research use only
Bicalutamide Androgen Receptor antagonist
Cat.No.S1190
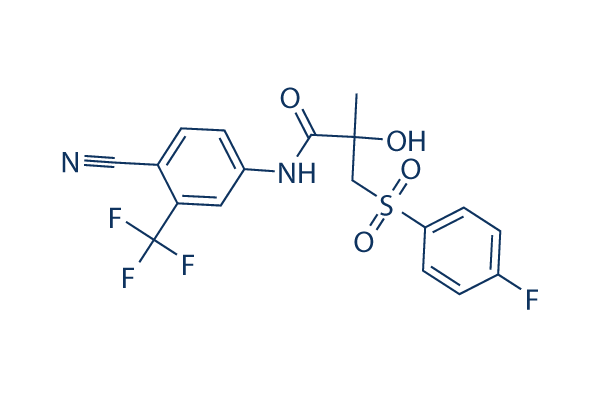
Chemical Structure
Molecular Weight: 430.37
Quality Control
| Related Targets | Adrenergic Receptor Estrogen/progestogen Receptor GPR Glucocorticoid Receptor ACE RAAS Progesterone Receptor Opioid Receptor PGES THR |
|---|---|
| Other Androgen Receptor Inhibitors | Bavdegalutamide (ARV-110) RU58841 EPI-001 Galeterone Cyproterone Acetate Ailanthone 3,3'-Diindolylmethane Proxalutamide (GT0918) GSK-2881078 Chlormadinone acetate |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| human MDA-MB-453 cells | Function assay | Displacement of [3H]R1881 from AR in human MDA-MB-453 cells, EC50=31 nM | ||||
| LNCaP cells | Function assay | Inhibition of [3H]-DHT binding to T877A androgen receptor of LNCaP cells, Ki=35 nM | ||||
| Freestyle293F cells | Function assay | Inhibition of wild type Androgen receptor (unknown origin) expressed in Freestyle293F cells, IC50=0.054 μM | ||||
| HEK293 cells | Function assay | 3 h | Displacement of [17-alpha-methyl-3H]mibolerone from androgen receptor expressed in HEK293 cells after 3 hrs, IC50=54 nM | |||
| MDA453 cells | Function assay | Displacement of [3H]DHT from human androgen receptor in MDA453 cells, Ki=64 nM | ||||
| human MDA-MB-453 cells | Function assay | Displacement of [3H]DHT from AR in human MDA-MB-453 cells, IC50=64 nM | ||||
| COS1 cells | Function assay | Antagonist activity against pSG5-tagged human androgen receptor expressed in COS1 cells assessed as reduction in receptor-mediated transcriptional activity by AR-regulated rat probasin promoter fragment driven firefly luciferase reporter assay, IC50=0.0869 μM | ||||
| HeLa cells | Function assay | Antagonist activity at human androgen receptor expressed in HeLa cells assessed as inhibition of dihydrotestosterone induced transcriptional activity by reporter gene assay, IC50=0.14 μM | ||||
| CV1 cells | Function assay | Binding affinity to human androgen receptor expressed in CV1 cells, Ki=0.151 μM | ||||
| monkey COS7 cells | Function assay | Binding affinity to human androgen receptor expressed in monkey COS7 cells by whole cell binding assay, Ki=0.151 μM | ||||
| COS7 cells | Function assay | Agonist activity at human androgen receptor W741C mutant expressed in COS7 cells assessed as luciferase activity after 24 hrs by reporter gene assay, EC50=0.18 μM | ||||
| CHO-K1 cells | Function assay | 2 h | Displacement of [3H]mibolerone from human AR expressed in CHO-K1 cells after 2 hrs by scintillation counting, IC50=0.2 μM | |||
| human HT-3 cell | Growth inhibition assay | Inhibition of human HT-3 cell growth in a cell viability assay, IC50=0.73134 μM | ||||
| human LNCAP cells | Proliferation assay | 3 days | Antiproliferative activity against human LNCAP cells after 3 days, IC50=0.7327 μM | |||
| human PC3 cells | Function assay | Displacement of [3H]R1881 from androgen receptor in human PC3 cells, EC50=4.3 μM | ||||
| human 22Rv1 cells | Function assay | 3 days | Antagonist activity at androgen receptor H874Y mutant (unknown origin) expressed in human 22Rv1 cells assessed as inhibition of DHT-induced cell growth after 3 days by WST-8 assay, IC50=4.6 μM | |||
| human CCF-STTG1 cell | Growth inhibition assay | Inhibition of human CCF-STTG1 cell growth in a cell viability assay, IC50=4.92929 μM | ||||
| human SCC-25 cell | Growth inhibition assay | Inhibition of human SCC-25 cell growth in a cell viability assay, IC50=6.08656 μM | ||||
| human MKN45 cell | Growth inhibition assay | Inhibition of human MKN45 cell growth in a cell viability assay, IC50=6.9605 μM | ||||
