research use only
Robo1 Antibody [A23H22]
Cat.No.: F4036
Application:
Reactivity:
Experiment Essentials
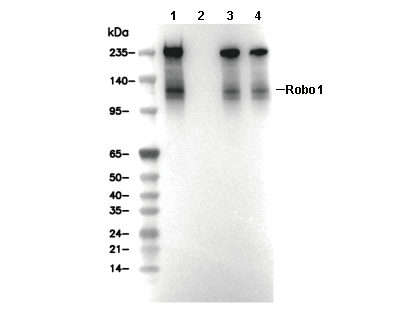
WB
Recommended SDS-PAGE separating gel concentration: 5%.
Recommended SDS-PAGE separating gel concentration: 5%.
Usage Information
| Dilution |
|---|
|
| Application |
|---|
| WB, IP, FCM |
| Reactivity |
|---|
| Mouse, Rat, Human |
| Source |
|---|
| Rabbit Monoclonal Antibody |
| Storage Buffer |
|---|
| PBS, pH 7.2+50% Glycerol+0.05% BSA+0.01% NaN3 |
| Storage (from the date of receipt) |
|---|
| -20°C (avoid freeze-thaw cycles), 2 years |
| Predicted MW Observed MW |
|---|
| 181 kDa 38 kDa, 120-130 kDa, 215,250 kDa |
| *Why do the predicted and actual molecular weights differ? The following reasons may explain differences between the predicted and actual protein molecular weight. Post-translational modifications(e.g., phosphorylation, glycosylation); Splice variants and isoforms; Relative charge; Multimerization. |
| Positive Control | Mouse E14.5 brain tissue; C57 E18 brain tissue; Rat E14 brain tissue; U-87 MG cells; HepG2 cells; HeLa cells; HAP1 cells |
|---|---|
| Negative Control | LoVo cells; MCF7 cells |
Experimental Methods
| WB |
|---|
Experimental Protocol:
Sample preparation
1. Tissue: Lyse the tissue sample by adding an appropriate volume of ice-cold RIPA/NP-40 Lysis Buffer (containing Protease Inhibitor Cocktail),and homogenize the tissue at a low temperature. 2. Adherent cell: Aspirate the culture medium and wash the cells with ice-cold PBS twice. Lyse the cells by adding an appropriate volume of RIPA/NP-40 Lysis Buffer (containing Protease Inhibitor Cocktail) and put the sample on ice for 5 min. 3. Suspension cell: Transfer the culture medium to a pre-cooled centrifuge tube. Centrifuge and aspirate the supernatant. Wash the cells with ice-cold PBS twice. Lyse the cells by adding an appropriate volume of RIPA/NP-40 Lysis Buffer (containing Protease Inhibitor Cocktail) and put the sample on ice for 5 min. 4. Place the lysate into a pre-cooled microcentrifuge tube. Centrifuge at 4°C for 15 min. Collect the supernatant;
5. Remove a small volume of lysate to determine the protein concentration;
6. Combine the lysate with protein loading buffer. Boil 20 µL sample under 95-100°C for 5 min. Centrifuge for 5 min after cool down on ice.
Electrophoretic separation
1. According to the concentration of extracted protein, load appropriate amount of protein sample and marker onto SDS-PAGE gels for electrophoresis. Recommended separating gel (lower gel) concentration: 5%. Reference Table for Selecting SDS-PAGE Separation Gel Concentrations 2. Power up 80V for 30 minutes. Then the power supply is adjusted (110 V~150 V), the Marker is observed, and the electrophoresis can be stopped when the indicator band of the predyed protein Marker where the protein is located is properly separated. (Note that the current should not be too large when electrophoresis, too large current (more than 150 mA) will cause the temperature to rise, affecting the result of running glue. If high currents cannot be avoided, an ice bath can be used to cool the bath.)
