research use only
TFIIB Antibody [H2N6]
Cat.No.: F4717
Application:
Reactivity:
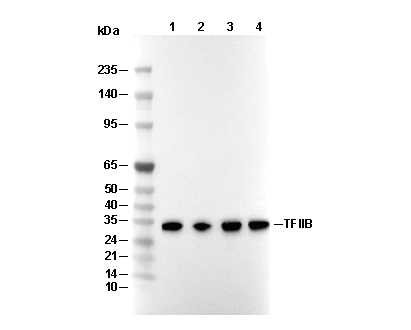
Experiment Essentials
WB
Recommended wet transfer conditions: 200 mA, 60 min.
Recommended wet transfer conditions: 200 mA, 60 min.
Usage Information
| Dilution |
|---|
|
| Application |
|---|
| WB, IP |
| Reactivity |
|---|
| Human, Mouse, Rat, Monkey |
| Source |
|---|
| Mouse Monoclonal Antibody |
| Storage Buffer |
|---|
| PBS, pH 7.2+50% Glycerol+0.05% BSA+0.01% NaN3 |
| Storage (from the date of receipt) |
|---|
| -20°C (avoid freeze-thaw cycles), 2 years |
| Predicted MW |
|---|
| 32 kDa |
| Positive Control | HeLa cells; NIH/3T3 cells; H-4-II-E cells; COS cells |
|---|---|
| Negative Control |
Experimental Methods
| WB |
|---|
Experimental Protocol:
Sample preparation
1. Tissue: Lyse the tissue sample by adding an appropriate volume of ice-cold RIPA/Nuclear Lysis Buffer (containing Protease Inhibitor Cocktail),and homogenize the tissue at a low temperature. 2. Adherent cell: Aspirate the culture medium and wash the cells with ice-cold PBS twice. Lyse the cells by adding an appropriate volume of RIPA/Nuclear Lysis Buffer (containing Protease Inhibitor Cocktail) and put the sample on ice for 5 min. 3. Suspension cell: Transfer the culture medium to a pre-cooled centrifuge tube. Centrifuge and aspirate the supernatant. Wash the cells with ice-cold PBS twice. Lyse the cells by adding an appropriate volume of RIPA/Nuclear Lysis Buffer (containing Protease Inhibitor Cocktail) and put the sample on ice for 5 min. 4. Place the lysate into a pre-cooled microcentrifuge tube. Centrifuge at 4°C for 15 min. Collect the supernatant;
5. Remove a small volume of lysate to determine the protein concentration;
6. Combine the lysate with protein loading buffer. Boil 20 µL sample under 95-100°C for 5 min. Centrifuge for 5 min after cool down on ice.
Electrophoretic separation
1. According to the concentration of extracted protein, load appropriate amount of protein sample and marker onto SDS-PAGE gels for electrophoresis. Recommended separating gel (lower gel) concentration: 10%. Reference Table for Selecting SDS-PAGE Separation Gel Concentrations 2. Power up 80V for 30 minutes. Then the power supply is adjusted (110 V~150 V), the Marker is observed, and the electrophoresis can be stopped when the indicator band of the predyed protein Marker where the protein is located is properly separated. (Note that the current should not be too large when electrophoresis, too large current (more than 150 mA) will cause the temperature to rise, affecting the result of running glue. If high currents cannot be avoided, an ice bath can be used to cool the bath.)
Transfer membrane
1. Take out the converter, soak the clip and consumables in the pre-cooled converter;
2. Activate PVDF membrane with methanol for 1 min and rinse with transfer buffer;
3. Install it in the order of "black edge of clip - sponge - filter paper - filter paper - glue -PVDF membrane - filter paper - filter paper - sponge - white edge of clip"; 4. The protein was electrotransferred to PVDF membrane. ( 0.45 µm PVDF membrane is recommended ) Reference Table for Selecting PVDF Membrane Pore Size Specifications Recommended conditions for wet transfer: 200 mA, 60 min. ( Note that the transfer conditions can be adjusted according to the protein size. For high-molecular-weight proteins, a higher current and longer transfer time are recommended. However, ensure that the transfer tank remains at a low temperature to prevent gel melting.)
Block
1. After electrotransfer, wash the film with TBST at room temperature for 5 minutes;
2. Incubate the film in the blocking solution for 1 hour at room temperature;
3. Wash the film with TBST for 3 times, 5 minutes each time.
Antibody incubation
1. Use 5% skim milk powder to prepare the primary antibody working liquid (recommended dilution ratio for primary antibody 1:1000), gently shake and incubate with the film at 4°C overnight; 2. Wash the film with TBST 3 times, 5 minutes each time;
3. Add the secondary antibody to the blocking solution and incubate with the film gently at room temperature for 1 hour;
4. After incubation, wash the film with TBST 3 times for 5 minutes each time.
Antibody staining
1. Add the prepared ECL luminescent substrate (or select other color developing substrate according to the second antibody) and mix evenly;
2. Incubate with the film for 1 minute, remove excess substrate (keep the film moist), wrap with plastic film, and expose in the imaging system. |
Biological Description
| Specificity |
|---|
| TFIIB Antibody [H2N6] detects endogenous levels of total TFIIB protein. |
| Subcellular Location |
|---|
| Chromosome, Nucleus |
| Uniprot ID |
|---|
| Q00403 |
| Clone |
|---|
| H2N6 |
| Synonym(s) |
|---|
| Transcription initiation factor IIB; General transcription factor TFIIB; S300-II; GTF2B; TF2B; TFIIB |
| Background |
|---|
| TFIIB is a ubiquitous general transcription factor essential for RNA polymerase II pre-initiation complex assembly at TATA-containing promoters, bridging promoter-bound TFIID, specifically TBP, with Pol II-TFIIF and precisely positioning the transcription start site. TFIIB includes an N-terminal zinc ribbon domain spanning amino acids 1 to 75 with a C(X)2C(X)12-15H(X)4-6C motif that coordinates zinc for interactions with RPB1 dock2 and RPB9, a B-reader helix-loop from amino acids 155 to 169 that inserts into the Pol II template tunnel and recognizes the initiator element through complementary T-A base pairing, a B-linker from amino acids 170 to 200 forming a flexible helix that constrains the active site metal B, and a C-terminal core domain of five imperfect tandem repeats forming a saddle-shaped platform with two DNA-binding modules contacting BREu and BREd elements flanking the TATA box via basic residues such as Arg180 and Lys35. TFIIB mediates ordered PIC assembly: its core clamps the TBP-induced DNA bend of about 90 degrees, extending protein contacts both upstream and downstream of the TATA box, recruits Pol II-TFIIF via the zinc ribbon, stabilizing the dock angle at around 45 degrees, the B-reader aligns the initiator template strand, with mutations shifting the transcription start site by about 10 nucleotides, and the linker allosterically rearranges the Pol II active site cleft by repositioning Glu414 and coordinating Mg2+B to enable promoter melting and abortive initiation followed by promoter clearance. Phosphorylation of TFIIB by TFIIH kinase triggers its dissociation after synthesis of a 10-nucleotide RNA. TFIIB enforces promoter specificity by integrating TATA, initiator, and BRE elements, responds to transcriptional activators such as VP16 and GAL4 via the N-terminal region, and regulates Pol II pause-release, while its archaeal homolog stimulates basal initiation by about fivefold. TFIIB mutations such as R178H cause neurodevelopmental disorders by impairing PIC stability, and its overexpression in certain cancers hijacks Pol II for oncogene transcription. |
| References |
|---|
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.