| human ES5 cell | Growth inhibition assay | Inhibition of human ES5 cell growth in a cell viability assay, IC50=8.61154 μM | ||||
| human SK-MEL-3 cell | Growth inhibition assay | Inhibition of human SK-MEL-3 cell growth in a cell viability assay, IC50=10.0964 μM | ||||
| human PC-3 cell | Growth inhibition assay | Inhibition of human PC-3 cell growth in a cell viability assay, IC50=10.2791 μM | ||||
| human NOS-1 cell | Growth inhibition assay | Inhibition of human NOS-1 cell growth in a cell viability assay, IC50=11.2917 μM | ||||
| human LB1047-RCC cell | Growth inhibition assay | Inhibition of human LB1047-RCC cell growth in a cell viability assay, IC50=12.253 μM | ||||
| human CAMA-1 cell | Growth inhibition assay | Inhibition of human CAMA-1 cell growth in a cell viability assay, IC50=12.3926 μM | ||||
| human SAS cell | Growth inhibition assay | Inhibition of human SAS cell growth in a cell viability assay, IC50=13.3081 μM | ||||
| human NCI-H2228 cell | Growth inhibition assay | Inhibition of human NCI-H2228 cell growth in a cell viability assay, IC50=13.7531 μM | ||||
| human NCI-H187 cell | Growth inhibition assay | Inhibition of human NCI-H187 cell growth in a cell viability assay, IC50=16.6616 μM | ||||
| human BFTC-905 cell | Growth inhibition assay | Inhibition of human BFTC-905 cell growth in a cell viability assay, IC50=17.4857 μM | ||||
| human G-361 cell | Growth inhibition assay | Inhibition of human G-361 cell growth in a cell viability assay, IC50=17.826 μM | ||||
| human DU145 cells | Cytotoxic assay | 72 h | Cytotoxicity against ERalpha-deficient human DU145 cells expressing ERbeta assessed as growth inhibition after 72 hrs by MTT assay, IC50=18 μM | |||
| human SW780 cell | Growth inhibition assay | Inhibition of human SW780 cell growth in a cell viability assay | ||||
| human BB49-HNC cell | Growth inhibition assay | Inhibition of human BB49-HNC cell growth in a cell viability assay, IC50=18.9532 μM | ||||
| human KALS-1 cell | Growth inhibition assay | Inhibition of human KALS-1 cell growth in a cell viability assay, IC50=19.6635 μM | ||||
| human AU565 cell | Growth inhibition assay | Inhibition of human AU565 cell growth in a cell viability assay, IC50=19.7402 μM | ||||
| human NCI-H2087 cell | Growth inhibition assay | Inhibition of human NCI-H2087 cell growth in a cell viability assay, IC50=21.0591 μM | ||||
| human RVH-421 cell | Growth inhibition assay | Inhibition of human RVH-421 cell growth in a cell viability assay, IC50=21.5795 μM | ||||
| human SK-CO-1 cell | Growth inhibition assay | Inhibition of human SK-CO-1 cell growth in a cell viability assay, IC50=21.8872 μM | ||||
| human KU-19-19 cell | Growth inhibition assay | Inhibition of human KU-19-19 cell growth in a cell viability assay, IC50=22.0242 μM | ||||
| human NB6 cell | Growth inhibition assay | Inhibition of human NB6 cell growth in a cell viability assay, IC50=22.9135 μM | ||||
| human RO82-W-1 cell | Growth inhibition assay | Inhibition of human RO82-W-1 cell growth in a cell viability assay, IC50=23.1318 μM | ||||
| human LNCAP cells | Cytotoxic assay | 2 days | Cytotoxicity against human LNCAP cells assessed as cell viability after 2 days by cell counting method, IC50=23.79 μM | |||
| human CTB-1 cell | Growth inhibition assay | Inhibition of human CTB-1 cell growth in a cell viability assay, IC50=24.5536 μM | ||||
| human SW48 cell | Growth inhibition assay | Inhibition of human SW48 cell growth in a cell viability assay, IC50=24.6546 μM | ||||
