research use only
Vadimezan (DMXAA) Vascular Disrupting Agent
Cat.No.S1537
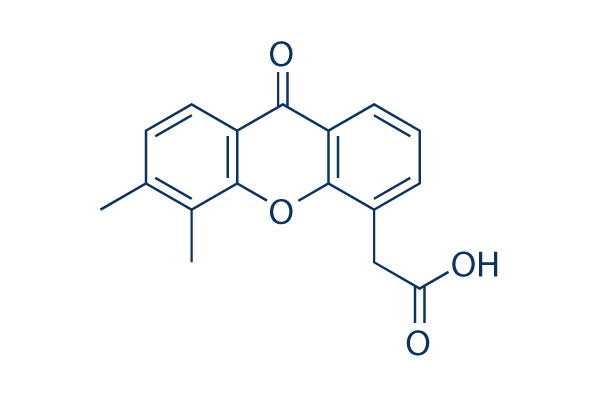
Chemical Structure
Molecular Weight: 282.29
Quality Control
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| human BJ cells | Cytotoxic assay | 24 h | Cytotoxicity against human BJ cells after 24 hrs by MTT assay, CC50=48.9 μM | 24518295 | ||
| HECPP cells | Function assay | 10 ug/mL | Activation of NF-kappaB in HECPP cells at 10 ug/mL | 17616114 | ||
| MCF7 | Antiproliferative assay | 24 hrs | Antiproliferative activity against human MCF7 cells co-treated with pyranoxanthone at 1:1 molar ratio after 24 hrs by MTT assay, IC50 = 11.89 μM. | 29129511 | ||
| MDA-MB-231 | Antiproliferative assay | 24 hrs | Antiproliferative activity against human MDA-MB-231 cells co-treated with pyranoxanthone at 1:1 molar ratio after 24 hrs by MTT assay, IC50 = 12.12 μM. | 29129511 | ||
| K562 | Antiproliferative assay | 24 hrs | Antiproliferative activity against human K562 cells co-treated with pyranoxanthone at 1:1 molar ratio after 24 hrs by MTT assay, IC50 = 19.14 μM. | 29129511 | ||
| HepG2 | Antiproliferative assay | 24 hrs | Antiproliferative activity against human HepG2 cells co-treated with pyranoxanthone at 1:1 molar ratio after 24 hrs by MTT assay, IC50 = 21.25 μM. | 29129511 | ||
| COLO320 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human COLO320 cells after 48 hrs by CCK8 assay, IC50 = 39.5 μM. | 28376372 | ||
| MDA-MB-231 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MDA-MB-231 cells after 48 hrs by CCK8 assay, IC50 = 48.4 μM. | 28376372 | ||
| MDA-MB-231 | Growth inhibition assay | 24 hrs | Growth inhibition of human MDA-MB-231 cells after 24 hrs by MTT assay, IC50 = 48.42 μM. | 29609121 | ||
| MDA-MB-231 | Antiproliferative assay | 24 hrs | Antiproliferative activity against human MDA-MB-231 cells after 24 hrs by MTT assay, IC50 = 48.44 μM. | 29129511 | ||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for MG 63 (6-TG R) cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| MDA-MB-231 | Apoptosis assay | 24 to 96 uM | 48 hrs | Induction of apoptosis in human MDA-MB-231 cells assessed as increase in cleaved caspase-3 expression at 24 to 96 uM after 48 hrs by Western blot analysis | 28376372 | |
| MDA-MB-231 | Apoptosis assay | 24 to 96 uM | 48 hrs | Induction of apoptosis in human MDA-MB-231 cells assessed as increase in cleaved PARP level at 24 to 96 uM after 48 hrs by Western blot analysis | 28376372 | |
| MDA-MB-231 | Function assay | 24 to 96 uM | 48 hrs | Decrease in caspase-3 level in human MDA-MB-231 cells at 24 to 96 uM after 48 hrs by Western blot analysis | 28376372 | |
| MDA-MB-231 | Function assay | 24 to 96 uM | 48 hrs | Increase in p53 level in human MDA-MB-231 cells at 24 to 96 uM after 48 hrs by Western blot analysis | 28376372 | |
| MDA-MB-231 | Function assay | 24 to 96 uM | 48 hrs | Decrease in caspase-9 level in human MDA-MB-231 cells at 24 to 96 uM after 48 hrs by Western blot analysis | 28376372 | |
| MDA-MB-231 | Function assay | 24 to 96 uM | 48 hrs | Decrease in MDM2 level in human MDA-MB-231 cells at 24 to 96 uM after 48 hrs by Western blot analysis | 28376372 | |
| HepG2 | Cell cycle arrest assay | 0.2 uM | 24 hrs | Cell cycle arrest in human HepG2 cells assessed as accumulation at S phase at 0.2 uM after 24 hrs by propidium iodide staining-based flow cytometric method relative to control | 29129511 | |
