research use only
Enalapril Maleate RAAS inhibitor
Cat.No.S1941
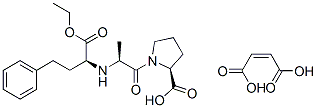
Chemical Structure
Molecular Weight: 492.52
Quality Control
Solubility
|
In vitro |
DMSO
: 98 mg/mL
(198.97 mM)
Ethanol : 17 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 492.52 | Formula | C20H28N2O5.C4H4O4 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 76095-16-4 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | MK-421 Maleate | Smiles | CCOC(=O)C(CCC1=CC=CC=C1)NC(C)C(=O)N2CCCC2C(=O)O.C(=CC(=O)O)C(=O)O | ||
Mechanism of Action
| Targets/IC50/Ki |
ACE
|
|---|---|
| In vitro |
Enalapril Maleate is rapidly converted by ester hydrolysis to enalaprilat, a potent ACE inhibitor; this compound itself is only a weak ACE inhibitor. It lowers peripheral vascular resistance without causing an increase in heart rate.
|
| In vivo |
Enalapril Maleate treatment abolishes the deleterious effects of eNOS deficiency on blood pressure (BP), atherosclerosis, and kidney dysfunction in nnee mice. This compound causes a dose-dependent increase in fore limb strength in the mdx mouse. It causes a dose-dependent reduction of superoxide anion production in tibialis anterior muscle of the mdx mouse as observed by dihydroethidium staining. This chemical (5 mg/kg) reduces the area of muscle necrosis in both gastrocnemious muscle and diaphragm, without significant effect on non-muscle area. It results in significant increases in kidney weight and in concentrations of urinary albumin, N-acetyl-fl-d-glucosamidase (NAG) and serum ET-1 in streptozotocin (STZ)-induced diabetic (STZ-DM) rats as compared with the non-diabetic rats, and the concentration of ET-1 in the kidneys tended to be increased. This compound results in increased mesangial cell proliferation, matrix expansion and enlarged mesangial area in the kidney of the diabetic rats. It reduces increased concentrations of urinary albumin and NAG in the STZ-DM rats to the control values with a slight improvement in the electron microscopic changes.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT04736329 | Completed | Heart Failure With Reduced Ejection Fraction|Renal Insufficiency |
Cairo University |
February 1 2021 | Not Applicable |
| NCT03265574 | Completed | Breast Cancer|Non Hodgkin Lymphoma |
South Tees Hospitals NHS Foundation Trust|Newcastle University|University of Durham|Newcastle-upon-Tyne Hospitals NHS Trust |
October 4 2017 | Phase 3 |
| NCT03051282 | Active not recruiting | Healthy Volunteers |
University of Michigan |
April 1 2017 | Phase 4 |
| NCT03460366 | Recruiting | Obesity Morbid |
Norwegian University of Science and Technology|St. Olavs Hospital|Volvat Medisinsk Senter Stokkan|Namsos Hospital|Alesund Hospital |
November 2 2016 | -- |
| NCT02654678 | Unknown status | Heart Failure|Dilated Cardiomyopathy|Congenital Heart Disease |
Ethicare GmbH |
March 2016 | Phase 2|Phase 3 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