| human TCCSUP cell | Growth inhibition assay | Inhibition of human TCCSUP cell growth in a cell viability assay, IC50=24.7232 μM | ||||
| human DK-MG cell | Growth inhibition assay | Inhibition of human DK-MG cell growth in a cell viability assay, IC50=24.8917 μM | ||||
| human ST486 cell | Growth inhibition assay | Inhibition of human ST486 cell growth in a cell viability assay, IC50=25.7464 μM | ||||
| human H4 cell | Growth inhibition assay | Inhibition of human H4 cell growth in a cell viability assay, IC50=26.9458 μM | ||||
| human SBC-1 cell | Growth inhibition assay | Inhibition of human SBC-1 cell growth in a cell viability assay, IC50=28.3507 μM | ||||
| human CAS-1 cell | Growth inhibition assay | Inhibition of human CAS-1 cell growth in a cell viability assay, IC50=28.6294 μM | ||||
| human OAW-42 cell | Growth inhibition assay | Inhibition of human OAW-42 cell growth in a cell viability assay, IC50=28.7195 μM | ||||
| human HCC1954 cell | Growth inhibition assay | Inhibition of human HCC1954 cell growth in a cell viability assay, IC50=28.7525 μM | ||||
| human MDA-MB-453 cell | Growth inhibition assay | Inhibition of human MDA-MB-453 cell growth in a cell viability assay, IC50=29.907 μM | ||||
| human MCF7 cell | Growth inhibition assay | Inhibition of human MCF7 cell growth in a cell viability assay, IC50=39.301 μM | ||||
| human PC3 cells | Function assay | 100 μM | 48 h | Inhibition of actin based pseudopodia formation in androgen-dependent human PC3 cells at 100 uM after 48 hrs by DAPI staining based fluorescence microscopy assay | ||
| human PC3 cells | Function assay | 0.1-1 μM | Agonist activity at androgen receptor W741C mutant expressed in human PC3 cells assessed as stimulation of receptor transactivation at 0.1 to 1 uM by luciferase reporter gene assay | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 86 mg/mL
(199.82 mM)
Ethanol : 7 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 430.37 | Formula | C18H14F4N2O4S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 90357-06-5 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | ICI-176334 | Smiles | CC(CS(=O)(=O)C1=CC=C(C=C1)F)(C(=O)NC2=CC(=C(C=C2)C#N)C(F)(F)F)O | ||
Mechanism of Action
| Targets/IC50/Ki |
Androgen Receptor
(LNCaP/AR(cs) cells) 0.16 μM
|
|---|---|
| In vitro |
Bicalutamide undergoes an antagonist-to-agonist switch, stimulating AR activity. This compound treatment of LNCaP/AR(cs) cells in absence of the synthetic androgen R1881 results in altered gene expression consistent with its well-documented agonist activity in context of AR overexpression. It induces cell proliferation in a dose-dependent manner, and only partially antagonized the effects of R1881. This chemical treatment also results in a significant amount of nuclear AR, although less than that observed with R1881. It exhibits partial agonist activity as evidenced by induction of DNA binding at AR target genes and incomplete antagonism of the effects of R1881. In absence of R1881, this agent partially activates VP16-AR–mediated transcription, indicative of AR binding to DNA. In LNCaP/AR-luc cells with a stably integrates AR-driven luciferase reporter construct. In the presence of R1881, it shows only weak partial antagonism of VP16-AR–mediated transcription with an IC50 of 0.35 μM. Micromolar bicalutamide causes a significant dose-dependent reduction in clonogenicity. Dual inhibition of the AR and mTOR signaling pathways provides further benefit with the ridaforolimus-this compound combination producing syner -gistic antiproliferative effects in prostate cancer cells in vitro when compared with each agent alone.