| HepG2 | Cell cycle arrest assay | 24 hrs | Cell cycle arrest in human HepG2 cells assessed as accumulation at S phase co-treated with pyranoxanthone at 1:1 molar ratio after 24 hrs by propidium iodide staining-based flow cytometric method | 29129511 | ||
| HepG2 | Apoptosis assay | 0.2 uM | 24 hrs | Induction of apoptosis in human HepG2 cells assessed as increase in cleaved caspase-3 levels at 0.2 uM after 24 hrs by Western blot method | 29129511 | |
| HepG2 | Apoptosis assay | 0.2 uM | 24 hrs | Induction of apoptosis in human HepG2 cells assessed as increase in cleaved caspase-9 levels at 0.2 uM after 24 hrs by Western blot method | 29129511 | |
| HepG2 | Apoptosis assay | 24 hrs | Induction of apoptosis in human HepG2 cells assessed as increase in cleaved PARP levels co-treated with pyranoxanthone at 1:1 molar ratio after 24 hrs by Western blot method | 29129511 | ||
| HepG2 | Apoptosis assay | 0.2 uM | 24 hrs | Induction of apoptosis in human HepG2 cells assessed as increase in cleaved PARP levels at 0.2 uM after 24 hrs by Western blot method | 29129511 | |
| HepG2 | Apoptosis assay | 24 hrs | Induction of apoptosis in human HepG2 cells assessed as increase in cleaved caspase-3 levels co-treated with pyranoxanthone at 1:1 molar ratio after 24 hrs by Western blot method | 29129511 | ||
| HepG2 | Apoptosis assay | 24 hrs | Induction of apoptosis in human HepG2 cells assessed as increase in cleaved caspase-9 levels co-treated with pyranoxanthone at 1:1 molar ratio after 24 hrs by Western blot method | 29129511 | ||
| HepG2 | Apoptosis assay | 24 hrs | Induction of apoptosis in human HepG2 cells assessed as downregulation of Bcl-xL expression co-treated with pyranoxanthone at 1:1 molar ratio after 24 hrs by Western blot method | 29129511 | ||
| HepG2 | Apoptosis assay | 24 hrs | Induction of apoptosis in human HepG2 cells assessed as upregulation of Bid expression co-treated with pyranoxanthone at 1:1 molar ratio after 24 hrs by Western blot method | 29129511 | ||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 16 mg/mL
(56.67 mM)
Warmed with 50°C water bath;
Ultrasonicated;
7.5%Sodium bicarbonate : 10 mg/mL (Ultrasonic and heating for 5 minutes.) Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 282.29 | Formula | C17H14O4 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 117570-53-3 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | ASA404, NSC 640488 | Smiles | CC1=C(C2=C(C=C1)C(=O)C3=CC=CC(=C3O2)CC(=O)O)C | ||
Mechanism of Action
| Targets/IC50/Ki |
DT-diaphorase
(Cell-free assay) 20 μM(Ki)
DT-diaphorase
(Cell-free assay) 20 μM(Ki)
|
|---|---|
| In vitro |
In DLD-1 human colon carcinoma cells, Vadimezan (DMXAA) inhibits DT-diaphorase activity without significant effects on the activity of cytochrome b5 reductase and cytochrome P450 reductase. Combination of menadione and this compound leads to an increase in the antiproliferative activity of DLD-1 cells. As an antiviral agent, it inhibits VSV-induced cytotoxicity and influenza virus replication in RAW 264.7 macrophages. A recent study shows that DMXAA has non-immune-mediated inhibitory effects against several kinase members of VEGFR (vascular endothelial growth factor receptor), such as VEGFR2 signalling in human umbilical vein endothelial cells.
