research use only
BX-912 PDK inhibitor
Cat.No.S1275
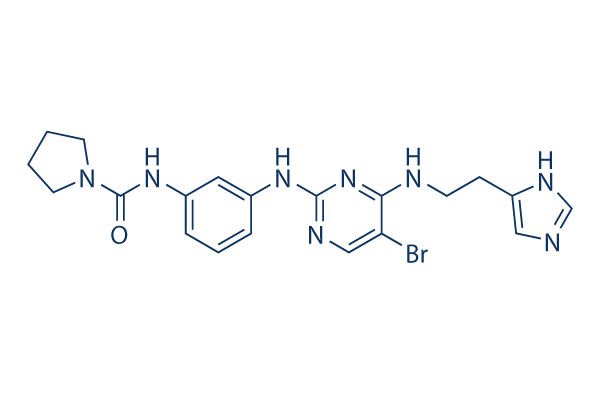
Chemical Structure
Molecular Weight: 471.35
Quality Control
Solubility
|
In vitro |
DMSO
: 94 mg/mL
(199.42 mM)
Ethanol : 94 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 471.35 | Formula | C20H23BrN8O |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 702674-56-4 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | C1CCN(C1)C(=O)NC2=CC=CC(=C2)NC3=NC=C(C(=N3)NCCC4=CN=CN4)Br | ||
Mechanism of Action
| Targets/IC50/Ki |
PDK-1
(Cell-free assay) 12 nM
PKA
(Cell-free assay) 110 nM
KDR
(Cell-free assay) 410 nM
CDK2/CyclinE
(Cell-free assay) 650 nM
Chk1
(Cell-free assay) 830 nM
c-Kit
(Cell-free assay) 850 nM
|
|---|---|
| In vitro |
BX912 prevents ChcK1, PKA, c-kit, and KDR with IC50 of 0.83, 0.11, 0.85, and 0.41 μM, resepectively. BX912 blocks PDK1/Akt signaling in tumor cells and suppresses the anchorage-dependent growth of a variety of tumor cell lines (such as PC-3 cells ) in culture or induces apoptosis. A number of cancer cell lines (such as MDA-468 breast cancer) with elevated Akt activity are >30-fold more sensitive to growth inhibition by PDK1 inhibitor BX912 in soft agar than on tissue culture plastic, consistent with the cell survival function of the PDK1/Akt signaling pathway, which is particularly important for unattached cells. BX912 potently blocks PDK1 enzyme activity in a direct kinase assay format, although BX912 fails to block preactivated AKT2 activity (IC50 > 10 μM). Therefore, BX-912 is a direct inhibitor of PDK1. BX912 is a competitive inhibitor of PDK1 activity with respect to its substrate, ATP, suggesting that BX912 binds to the ATP binding pocket of PDK1. The aminopyrimidine backbone of BX912 adopts a similar orientation in the active site of PDK1. BX912 promotes a pronounced increase in the population of MDA-468 cells with 4 N DNA content, indicative of a block at the G2/M phase of the cell cycle. BX912 also potently inhibits the growth of HCT-116 cells in soft agar, showing a 96% inhibitory effect at a dose of 1 μM. BX912 potently inhibits the growth of PC-3 cells in soft agar, displaying IC50 of 0.32 μM.
|
| Kinase Assay |
Kinase assays
|
|
PDK1 is assayed in a direct kinase assay and a coupled assay format measuring PDK1- and PtdIns-3,4-P2-mediated activation of AKT2. For the coupled assay, the final assay mixture (60 μL) contained: 15 mM MOPS, pH 7.2, 1 mg/mL bovine serum albumin, 18 mM β-glycerol phosphate, 0.7 mM dithiothreitol, 3 mM EGTA, 10 mM MgOAc, 7.5 μM ATP, 0.2 μCi of [γ- 33P]ATP, 7.5 μM biotinylated peptide substrate (biotin-ARRRDGGGAQPFRPRAATF), 0.5 μL of PtdIns-3,4-P2-containing phospholipid vesicles, 60 pg of purified recombinant human PDK1, and 172 ng of purified recombinant human AKT2. After incubation for 2 hours at room temperature, the biotin-labeled peptide is captured from 10 μL of the assay mixture on streptavidin-coated SPA beads, and product formation is measured by scintillation proximity in a Wallac MicroBeta counter. The product formed is proportional to the time of incubation and to the amount of PDK1 and inactive AKT2 added. PDK1 is added at suboptimal levels so that the assay could sensitively detect inhibitors of AKT2 activation as well as direct inhibitor BX912 of PDK1 or AKT2. To measure PDK1 activity directly, the final assay mixture (60 μL) contained 50 mM Tris-HCl, pH 7.5, 0.1 mM EGTA, 0.1 mM EDTA, 0.1% β-mercaptoethanol, 1 mg/mL bovine serum albumin, 10 mM MgOAc, 10 μM ATP, 0.2 μCi of [γ-33P]ATP, 7.5 μM substrate peptide (H2N-ARRRGVTTKTFCGT), and 60 ng of purified recombinant human PDK1. After 4 hours at room temperature, 25 mM EDTA is added and a portion of the reaction mixture on P81 phosphocellulose paper is spotted. The filter paper is washed three times with 0.75% phosphoric acid and once with acetone. After drying, the filter-bound labeled peptide is quantified using a phosphorimager.
|
References |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































