EGFR is a trans-membrane receptor belonging to the erbB/HER-family of RTK. EGFR exists on the cell surface and can be activated by EGF and TGF-alpha. Many important signaling cascades, like MAPK, Akt, and JNK pathways, could be the downstream of EGFR. [show the full text]
EGFR Inhibitors
Other Protein Tyrosine Kinase Related Inhibitors
Isoform-selective Products
| Cat.No. | Product Name | Information | Product Use Citations | Product Validations |
|---|---|---|---|---|
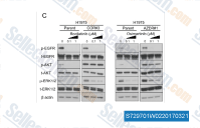
| S7297 | AZD9291 (Osimertinib) | Osimertinib (AZD9291) is an oral, irreversible, and mutant-selective EGFR inhibitor with IC50 of 12.92, 11.44 and 493.8 nM for Exon 19 deletion EGFR, L858R/T790M EGFR, and WT EGFR in LoVo cells, respectively. Phase 3. |
|
|
| S1011 | Afatinib (BIBW2992) | Afatinib inhibits EGFR/ErbB irreversibly in vitro with IC50 of 0.5, 0.4, 10, 14, 1 nM for EGFRwt, EGFR L858R , EGFR L858R/T790M ErbB2 (HER2) and ErbB4 (HER4), respectively. This compound induces autophagy. |
|
|
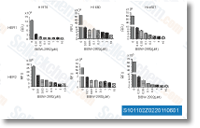
| S8724 | Lazertinib (YH25448) | Lazertinib (YH25448,GNS-1480) is a potent, highly mutant-selective and irreversible EGFR-TKI with IC50 values of 1.7 nM, 2 nM, 5 nM, 20.6 nM and 76 nM for Del19/T790M, L858R/T790M, Del19, L85R and Wild type EGFR respectively, showing much higher IC50 values aganist ErbB2 and ErbB4. |
|
|
| S1025 | Gefitinib (ZD1839) | Gefitinib is an EGFR inhibitor for Tyr1173, Tyr992, Tyr1173 and Tyr992 in the NR6wtEGFR and NR6W cells with IC50 of 37 nM, 37nM, 26 nM and 57 nM, respectively. This compound promotes autophagy and apoptosis of lung cancer cells via blockade of the PI3K/AKT/mTOR pathway. |
|

|
| E4884 | Icotinib Hydrochloride | Icotinib hydrochloride (BPI-2009H) is a highly potent and selective epidermal growth factor receptor tyrosine kinase inhibitor (EGFR-TKI) with an IC50 of 5 nM, utilized in the treatment of non-small cell lung cancer (NSCLC). | ||
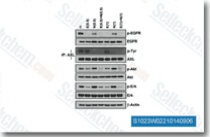
| S7786 | Erlotinib (CP-358774) | Erlotinib is an EGFR inhibitor with IC50 of 2 nM, >1000-fold more sensitive for EGFR than human c-Src or v-Abl. Erlotinib induces autophagy. |
|
|
| E5906New | Sunvozertinib | Sunvozertinib (DZD9008) is an oral, potent, and selective inhibitor of ErbBs (EGFR or Her2), particularly mutant forms of ErbBs. It also inhibits EGFR exon 20 NPH insertion, EGFR exon 20 ASV insertion, EGFR L858R and T790M mutations, Her2 exon 20 YVMA, and EGFR WT A431, with IC50 values of 20.4 nM, 20.4 nM, 1.1 nM, 7.5 nM, and 80.4 nM, respectively. |
|
|
| S1023 | Erlotinib (CP-358774) Hydrochloride | Erlotinib HCl is an EGFR inhibitor with IC50 of 2 nM in cell-free assays, >1000-fold more sensitive for EGFR than human c-Src or v-Abl. |
|

|
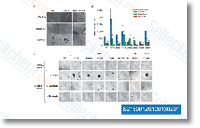
| S2150 | Neratinib (HKI-272) | Neratinib is a highly selective HER2 and EGFR inhibitor with IC50 of 59 nM and 92 nM in cell-free assays; weakly inhibits KDR and Src, no significant inhibition to Akt, CDK1/2/4, IKK-2, MK-2, PDK1, c-Raf and c-Met. Phase 3. |
|
|
| S8362 | Tucatinib (Irbinitinib, ONT-380, Arry-380) | Tucatinib (Irbinitinib, ONT-380, ARRY-380) is an oral, potent, selective, reversible and ATP-competitive small-molecule inhibitor of ErbB-2 (also called HER2) with IC50s of 8 nM and 7 nM for ErbB-2 and p95 HER2, respectively in cell-based assays, showing ~500-fold selective for HER2 vs EGFR. It has potential antineoplastic activity. |
|
|
Signaling Pathway Map
Epidermal growth factor receptor (EGFR) was initially discovered in 1962 following the identification of the ligand EGF. Following this, the role of EGFR in protein phosphorylation and tumorigenesis has been established, and the EGF-EGFR signaling axis has consequently been the focus of research in oncology and developmental biology. Situated in the plasma membrane, EGFR has been an attractive target for anticancer therapy as it becomes activated upon ligand binding, recruiting a number of downstream molecules that leads to the activation of major pathways implicated in tumor growth, progression, and survival.[1][2]