|
| Kinase Assay |
DT-diaphorase activity and kinetic analysis of enzyme inhibition
|
|
Purified DT-diaphorase enzyme activity is assayed by measuring the reduction of cytochrome c at 550 nm on a Beckman DU 650 spectrophotometer. Each assay contains cytochrome c (70 μM), NADH (variable concentrations), purified DT-diaphorase (0.032 μg), and menadione (variable concentrations) in a final volume of 1 mL Tris–HCl buffer (50 mM, pH 7.4) containing 0.14% BSA. The reaction is started by the addition of NADH. Rates of reduction are calculated over the initial part of the reaction curve (30 seconds), and results are expressed in terms of μmol cytochrome c reduced/min/mg protein using a molar extinction coefficient of 21.1 mM−1 cm−1 for reduced cytochrome c. Enzyme assays are carried out at room temperature and all reactions are performed in triplicate. Inhibition of purified DT-diaphorase activity is performed by the inclusion of Vadimezan (DMXAA) at various concentrations in the reaction, and inhibition characteristics are determined by varying the concentration of NADH (constant menadione) or menadione (constant NADH) at several concentrations of this compound. Ki values are obtained by plotting 1/V against. The activity of DT-diaphorase in DLD-1 cells is determined by measuring the dicumarol-sensitive reduction of DCPIP at 600 nm. Briefly, DLD-1 cells in mid-exponential growth are harvested by scraping into ice-cold buffer (Tris–HCl, 25 mM, pH 7.4 and 250 mM sucrose) and sonicated on ice. Enzyme assay conditions are 2 mM NADH, 40 μM DCPIP, 20 μL of dicumarol (when required) in a final volume of 1 mL Tris–HCl (25 mM, pH 7.4) containing BSA (0.7 mg/mL). Results are expressed as the dicumarol-sensitive reduction of DCPIP using a molar extinction coefficient of 21 mM−1 cm−1. Protein levels are determined using the Bradford assay
|
|
| In vivo |
Vadimezan (DMXAA) treatment significantly protects C57BL/6J mice infected i.n. with 200 p.f.u. mouse-adapted H1N1 influenza PR8 virus with 60% survival, while the control group only exhibited 20% survival. This compound significantly delays tumor growth induced by chemical carcinogen, increases the time to tumor doubling and increases time from treatment to euthanasia. After its treatment, median tumor doubling time, median tumour tripling time and median time from treatment to euthanasia in tumor-bearing animals are increased by approximately 4.4-, 1.8- and 2.7-fold, respectively.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p-p38 / p38 p-MK2 / pERK / p-JNK |
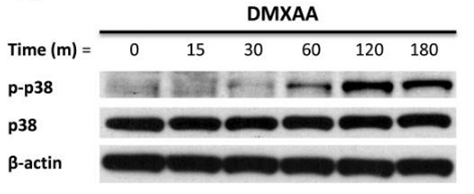
|
21819972 |
| Growth inhibition assay | Cell proliferation |
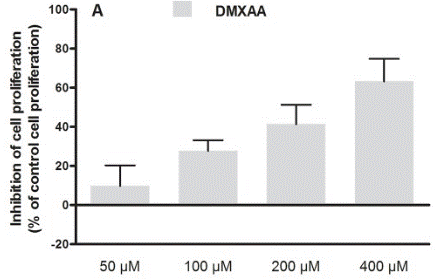
|
30138430 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT00856336 | Completed | Refractory Tumors |
Antisoma Research |
May 2003 | Phase 1 |
| NCT00863733 | Completed | Solid Tumors |
Cancer Research UK|Cancer Society Auckland |
May 1996 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































