research use only
Tolcapone COMT inhibitor
Cat.No.S4021
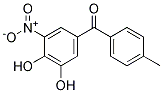
Chemical Structure
Molecular Weight: 273.24
Quality Control
Solubility
|
In vitro |
DMSO
: 55 mg/mL
(201.28 mM)
Ethanol : 55 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 273.24 | Formula |
C14H11NO5
|
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 134308-13-7 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Ro 40-7592 | Smiles | CC1=CC=C(C=C1)C(=O)C2=CC(=C(C(=C2)O)O)[N+](=O)[O-] | ||
Mechanism of Action
| Targets/IC50/Ki |
COMT
30 nM(Ki)
|
|---|---|
| In vitro |
Tolcapone functions as a selective peripheral and central COMT inhibitor, exerting no effect on adrenergic, serotonergic, or cholinergic receptors or other enzymes involved in synthesis or catabolism of catecholamines. This compound produces a concentration-dependent decrease in COMT activity in liver homogenates of developing (3 days-old) and adult (60 days-old) rats with Vmax, Km and IC50 of 5.3 nM/mg/h, 3.3 μM, 41 nM, and 2.9 nM/mg/h, 13.1 μM, 720 nM, respectively. It also produces a concentration-dependent decrease in COMT activity in kidney of developing (3 days-old) and adult (60 days-old) rats with Vmax, Km and IC50 of 2.6 nM/mg/h, 2.7 μM, 8 nM, and 3.5 nM/mg/h, 24 μM, 177 nM, respectively.
|
| In vivo |
Tolcapone orally administrated is able to crosses the blood-brain barrier. Acute administration of this compound increases basal levels of L-DOPA and dihydroxyphenylacetic acid (DOPAC) and decreases basal homovanillic acid (HVA) levels, but does not affect basal dopamine levels. This compound (30 mg/kg p.o.) combined with benserazide (15 mg/kg p.o.) and a low dose of L-dopa (10 mg/kg p.o.) almost completely blockes (for about 6 h) the formation of 3-O-methyldopa (3-OMD) in brain and plasma, producing a long-lasting increase of L-DOPA in plasma and a parallel marked increase of L-DOPA and dopamine in the brain.
It displays behavioural and neurochemical benefits on animals. This chemical (30 mg/kg p.o.) increases the effect of L-DOPA (plus benserazide) on locomotor activity, reserpine-induced hypothermia, and catalepsy induced by pimozide, haloperidol and fluphenazine. It also increases locomotor hyperactivity induced by amphetamine or nomifensine, as well as stereotypy induced by amphetamine (but not apomorphine).
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT03591757 | Completed | Transthyretin Amyloidosis|Amyloidosis Leptomeningeal Transthyretin-Related |
Boston University|Corino Therapeutics Inc. |
October 30 2018 | Early Phase 1 |
| NCT02630043 | Terminated | Neuroblastoma |
Giselle Sholler|Milton S. Hershey Medical Center |
December 2015 | Phase 1 |
| NCT02929485 | Withdrawn | Addiction |
University of California Berkeley|University of California San Francisco |
July 2013 | Phase 4 |
| NCT02080715 | Completed | Healthy |
University of Zurich |
June 2013 | Phase 1 |
| NCT00604591 | Completed | Frontotemporal Lobar Degeneration |
Columbia University|National Institute of Neurological Disorders and Stroke (NINDS) |
July 2011 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