|
| Kinase Assay |
Whole-cell competitive binding assays
|
|
Whole-cell competitive binding assays are performed in LNCaP/AR(codon-switch) (LNCaP/AR(cs)) (harbors a mixture of exogenous wild-type AR and endogenous mutant AR (T877A)) and cells propagated in Iscove's or RPMI media supplemented with 10% fetal bovine serum, or during the assay with 10% charcoal-stripped, dextran-treated fetal bovine serum (CSS). Cells are pre-incubated with 18F-FDHT, increasing concentrations (1pM to 1μM) of cold this compound are added, and the assay is performed to measure specific uptake of 18F-FDHT (4). IC50 values are determined using a one site binding model with least squares curve fitting and R2 > 0.99.
|
|
| In vivo |
Single bicalutamide reduces tumor growth by 79%, at defined submaximal doses. The ridaforolimus-this compound combination exhibits improved and potent antitumor activity, almost completely abrogating tumor growth. The combination is also well tolerated, as evidenced by no significant changes in body weight over the course of treatment. Plasma PSA levels are again tightly linked to tumor growth in the combination-treated mice.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | Cytosolic AR / Nuclear AR |
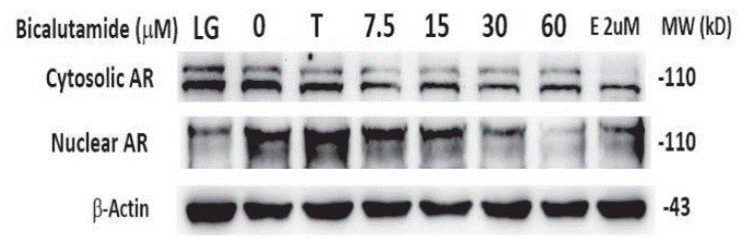
|
30833616 |
| Growth inhibition assay | Cell viability |
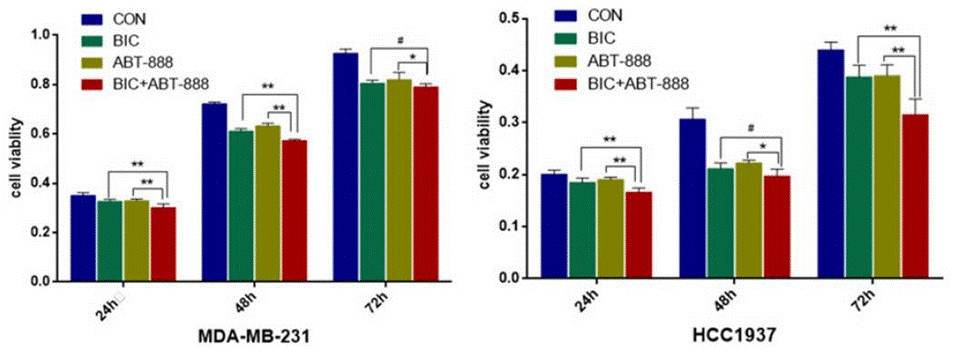
|
27994514 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06222593 | Not yet recruiting | Carcinoma Renal Cell |
State University of New York at Buffalo |
June 1 2024 | Phase 1|Phase 2 |
| NCT04573231 | Recruiting | Breast Cancer|HER2-negative Breast Cancer|Metastatic Breast Cancer |
University of Wisconsin Madison |
May 24 2021 | Phase 2 |
| NCT04443062 | Recruiting | Prostate Cancer |
Radboud University Medical Center|Prostaatkankerstichting|Advanced Accelerator Applications |
July 20 2020 | Phase 2 |
| NCT02910050 | Unknown status | Breast Cancer |
Xu fei|Sun Yat-sen University |
January 2016 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































