of human U-266 cell growth in a cell viability assay, IC50 = 15.5375 μM. | SANGER | |||
| HOP-62 | Growth Inhibition Assay | Inhibition of human HOP-62 cell growth in a cell viability assay, IC50 = 16.6013 μM. | SANGER | |||
| CaR-1 | Growth Inhibition Assay | Inhibition of human CaR-1 cell growth in a cell viability assay, IC50 = 16.6623 μM. | SANGER | |||
| LN-405 | Growth Inhibition Assay | Inhibition of human LN-405 cell growth in a cell viability assay, IC50 = 17.1486 μM. | SANGER | |||
| DJM-1 | Growth Inhibition Assay | Inhibition of human DJM-1 cell growth in a cell viability assay, IC50 = 17.17 μM. | SANGER | |||
| KY821 | Growth Inhibition Assay | Inhibition of human KY821 cell growth in a cell viability assay, IC50 = 17.2008 μM. | SANGER | |||
| RMG-I | Growth Inhibition Assay | Inhibition of human RMG-I cell growth in a cell viability assay, IC50 = 18.5568 μM. | SANGER | |||
| SBC-5 | Growth Inhibition Assay | Inhibition of human SBC-5 cell growth in a cell viability assay, IC50 = 19.5065 μM. | SANGER | |||
| NCI-H1838 | Growth Inhibition Assay | Inhibition of human NCI-H1838 cell growth in a cell viability assay, IC50 = 19.9055 μM. | SANGER | |||
| NB6 | Growth Inhibition Assay | Inhibition of human NB6 cell growth in a cell viability assay, IC50 = 20.2853 μM. | SANGER | |||
| OVCAR-3 | Growth Inhibition Assay | Inhibition of human OVCAR-3 cell growth in a cell viability assay, IC50 = 20.3154 μM. | SANGER | |||
| MKN7 | Growth Inhibition Assay | Inhibition of human MKN7 cell growth in a cell viability assay, IC50 = 20.3314 μM. | SANGER | |||
| HCC1569 | Growth Inhibition Assay | Inhibition of human HCC1569 cell growth in a cell viability assay, IC50 = 21.9995 μM. | SANGER | |||
| OE19 | Growth Inhibition Assay | Inhibition of human OE19 cell growth in a cell viability assay, IC50 = 22.4401 μM. | SANGER | |||
| LU-139 | Growth Inhibition Assay | Inhibition of human LU-139 cell growth in a cell viability assay, IC50 = 22.4402 μM. | SANGER | |||
| NB13 | Growth Inhibition Assay | Inhibition of human NB13 cell growth in a cell viability assay, IC50 = 22.504 μM. | SANGER | |||
| SW1417 | Growth Inhibition Assay | Inhibition of human SW1417 cell growth in a cell viability assay, IC50 = 22.5851 μM. | SANGER | |||
| NCI-H1975 | Growth Inhibition Assay | Inhibition of human NCI-H1975 cell growth in a cell viability assay, IC50 = 23.8807 μM. | SANGER | |||
| LCLC-103H | Growth Inhibition Assay | Inhibition of human LCLC-103H cell growth in a cell viability assay, IC50 = 24.5205 μM. | SANGER | |||
| HuH-7 | Growth Inhibition Assay | Inhibition of human HuH-7 cell growth in a cell viability assay, IC50 = 24.6147 μM. | SANGER | |||
| LU-135 | Growth Inhibition Assay | Inhibition of human LU-135 cell growth in a cell viability assay, IC50 = 26.5933 μM. | SANGER | |||
| NCI-H1793 | Growth Inhibition Assay | Inhibition of human NCI-H1793 cell growth in a cell viability assay, IC50 = 26.6093 μM. | SANGER | |||
| SK-MEL-30 | Growth Inhibition Assay | Inhibition of human SK-MEL-30 cell growth in a cell viability assay, IC50 = 27.3999 μM. | SANGER | |||
| HCC1937 | Growth Inhibition Assay | Inhibition of human HCC1937 cell growth in a cell viability assay, IC50 = 27.5398 μM. | SANGER | |||
| 639-V | Growth Inhibition Assay | Inhibition of human 639-V cell growth in a cell viability assay, IC50 = 27.5914 μM. | SANGER | |||
| HCC1187 | Growth Inhibition Assay | Inhibition of human HCC1187 cell growth in a cell viability assay, IC50 = 28.6541 μM. | SANGER | |||
| no-10 | Growth Inhibition Assay | Inhibition of human no-10 cell growth in a cell viability assay, IC50 = 29.8844 μM. | SANGER | |||
| HCC1954 | Growth Inhibition Assay | Inhibition of human HCC1954 cell growth in a cell viability assay, IC50 = 30.3937 μM. | SANGER | |||
| SW837 | Growth Inhibition Assay | Inhibition of human SW837 cell growth in a cell viability assay, IC50 = 30.8538 μM. | SANGER | |||
| CCRF-CEM | Growth Inhibition Assay | Inhibition of human CCRF-CEM cell growth in a cell viability assay, IC50 = 32.1211 μM. | SANGER | |||
| CAMA-1 | Growth Inhibition Assay | Inhibition of human CAMA-1 cell growth in a cell viability assay, IC50 = 32.163 μM. | SANGER | |||
| T47D | Growth Inhibition Assay | Inhibition of human T47D cell growth in a cell viability assay, IC50 = 32.1884 μM. | SANGER | |||
| KNS-42 | Growth Inhibition Assay | Inhibition of human KNS-42 cell growth in a cell viability assay, IC50 = 32.2443 μM. | SANGER | |||
| COLO-320-HSR | Growth Inhibition Assay | Inhibition of human COLO-320-HSR cell growth in a cell viability assay, IC50 = 32.4293 μM. | SANGER | |||
| UACC-893 | Growth Inhibition Assay | Inhibition of human UACC-893 cell growth in a cell viability assay, IC50 = 32.6355 μM. | SANGER | |||
| KALS-1 | Growth Inhibition Assay | Inhibition of human KALS-1 cell growth in a cell viability assay, IC50 = 33.3928 μM. | SANGER | |||
| ChaGo-K-1 | Growth Inhibition Assay | Inhibition of human ChaGo-K-1 cell growth in a cell viability assay, IC50 = 33.7009 μM. | SANGER | |||
