research use only
Doravirine Reverse Transcriptase inhibitor
Cat.No.S6492
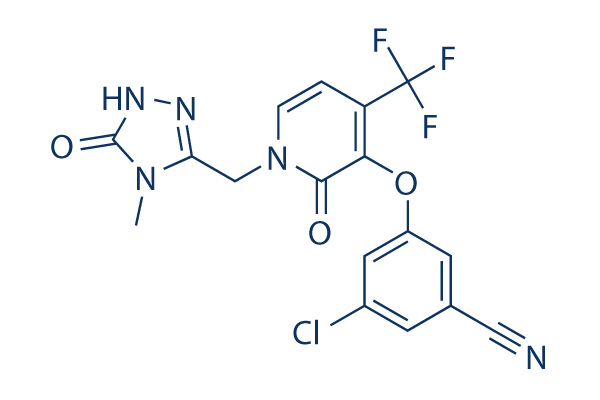
Chemical Structure
Molecular Weight: 425.75
Quality Control
| Related Targets | Integrase Bacterial Antibiotics Anti-infection Fungal Antiviral COVID-19 Parasite HIV HCV Protease |
|---|---|
| Other Reverse Transcriptase Inhibitors | Dapivirine (TMC120) Fangchinoline 3'-Fluoro-3'-deoxythymidine (Alovudine) Salicylanilide Bifendate Ulonivirine Lersivirine (UK-453061) 4-Chloro-2-(trifluoroacetyl)aniline hydrochloride |
Solubility
|
In vitro |
DMSO
: 85 mg/mL
(199.64 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 425.75 | Formula | C17H11C1F3N5O3 |
Storage (From the date of receipt) | 3 years -20°C powder |
|---|---|---|---|---|---|
| CAS No. | 1338225-97-0 | -- | Storage of Stock Solutions |
|
|
| Synonyms | MK-1439 | Smiles | CN1C(=NNC1=O)CN2C=CC(=C(C2=O)OC3=CC(=CC(=C3)C#N)Cl)C(F)(F)F | ||
Mechanism of Action
| Targets/IC50/Ki |
K103N HIV-1 RT
9.7 nM
Y181C HIV-1 RT
9.7 nM
WT HIV-1 RT
12 nM
|
|---|---|
| In vitro |
MK-1439 exhibits greater than 10,000-fold selectivity with respect to the cellular DNA polymerases α, ß, and γ with IC50s of >100 μM. In the screen with more than 110 protein targets including enzymes, transporters, ion channels, and receptor, MK-1439 shows an IC50 of greater than 10 μM against all targets except 5-HT2ß, where an IC50 of 2.5 μM is noted in a ligand binding assay. However, the 5-HT2ß activity is not observed in a functional cell-based assay monitoring the accumulation of inositol-1-phosphate. Therefore, the binding activity does not appear to translate into a 5-HT2ß functional response. MK-1439 does not display cytotoxicity in activated CD4+ T cells, PBMCs, monocytes, macrophages proliferating transformed cell lines, such as MT4, SupT1, and HL60 cell lines at concentrations of up to 100 μM. |
| In vivo |
In rats dosed IV at 1 mpk (60%PEG200), the clearance (CL) of MK-1439 is 5.4 mL/min/kg and the half time is 4.4 hr. Its volume of distribution (Vd) is 2.3 L/kg. The oral bioavailability of MK-1439 is 57% (doesed PO at 5 mpk). In dogs dosed IV at 0.5 mpk, the CL of MK-1439 is 0.36 mL/min/kg and half-time is 37 hr. The oral bioavailability of MK-1439 is 52% (doesed PO at 1 mpk). Overall, The pharmacokinetic profile of MK-1439 in preclinical species is favorable. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06155019 | Recruiting | HIV Infections |
Centre de Recherches et d''Etude sur la Pathologie Tropicale et le Sida |
December 15 2023 | -- |
| NCT05630638 | Recruiting | HIV |
University of Liverpool|Liverpool School of Tropical Medicine|Desmond Tutu Health Foundation |
October 10 2023 | Phase 4 |
| NCT05761509 | Active not recruiting | HIV Infections |
Institut de Médecine et d''Epidémiologie Appliquée - Fondation Internationale Léon M''Ba|Merck Sharp & Dohme LLC |
June 8 2023 | -- |
| NCT05536466 | Not yet recruiting | HIV Infections|Bariatric Surgery Candidate |
Radboud University Medical Center |
September 30 2022 | Not Applicable |
| NCT04900974 | Recruiting | HIV Infections|Pregnancy Related |
University of North Carolina Chapel Hill|Merck Sharp & Dohme LLC |
June 9 2022 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































