research use only
TAK-733 MEK inhibitor
Cat.No.S2617
Chemical Structure
Molecular Weight: 504.23
Quality Control
| Related Targets | ERK p38 MAPK Raf JNK Ras KRas S6 Kinase MAP4K TAK1 Mixed Lineage Kinase |
|---|---|
| Other MEK Inhibitors | PD0325901 (Mirdametinib) U0126-EtOH PD 98059 CI-1040 (PD184352) (E/Z)-BIX02189 Pimasertib (AS-703026) Refametinib (RDEA119) AZD8330 GDC-0623 SL-327 |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| COLO205 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human COLO205 cells after 72 hrs by MTS assay, EC50=0.0021μM. | 21310613 | ||
| A375 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A375 cells after 72 hrs by MTS assay, EC50=0.0031μM. | 21310613 | ||
| COLO205 | Function assay | Inhibition of MEK1 in human COLO205 cells, IC50=0.00692μM. | 24755426 | |||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | |||
| U-2 OS | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for U-2 OS cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 101 mg/mL
(200.3 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 504.23 | Formula | C17H15F2IN4O4 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1035555-63-5 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CN1C2=C(C(=C(C1=O)F)NC3=C(C=C(C=C3)I)F)C(=O)N(C=N2)CC(CO)O | ||
Mechanism of Action
| Targets/IC50/Ki |
MEK1
(Cell-free assay) 3.2 nM
|
|---|---|
| In vitro |
TAK-733 is highly potent and selective MEK allosteric site inhibitor with IC50 of 3.2 nM. This compound shows potent enzymatic and cell activity with an EC50 of 1.9 nM against ERK phosphorylation in cells.
|
| In vivo |
TAK-733 demonstrates broad antitumor activity in mouse xenograft models of human cancer including models of melanoma, colorectal, NSCLC, pancreatic and breast cancer. This compound is well tolerated with pharmacokinetics and pharmacodynamics that support once-daily oral dosing in humans. It shows maximally efficacious doses at once daily orally doses of 10 mg/kg.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Growth inhibition assay | Cell viability |
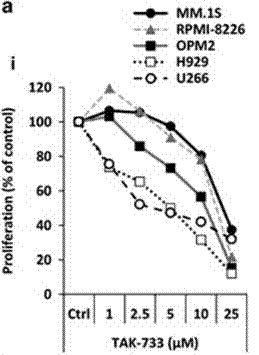
|
26918363 |
| Western blot | p-ERK / p-AKT / p-S6R / p-GSK3 pRb / Cyclin E |
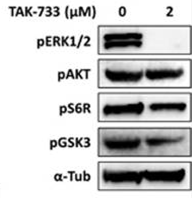
|
26918363 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT01613261 | Withdrawn | Advanced Nonhematologic Malignancies |
Millennium Pharmaceuticals Inc. |
August 2013 | Phase 1 |
| NCT00948467 | Completed | Advanced Non-hematologic Malignancies|Advanced Metastatic Melanoma |
Millennium Pharmaceuticals Inc. |
December 2009 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