| MFM-223 | Growth Inhibition Assay | Inhibition of human MFM-223 cell growth in a cell viability assay, IC50 = 34.0243 μM. | SANGER | |||
| C8166 | Growth Inhibition Assay | Inhibition of human C8166 cell growth in a cell viability assay, IC50 = 40.3675 μM. | SANGER | |||
| NCI-H2029 | Growth Inhibition Assay | Inhibition of human NCI-H2029 cell growth in a cell viability assay, IC50 = 40.9728 μM. | SANGER | |||
| DMS-79 | Growth Inhibition Assay | Inhibition of human DMS-79 cell growth in a cell viability assay, IC50 = 41.1583 μM. | SANGER | |||
| T84 | Growth Inhibition Assay | Inhibition of human T84 cell growth in a cell viability assay, IC50 = 41.246 μM. | SANGER | |||
| NCI-H520 | Growth Inhibition Assay | Inhibition of human NCI-H520 cell growth in a cell viability assay, IC50 = 43.4761 μM. | SANGER | |||
| COLO-680N | Growth Inhibition Assay | Inhibition of human COLO-680N cell growth in a cell viability assay, IC50 = 45.2396 μM. | SANGER | |||
| RH-18 | Growth Inhibition Assay | Inhibition of human RH-18 cell growth in a cell viability assay, IC50 = 46.3786 μM. | SANGER | |||
| BT-474 | Growth Inhibition Assay | Inhibition of human BT-474 cell growth in a cell viability assay, IC50 = 49.4631 μM. | SANGER | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 49 mg/mL
(201.46 mM)
Water : 49 mg/mL Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 243.22 | Formula | C9H13N3O5 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 147-94-4 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Cytarabin,Ara-C,Arabinofuranosyl Cytidine,Cytosine β-D-arabinofuranoside,Cytosine arabinoside,NSC 63878,NSC 287459,U-19920A | Smiles | C1=CN(C(=O)N=C1N)C2C(C(C(O2)CO)O)O | ||
Mechanism of Action
| Features |
The 1st of a series of cancer drugs that alters the sugar component of nucleosides.
|
|---|---|
| Targets/IC50/Ki |
DNA synthesis
(CCRF-CEM cells) 16 nM
|
| In vitro |
Cytarabine (AraC) is phosphorylated into a triphosphate form (Ara-CTP) involving deoxycytidine kinase (dCK), which competes with dCTP for incorporation into DNA, and then blocks DNA synthesis by inhibiting the function of DNA and RNA polymerases. This compound displays a higher growth inhibitory activity towards wild-type CCRF-CEM cells compared to other acute myelogenous leukemia (AML) cells with IC50 of 16 nM. Increasing concentrations of this chemical (IC50 of 0.69 μM) results in decreased metabolic activity of sensitive rat leukemic cell line RO/1, and the cell toxity can be highly enhanced by transfection with human wt dCK (IC50 of 0.037 μM) but not the inactive, alternatively spliced dCK forms. It apparently induces apoptosis of rat sympathetic neurons at 10 μM, of which 100 μM shows the highest toxicity and kills over 80% of the neurons by 84 hours, involving the release of mitochondrial cytochrome-c and the activation of caspase-3, and the toxicity can be attenuated by p53 knockdown and delayed by bax deletion. |
| Kinase Assay |
In Vitro Growth Inhibition Assay
|
|
Stock solution of Cytarabine is prepared in absolute ethanol, and serial dilutions of this compound are prepared. CCRF-CEM cells are suspended in RPMI medium supplemented with 10% FBS, 0.1% gentamicin, and 1% sodium pyruvate. The cells are suspended in their respective media to give 10 mL volumes of cell suspension at a final density of 3-6 × 104 cells/mL. Appropriate volumes of this chemical solution are transferred to the cell suspensions, and incubation is continued for 72 hours. The cells are spun down and resuspended in fresh this compound -free medium, and final cell counts are determined. The data are analyzed by sigmoidal curve fitting of the cell count versus this chemical concentration, and the results are expressed as the IC50 (this compound concentration that inhibits cell growth to 50% of the control value).
|
|
| In vivo |
Cytarabine is highly effective against acute leukaemias, which causes the characteristic G1/S blockage and synchronization, and increases the survival time for leukaemic Brown Norway rats in a weak dose-related fashion indicating that the use of higher dosages of this compound does not contribute to its antileukaemic effectiveness in man. This chemical also causes placental growth retardation and increases placental trophoblastic cells apoptosis in the placental labyrinth zone of the pregnant Slc:Wistar rats, which increases from 3 hour after the treatment and peaks at 6 hour before returning to control levels at 48 hour, with remarkably enhanced p53 protein, p53 trancriptional target genes such as p21, cyclinG1 and fas and caspase-3 activity. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p-mTOR / mTOR / p-S6K / S6K / p-AMPK / AMPK / p-AKT / AKT / p-ERK / ERK |
|
24714637 |
| Growth inhibition assay | Cell viability |
|
24714637 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05697510 | Recruiting | Acute Myeloid Leukemia (AML) |
Nantes University Hospital |
March 16 2023 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































