research use only
Sertaconazole nitrate Fungal inhibitor
Cat.No.S3161
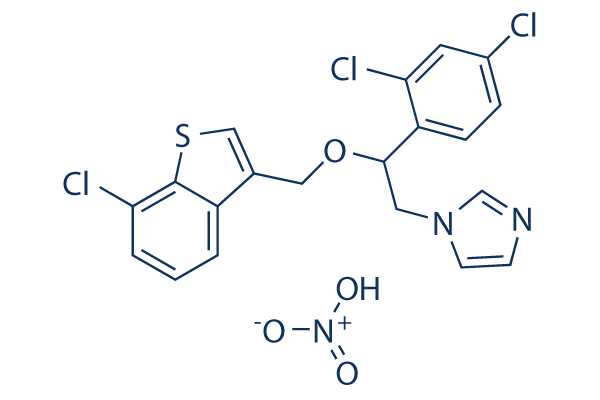
Chemical Structure
Molecular Weight: 500.78
Quality Control
| Related Targets | Integrase Bacterial Antibiotics Anti-infection Antiviral COVID-19 Parasite Reverse Transcriptase HIV HCV Protease |
|---|---|
| Other Fungal Inhibitors | Cycloheximide Tolnaftate Amorolfine HCl Isavuconazole Juglone Bifonazole Manogepix (E1210) Thimerosal Pseudolaric Acid B Butylparaben |
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(199.68 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 500.78 | Formula | C20H15Cl3N2OS.HNO3 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 99592-39-9 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | FI-7045,FI7056 | Smiles | C1=CC2=C(C(=C1)Cl)SC=C2COC(CN3C=CN=C3)C4=C(C=C(C=C4)Cl)Cl.[N+](=O)(O)[O-] | ||
Mechanism of Action
| In vitro |
Sertaconazole nitrate reduces the release of cytokines from activated lymphocytes and mitigated inflammation in animal models of irritant contact dermatitis and neurogenic inflammation in a dose-dependent fashion. This compound is found to inhibit the proliferation of stimulated human lymphocytes with IC50 of 4 μg/mL. It inhibits ergosterol synthesis by blockade of the P450-dependent enzyme pathway that catalyzes the methylation of lanosterol to ergosterol, thus inhibits fungal cell growth. This chemical binds directly to nonsterol lipids in the membrane, which interferes with the regulation of the permeability of fungal cell membranes, thus induces fungal cell death. This compound (< 100 μg/mL) activates p38 MAP kinase and Hsp27 in keratinocytes. It induces (< 2 μg/mL) COX-2 protein expression and PGE2 production in keratinocytes, which dependent on p38 activation. PGE2 is involved in this compound (1 μg/mL)-mediated suppression of cytokine production in keratinocytes and PBMCs. |
|---|---|
| In vivo |
The mean ear weight of Tetradecanoyl phorbol acetate (TPA)-challenged murine treated with sertaconazole nitrate (1%) is 7.23 mg compared with 14.7 mg for controls, indicating a statistically significant reduction in irritant dermatitis. This compound elicits a significant reduction in Resiniferatoxin-induced ear edema when compared with controls in CD-1 mice. Topical treatment with this chemical significantly inhibits contact hypersensitivity and decreases the content of the pro-inflammatory cytokines TNFα, IL-2, and IFNγ in oxazolone exposed murine skin. Clinical trials with this compound cream 2% show efficacy in the treatment of superficial cutaneous fungal infections. It reduces inflammation via inducing PGE2 production and the COX-2 inhibitor blocks it from exerting its anti-inflammatory effects in a mouse model of TPA-induced ear edema. The dermal administration of this chemical to rats, in which edema is induced by application of croton oil in the external ear, induced a 39.8% reduction of edema. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT00856596 | Unknown status | Tinea Pedis|Athlete''s Foot|Foot Fungus|Ringworm |
Jamaica Hospital Medical Center|Johnson & Johnson |
March 2009 | Phase 3 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































