research use only
Glyburide (Glibenclamide) Potassium Channel inhibitor
Cat.No.S1716
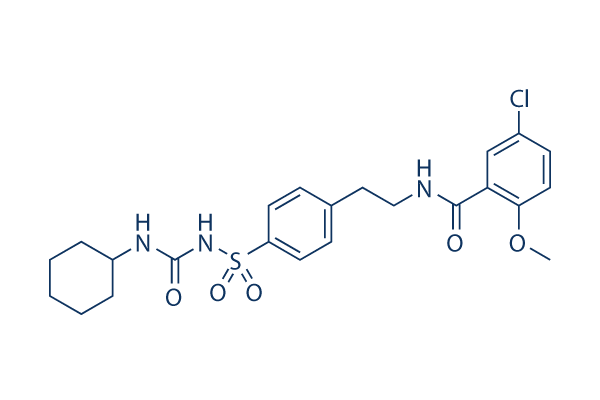
Chemical Structure
Molecular Weight: 494
Quality Control
| Related Targets | CFTR CRM1 CD markers AChR Calcium Channel Sodium Channel GABA Receptor TRP Channel ATPase GluR |
|---|---|
| Other Potassium Channel Inhibitors | Nicorandil TRAM-34 Sophocarpine ML133 HCl Hydralazine HCl Gliquidone PAP-1 VU0071063 Apamin A2793 |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| INS-1E cells | Function assay | 1 h | Stimulation of insulin secretion in rat INS-1E cells after 1 hr by alphaLISA assay in presence of 5 mM glucose, EC50=0.003 μM | |||
| CHO cells | Function assay | Inhibition of human SUR1/Kir6.2 expressed in CHO cells, IC50=0.0043 μM | ||||
| HEK293 cells | Function assay | Inhibition of OATP1B1 (unknown origin) expressed in HEK293 cells using estradiol-17beta-glucuronide substrate, IC50=1.4 μM | ||||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 99 mg/mL
(200.4 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 494 | Formula | C23H28ClN3O5S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 10238-21-8 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Glibenclamide | Smiles | COC1=C(C=C(C=C1)Cl)C(=O)NCCC2=CC=C(C=C2)S(=O)(=O)NC(=O)NC3CCCCC3 | ||
Mechanism of Action
| Targets/IC50/Ki |
Potassium channel
|
|---|---|
| In vitro |
Glyburide (Glibenclamide), a sulfonylurea which has been shown to block the ATP-modulated potassium channel in insulin-secreting cells, causes concentration-dependent shifts to the right (up to 100-fold) of the IC50 value for BRL 34915 and diazoxide, and at 1 μM, abolishes the relaxation response to minoxidil sulfate. This compound increases the apparent affinity of HDL binding to Scavenger receptor class B type I (SR-BI). It blocks SR-BI-mediated selective lipid uptake and efflux at a potency similar to that for its inhibition of ABCA1 (IC50 approximately 275-300 mM). At 6 mM, it reduces the opening of KATP channels, aggravating Ca2+ loading only when applied to dinitrophenol-pretreated myocytes but not when applied with dinitrophenol treatment. In concentrations of 10-500 nM, it produces a dose-dependent inhibition of the potassium channel openers (PCOs) relaxation time course. It also reverses existing Pinacidil relaxation regardless of the degree of pre-existing relaxation, and is able to produce its blockade regardless of the state of K+ channel activation.
|
| In vivo |
S1716 dose-dependently increases urinary Na+ excretion with little change in urinary K+ excretion after i.p. administration (10-100 mg/kg) in saline-loaded conscious rats. S1716 (25 mg/kg i.v.) increases Na+ excretion 350% during the first hour post-treatment without affecting K+ excretion, glomerular filtration rate, mean arterial pressure or heart rate.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05426681 | Recruiting | Acute Spinal Cord Injury |
University of Kentucky |
July 7 2022 | Phase 1 |
| NCT03832595 | Completed | Chronic Kidney Diseases |
University of Pittsburgh|Vanderbilt University Medical Center|National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) |
May 1 2019 | Not Applicable |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































