research use only
Phospho-eIF4E (Ser209) Antibody [G23F17]
Cat.No.: F5399
Application:
Reactivity:
Usage Information
| Dilution |
|---|
|
| Application |
|---|
| WB, IP |
| Reactivity |
|---|
| Human, Mouse, Rat |
| Source |
|---|
| Rabbit Monoclonal Antibody |
| Storage Buffer |
|---|
| PBS, pH 7.2+50% Glycerol+0.05% BSA+0.01% NaN3 |
| Storage (from the date of receipt) |
|---|
| -20°C (avoid freeze-thaw cycles), 2 years |
| Predicted MW |
|---|
| 25 kDa |
Biological Description
| Specificity |
|---|
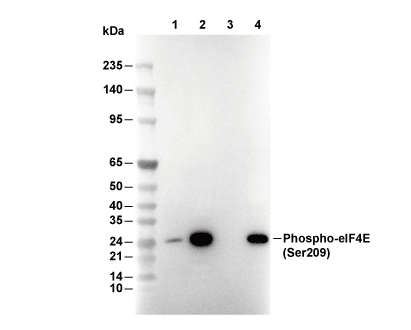
| Phospho-eIF4E (Ser209) Antibody [G23F17] detects endogenous levels of total eIF4E protein only when it is phosphorylated at Ser209. |
| Clone |
|---|
| G23F17 |
| Synonym(s) |
|---|
| Eukaryotic translation initiation factor 4E; eIF-4E; EIF4E |
| Background |
|---|
| Phospho‑eIF4E (Ser209) marks a key regulatory site on the mRNA 5′ cap‑binding initiation factor 4E (eIF4E), a central node in the control of cap‑dependent translation that functions within the eIF4F complex alongside the scaffold eIF4G and the RNA helicase eIF4A. eIF4E recognizes the 7‑methylguanosine cap of 5′‑capped mRNAs, and its interaction with eIF4G stabilizes the eIF4F complex and facilitates ribosome recruitment, while post‑translational modification at Ser209 by the MAPK‑activated kinases Mnk1 and Mnk2 fine‑tunes both the affinity and functional output of eIF4E without disrupting eIF4E–eIF4G binding per se. Activation of ERK and p38 MAPK cascades mobilizes Mnk1/2 to phosphorylate eIF4E at Ser209 within the C‑terminal domain, an event that enhances the translation of a subset of mRNAs encoding growth‑ and survival‑related proteins such as cyclins and anti‑apoptotic factors, thereby coupling extracellular cues to selective production of malignancy‑associated proteins. Ser209 phosphorylation increases resistance to oxidative, nutrient, and cytotoxic stresses by promoting recovery‑phase protein synthesis and upregulating pro‑survival factors such as Mcl‑1, and in tumorigenesis persistent eIF4E Ser209 phosphorylation drives proliferation, metastasis, and therapeutic resistance, whereas blocking Mnk‑dependent eIF4E phosphorylation suppresses eIF4E‑driven oncogenic translation and tumor growth. |
| References |
|---|
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.