EGFR belongs to the ErbB/HER receptor tyrosine kinase family that encompasses HER2 (ErbB2), HER3 (ErbB3), and HER4 (ErbB4).ii Each constituent of the EGFR family shares a basic domain structure including an N-terminal extracellular domain (subdivided in to domains I through IV), a transmembrane domain, an intracellular kinase domain, and a cytoplasmic C-terminal tail containing several phosphorylation sites that serve as signal transduction modules. The binding of one of several ligands to the extracellular ligand-binding domain induces receptor homo-dimerization or hetero-dimerization and results in kinase activation. Among the EGFR family, it should be noted that ErbB2 is an orphan receptor, characterized by ligand-induced hetero-dimerization with any other family member for activation. Research on ErbB3 initially did not show any intrinsic kinase activity, although recent research findings suggest otherwise.[3]
In the initial activation response of HER family constituents and similar receptor tyrosine kinases are downregulation events, involving ligand-stimulated endocytosis of occupied receptor accompanied by receptor ubiquitination and followed by lysosomal degradation of both ligands and receptors. Another possibility is that receptor tyrosine kinases are recycled from endosomes to the plasma membrane. Interestingly, HER2 and HER3 are internalized and targeted to lysosomes less efficiently than EGFR, and increased expression of HER2 or HER3 can have a dominant-negative effect on EGFR downregulation and degradation. Whether degradation and/or recycling events occur will affect the number of receptors and thus downstream signalling, profoundly changing the biological response.[2]
Ligands of EGFR include EGF, transforming growth factor-α, and heparin-binding EGF-like growth factor. Once bound to its ligand, EGFR recruits, phosphorylates and activates all of the following downstream signalling cascades: MAPK, PLC-ϒ/PKC, Ras-Raf-Mek, PI3K-Akt-mTOR/PKB and JAK2-STAT3.[1][2] EGFR can also mediate cellular processes through the physical interaction with other proteins in the absence of kinase activity or ligand activation.[1]
Several EGFR-targeting small molecule kinase inhibitors and therapeutic antibodies have been approved by the FDA to treat patients with breast cancer, colorectal cancer, non-small cell lung cancer (NSCLC), head and neck-related squamous cell carcinoma and pancreatic cancer. Despite the role that the EGFR signalling pathway plays in several downstream events that lead to tumorigenesis, currently approved EGFR-targeted therapies show only modest effect on most cancer types.[1]
Of the classes of compounds that are in clinical trials and/or are approved for clinical practice use, there are two molecular approaches to target EGFR: (1) monoclonal antibodies, directed against the external ligand-binding site of the receptor (i.e. cetuximab and panitumumab), and (2) small molecule tyrosine kinase inhibitors, directed against intracellular tyrosine kinase domain (i.e. gefitinib, erlotinib, and lapatinib). In either case, both classes of compounds target EGFR homodimers and heterodimers. Evidence of this includes the ability of erlotinib to target both EGFR and HER3. Interestingly, the impact of monoclonal antibodies targeting EGFR or HER2 is observed to impact VEGFR expression. EGFR affects VEGFR activity through the MAPK and PI3K signalling pathways and at least three different transcription factors, STAT3, Sp1 and hypoxia-inducible factors (HIFs).ii A close relative of EGFR is EGFRvIII, found to be localized on the cell-surface where it activates several signalling molecules; overexpression of EGFRvIII has been implicated in malignant gliomas. In tumorigenesis, both EGFR and EGFRvIII are observed to be involved in malignant phenotypes in human cancers.[1]
ErbB proteins are highlighted in oncology research for their ability to drive proliferation, survival, and differentiation. Their overexpression in a variety of human cancers qualifies them as an ideal target for further investigation.[3]





































