research use only
Clevudine DNA/RNA Synthesis inhibitor
Cat.No.S3001
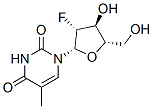
Chemical Structure
Molecular Weight: 260.22
Quality Control
| Related Targets | HDAC PARP ATM/ATR DNA-PK WRN Topoisomerase PPAR Sirtuin Casein Kinase eIF |
|---|---|
| Other DNA/RNA Synthesis Inhibitors | CX-5461 (Pidnarulex) B02 SCR7 Favipiravir (T-705) EED226 RK-33 BMH-21 Triapine (3-AP) Carmofur YK-4-279 |
Solubility
|
In vitro |
DMSO
: 52 mg/mL
(199.83 mM)
Water : 52 mg/mL Ethanol : 4 mg/mL |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 260.22 | Formula | C10H13FN2O5 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 163252-36-6 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Levovir,L-FMAU | Smiles | CC1=CN(C(=O)NC1=O)C2C(C(C(O2)CO)O)F | ||
Mechanism of Action
| Targets/IC50/Ki |
RNA polymerase
DNA polymerase
|
|---|---|
| In vitro |
Clevudine is a potent antiviral agent against HBV (EC50 0.1 μM in HepG2 2.2.15 cells) as well as EBV, which has low cytotoxicities in a variety of cell lines including MT2, CEM, H1 and HepG2 2.2.15 and bone marrow progenitor cells. This compound is metabolized in cells by the cellular thymidine kinase as well as deoxycytidine kinase to its monophosphate, and subsequently to the di- and triphosphate. It is known to act specifically on viral DNA synthesis, and its triphosphate inhibits the HBV DNA synthesis in a dose-dependent manner without being incorporated into the DNA or chain termination. This agent results in increase of the amounts of the diphosphate and triphosphate metabolites of these analogs. Its monophosphate (L-FMAUMP) is a poorer substrate than its D-configuration anomer. It is readily phosphorylated to the corresponding 5'-triphosphate form of the compound in cell culture, which involves the mechanism of action of this chemical. |
| In vivo |
Clevudine is a potent antiviral agent against HBV (EC50 0.1 μM in HepG2 2.2.15 cells) as well as EBV, which has low cytotoxicities in a variety of cell lines including MT2, CEM, H1 and HepG2 2.2.15 and bone marrow progenitor cells. This compound is metabolized in cells by the cellular thymidine kinase as well as deoxycytidine kinase to its monophosphate, and subsequently to the di- and triphosphate. It is known to act specifically on viral DNA synthesis, and its triphosphate inhibits the HBV DNA synthesis in a dose-dependent manner without being incorporated into the DNA or chain termination. This agent results in increase of the amounts of the diphosphate and triphosphate metabolites of these analogs. Its monophosphate (L-FMAUMP) is a poorer substrate than its D-configuration anomer. This chemical is readily phosphorylated to the corresponding 5'-triphosphate form of the compound in cell culture, which involves the mechanism of action of this compound. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05298332 | Terminated | Chronic Hepatitis b |
Antios Therapeutics Inc |
March 29 2022 | Phase 1 |
| NCT05137548 | Completed | Drug Interaction |
Antios Therapeutics Inc |
October 27 2021 | Phase 1 |
| NCT04847440 | Terminated | Hepatitis B Chronic|Hepatitis D |
Antios Therapeutics Inc |
March 30 2021 | Phase 2 |
| NCT04248426 | Completed | Hepatitis B Chronic |
Antios Therapeutics Inc |
February 5 2020 | Phase 1 |
| NCT00558818 | Completed | Chronic Hepatitis B |
Bukwang Pharmaceutical |
June 2007 | Phase 4 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































