research use only
PRN1371 FGFR inhibitor
Cat.No.S8578
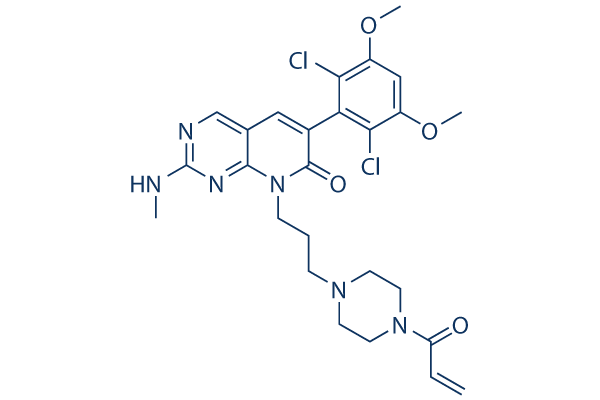
Chemical Structure
Molecular Weight: 561.46
Quality Control
| Related Targets | EGFR VEGFR JAK PDGFR Src HIF FLT FLT3 HER2 Bcr-Abl |
|---|---|
| Other FGFR Inhibitors | PD173074 AZD4547 (Fexagratinib) BLU9931 H3B-6527 Futibatinib (TAS-120) LY2874455 PD-166866 Zoligratinib (Debio-1347) SSR128129E Fisogatinib (BLU-554) |
Solubility
|
In vitro |
DMSO
: 56 mg/mL
(99.73 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 561.46 | Formula | C26H30Cl2N6O4 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1802929-43-6 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CNC1=NC=C2C=C(C(=O)N(C2=N1)CCCN3CCN(CC3)C(=O)C=C)C4=C(C(=CC(=C4Cl)OC)OC)Cl | ||
Mechanism of Action
| Targets/IC50/Ki |
FGFR1
(Cell-free assay) 0.6 nM
FGFR2
(Cell-free assay) 1.3 nM
FGFR3
(Cell-free assay) 4.1 nM
CSF1R
(Cell-free assay) 8.1 nM
FGFR4
(Cell-free assay) 19.3 nM
|
|---|---|
| In vitro |
PRN1371 is an irreversible nanomolar inhibitor of FGFR1−4. This compound presents a unique profile of high biochemical and cellular potency (FGFR1 IC50 = 0.6 nM, SNU16 IC50 = 2.6 nM), prolonged target engagement (FGFR1 occupancy 24 h = 96%), <30% hERG inhibition at 1 μM, and good predicted ADME stability with BME reactivity Kd>100 μM. This chemical maintained high FGFR1 occupancy with improved solubility and exceptional oral bioavailability.
|
| In vivo |
A rat iv (2 mg/kg) PK study of PRN1371 showed rapid clearance (Cl = 160 ml/min/kg), yet dosing po (20 mg/kg) demonstrated high oral exposure (AUC = 4348 h·ng/mL) and a reasonable half-life (t1/2 = 3.8 h). PK studies of this compound in rat, dog, and cynomolgus monkey showed rapid iv clearance in all species; however there were large species differences in oral exposure and bioavailability for monkey compared to rat and dog. In rat, high exposure upon oral dosing (e.g., Cmax = 1785 ng/mL, AUC = 4348 ng·h/mL) and >100% bioavailability (F) suggested good absorption and partial saturation of clearance mechanisms at the 20 mg/kg dose. Unique to the rat, there is a large difference in half-life between the iv (t1/2 = 0.8 h) and po (t1/2 = 3.8 h) routes of administration, also indicative of possible saturation of a clearance mechanism upon oral dosing. In the dogs, the same methylcellulose suspension formulation used for the rat gave low oral absorption and bioavailability (F < 15%). In SNU16 gastric cancer xenograft mouse model, this compound induced a dose-dependent reduction in tumor volume and up to 68% tumor growth inhibition at the highest dose of 10 mg/kg b.i.d. following 27 days of treatment. All doses were well tolerated with no significant body weight loss.
|
References |
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p-STAT5 / STAT5 / p-STAT3 / STAT3 |
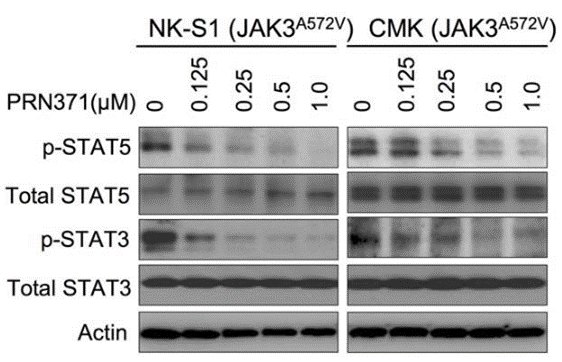
|
29434279 |
| Growth inhibition assay | Cell viability |
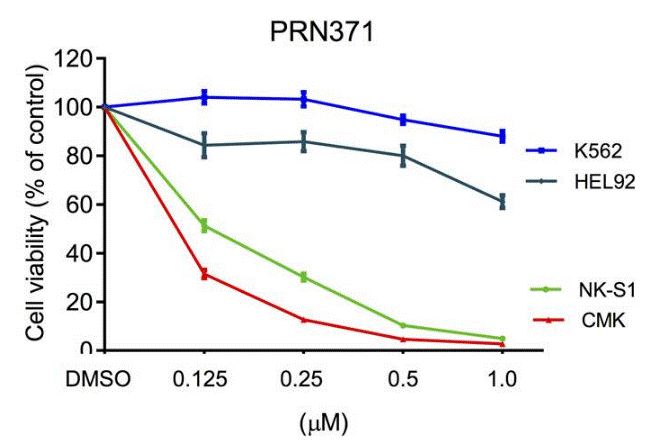
|
29434279 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02608125 | Terminated | Solid Tumors|Metastatic Urothelial Carcinoma & Renal Pelvis & Ureter |
Principia Biopharma a Sanofi Company |
October 28 2015 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































