research use only
Methyclothiazide
Cat.No.S4057
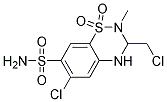
Chemical Structure
Molecular Weight: 360.24
Quality Control
Solubility
|
In vitro |
DMSO
: 72 mg/mL
(199.86 mM)
Ethanol : 4 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 360.24 | Formula | C9H11Cl2N3O4S2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 135-07-9 | -- | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CN1C(NC2=CC(=C(C=C2S1(=O)=O)S(=O)(=O)N)Cl)CCl | ||
Mechanism of Action
| Features |
A diuretic-antihypertensive agent, and is a member of the benzothiadiazine (thiazide) class of drugs.
|
|---|---|
| In vitro |
Methyclothiazide (0.1 mM) reduces Ca2+ contractures with maximal inhibition of 90.4% in SHR aortic rings with endothelium. The inhibitory effect of this compound on Ca2+ contracture is significantly but not totally abolished by the NO synthase inhibitor in SHR aortic rings with functional endothelium. This chemical induces endothelium-dependent inhibition of the vasoconstrictor responses to NE and AVP only in aortas from spontaneously hypertensive rats (SHR) rather than normotensive Wistar Kyoto rats (WKY), and the maximal vasoconstrictive effect of NE and AVP is decreased by 59% and 32.3%, respectively. It inhibits renin release in rat kidney slices.
|
| In vivo |
Methyclothiazide (0.1 g/L for the first 5 weeks and 0.2 g/L for the second 2 weeks, drinking water) attenuates development of hypertension in salt-sensitive (DS) rats, but does not affect blood pressure in salt-resistant (DR) ones. The compound-treated DOCA-salt rats weigh less than controls or the nonsupplemented DOCA-salt rats at weeks 3 and 4. This chemical added to the drinking water of DOCA-salt rats attenuates the elevation of blood pressure.
|
References |
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































