research use only
Pyrazinamide Bacterial inhibitor
Cat.No.S1762
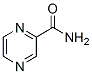
Chemical Structure
Molecular Weight: 123.11
Quality Control
| Related Targets | Integrase Antibiotics Anti-infection Fungal Antiviral COVID-19 Parasite Reverse Transcriptase HIV HCV Protease |
|---|---|
| Other Bacterial Inhibitors | Berberine BTZ043 Racemate Teicoplanin Pefloxacin Mesylate Ornidazole Proanthocyanidins Solithromycin Skatole Berberine Sulfate Furagin |
Solubility
|
In vitro |
DMSO
: 25 mg/mL
(203.07 mM)
Water : 12 mg/mL Ethanol : 2 mg/mL |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 123.11 | Formula | C5H5N3O |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 98-96-4 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Pyrazinoic Acid Amide | Smiles | C1=CN=C(C=N1)C(=O)N | ||
Mechanism of Action
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05329415 | Active not recruiting | Tuberculosis|Diabetes Mellitus |
London North West Healthcare NHS Trust |
February 1 2022 | -- |
| NCT05840809 | Recruiting | Tuberculosis Active|Breastfeeding|Tuberculosis |
University of Liverpool|Infectious Diseases Institute Makerere University College of Health Sciences |
January 20 2022 | -- |
| NCT04972903 | Unknown status | Tuberculosis|Pulmonary |
Institut National de la Santé Et de la Recherche Médicale France|National Agency for Research on AIDS and Viral Hepatitis (ANRS) |
August 2021 | -- |
| NCT04239326 | Completed | Tuberculosis Multidrug-Resistant |
Foundation for Innovative New Diagnostics Switzerland |
April 16 2021 | -- |
| NCT03800381 | Recruiting | Tuberculosis|Human Immunodeficiency Virus|Coinfection |
University of Florida|Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) |
January 28 2019 | -- |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































