research use only
SHOC2 Antibody [H23M19]
Cat.No.: F5330
Application:
Reactivity:
Usage Information
| Dilution |
|---|
|
| Application |
|---|
| WB |
| Reactivity |
|---|
| Human, Mouse, Rat, Monkey |
| Source |
|---|
| Rabbit Monoclonal Antibody |
| Storage Buffer |
|---|
| PBS, pH 7.2+50% Glycerol+0.05% BSA+0.01% NaN3 |
| Storage (from the date of receipt) |
|---|
| -20°C (avoid freeze-thaw cycles), 2 years |
| Predicted MW |
|---|
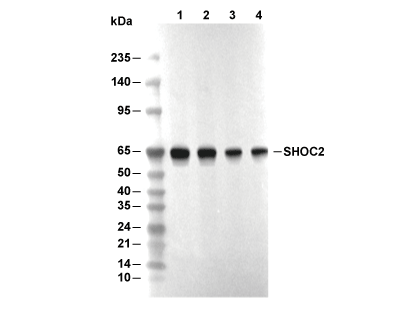
| 64 kDa |
Biological Description
| Specificity |
|---|
| SHOC2 Antibody [H23M19] detects endogenous levels of total SHOC2 protein. |
| Clone |
|---|
| H23M19 |
| Synonym(s) |
|---|
| Leucine-rich repeat protein SHOC-2; Ras-binding protein Sur-8 homolog; SHOC2 |
| Background |
|---|
| SHOC2 is a leucine‑rich repeat (LRR)‑containing scaffold protein that positively regulates RAS‑MAPK signaling and intersects with the PI3K/Akt axis, functioning as a modular platform that coordinates the assembly and dynamics of specific signaling complexes. SHOC2 folds into a concave LRR‑rich domain that mediates multiple protein–protein interactions, enabling its recruitment to activated RAS‑GTP and to the catalytic subunit of protein phosphatase 1 (PP1), while its N‑terminal region participates in competitive membrane association and regulatory switches. SHOC2 forms a ternary complex with a regulatory PP1 subunit and the GTP‑bound small GTPase M‑RAS, and this complex serves as a dedicated accelerator of RAF–MEK–ERK signaling, promoting RAF dimerization and MEK/ERK phosphorylation downstream of receptor‑tyrosine‑kinase‑driven growth‑factor inputs. SHOC2 engages the p110α catalytic subunit of PI3K, thereby linking RAS‑MAPK activation to PI3K/Akt stimulation and broadening the range of downstream transcriptional and metabolic outputs that depend on coordinated ERK and Akt signaling. This dual engagement of ERK and PI3K/Akt pathways underlies SHOC2’s role in promoting cell motility, cytoskeletal remodeling, and invasive behavior in epithelial and cancer cell contexts, where its expression supports processes that increase migratory capacity and metastatic potential. A germline SHOC2 mutation that introduces an N‑terminal myristoylation motif causes aberrant constitutive membrane targeting of the protein, leading to sustained, growth‑factor‑independent enhancement of RAF–ERK signaling and underpinning the hyperactive RAS–MAPK phenotype in Noonan‑like syndrome with loose anagen hair. |
| References |
|---|
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.