Transfer membrane
1. Take out the converter, soak the clip and consumables in the pre-cooled converter;
2. Activate PVDF membrane with methanol for 1 min and rinse with transfer buffer;
3. Install it in the order of "black edge of clip - sponge - filter paper - filter paper - glue -PVDF membrane - filter paper - filter paper - sponge - white edge of clip"; 4. The protein was electrotransferred to PVDF membrane. ( 0.45 µm PVDF membrane is recommended ) Reference Table for Selecting PVDF Membrane Pore Size Specifications Recommended conditions for wet transfer: 200 mA, 120 min. ( Note that the transfer conditions can be adjusted according to the protein size. For high-molecular-weight proteins, a higher current and longer transfer time are recommended. However, ensure that the transfer tank remains at a low temperature to prevent gel melting.)
Block
1. After electrotransfer, wash the film with TBST at room temperature for 5 minutes;
2. Incubate the film in the blocking solution for 1 hour at room temperature;
3. Wash the film with TBST for 3 times, 5 minutes each time.
Antibody incubation
1. Use 5% skim milk powder to prepare the primary antibody working liquid (recommended dilution ratio for primary antibody 1:1000), gently shake and incubate with the film at 4°C overnight; 2. Wash the film with TBST 3 times, 5 minutes each time;
3. Add the secondary antibody to the blocking solution and incubate with the film gently at room temperature for 1 hour;
4. After incubation, wash the film with TBST 3 times for 5 minutes each time.
Antibody staining
1. Add the prepared ECL luminescent substrate (or select other color developing substrate according to the second antibody) and mix evenly;
2. Incubate with the film for 1 minute, remove excess substrate (keep the film moist), wrap with plastic film, and expose in the imaging system. |
Biological Description
| Specificity |
|---|
| Robo1 Antibody [A23H22] detects endogenous levels of total Robo1 protein. |
| Subcellular Location |
|---|
| Cell membrane, Cell projection, Membrane |
| Uniprot ID |
|---|
| Q9Y6N7 |
| Clone |
|---|
| A23H22 |
| Synonym(s) |
|---|
| DUTT1; ROBO1; Roundabout homolog 1; Deleted in U twenty twenty; H-Robo-1 |
| Background |
|---|
| Robo1, or Roundabout guidance receptor 1, also known as DUTT1, is the founding member of the Robo family of single-pass transmembrane glycoproteins, mediating Slit-dependent repulsion that is critical for midline axon guidance, neuronal migration, and tissue morphogenesis. Its extracellular domain comprises five immunoglobulin-like domains Ig1 to Ig5, with Ig1 and Ig2 forming the primary Slit2-D2 binding site and assembling into compact dimers or tetramers via Ig4 contacts, further rigidified by heparan sulfate binding on Ig1 and Ig2. This is followed by three cysteine-rich fibronectin type III domains, Fn1 to Fn3, with Fn2 and Fn3 near the membrane for cell surface positioning, a transmembrane domain, and an intracellular domain of about 150 amino acids containing four conserved CC0 to CC3 motifs. The CC2 and CC3 motifs recruit Abelson kinase and Dock for cytoskeletal repulsion, while CC1 mediates LogR and DCC antagonism. Robo1's core function centers on Slit2-induced signaling, where the ternary Slit2N-Ig1-heparan sulfate complex triggers Robo1 oligomerization without major conformational shift, and the cytoplasmic CC motifs recruit Dock and ELMO, leading to Rac1 and CDC42 activation, Arp2/3 and WAVE complex engagement, and branched F-actin-mediated repulsion at growth cones and leading edges, thereby inhibiting Netrin and DCC attraction and establishing anterior-posterior boundaries. Robo1 patterns commissural axons by facilitating the anterior turn after midline crossing, restricts glial cells to the midline, governs vascular branching in the lung and kidney, and regulates tumor cell migration, with redundancy among Robo1, Robo2, Robo3, and Robo4 ensuring developmental robustness. Epigenetic silencing through hypermethylation or mutations in Robo1 promotes metastasis in breast and lung cancers by increasing invasion, advances glioma progression, and contributes to autism through neural circuit defects, while reactivation of the Slit2-Robo1 pathway restores tumor suppression. |
| References |
|---|
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.