research use only
TRIM5α Antibody [E3H20]
Cat.No.: F8463
Application:
Reactivity:
Usage Information
| Dilution |
|---|
|
| Application |
|---|
| WB, IP |
| Reactivity |
|---|
| Human, Monkey |
| Source |
|---|
| Rabbit Monoclonal Antibody |
| Storage Buffer |
|---|
| PBS, pH 7.2+50% Glycerol+0.05% BSA+0.01% NaN3 |
| Storage (from the date of receipt) |
|---|
| -20°C (avoid freeze-thaw cycles), 2 years |
| Predicted MW |
|---|
| 56 kDa |
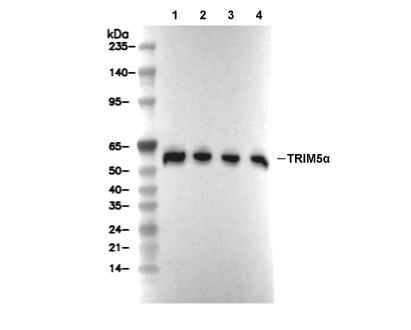
| Positive Control | RL cells, THP-1 + TPA cells, Jurkat cells, Hep G2 cells, MCF7 cells, COS-7 cells |
|---|---|
| Negative Control |
Biological Description
| Specificity |
|---|
| TRIM5α Antibody [E3H20] detects endogenous levels of total TRIM5α protein. |
| Clone |
|---|
| E3H20 |
| Synonym(s) |
|---|
| Tripartite motif-containing protein 5; Ring finger protein 88; TRIM5; RNF88 |
| Background |
|---|
| TRIM5α belongs to the tripartite motif (TRIM) family of proteins characterized by an N-terminal RING domain with E3 ubiquitin ligase activity, B-box domains, and a coiled-coil region that mediates oligomerization, followed by a C-terminal B30.2/SPRY domain responsible for specific ligand recognition. The coiled-coil domain enables higher-order assembly into hexagonal lattices that match the symmetry of retroviral capsid surfaces, while the SPRY domain directly engages determinants on the viral capsid lattice in a species-variable manner, with rhesus monkey TRIM5α showing high avidity for HIV-1 capsid and human TRIM5α preferring N-tropic murine leukemia virus capsid. Upon binding the intact incoming retroviral core via the SPRY domain, TRIM5α accelerates premature uncoating, converting the particulate capsid into soluble forms in the cytosol without relying on proteasomal degradation or major covalent modifications to the capsid protein, thereby disrupting early post-entry events prior to reverse transcription. Concurrently, TRIM5α partners with the UBC13/UEV1A heterodimeric E2 ubiquitin-conjugating enzyme to synthesize free, unattached K63-linked polyubiquitin chains through its RING domain, which bind TAB2/3 and trigger autophosphorylation of the TAK1 kinase complex at threonine 187, leading to downstream activation of AP-1 and NF-κB transcription factors that drive inflammatory cytokine and chemokine expression. This dual mechanism integrates retroviral restriction with innate immune sensing, where capsid lattice interaction amplifies ubiquitin chain production and signaling potency scales with TRIM5α binding avidity to the capsid, enhancing production of mediators like IL-6, IL-8, CXCL10, and PTGS2 upon challenge by restriction-sensitive retroviruses. TRIM5α operates in myeloid cells and restricts diverse retroviruses, including HIV-1 in Old World monkeys and MLV strains in humans, contributing to species-specific viral tropism and bolstering antiviral states induced by pathogen-associated patterns like LPS through TAK1-dependent pathways. |
| References |
|---|
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.