research use only
Phospho-α-Synuclein (Ser129) Antibody [G19P15]
Cat.No.: F4724
Application:
Reactivity:
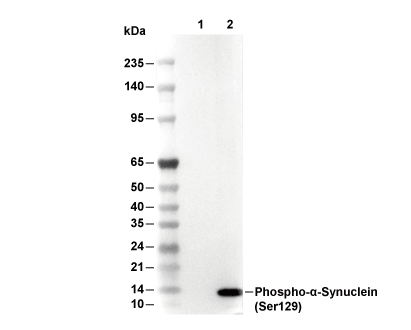
Usage Information
| Dilution |
|---|
|
| Application |
|---|
| WB, IP, IF |
| Reactivity |
|---|
| Human, Mouse, Rat |
| Source |
|---|
| Rabbit Monoclonal Antibody |
| Storage Buffer |
|---|
| PBS, pH 7.2+50% Glycerol+0.05% BSA+0.01% NaN3 |
| Storage (from the date of receipt) |
|---|
| -20°C (avoid freeze-thaw cycles), 2 years |
| Predicted MW |
|---|
| 18 kDa |
Biological Description
| Specificity |
|---|
| Phospho-α-Synuclein (Ser129) Antibody [G19P15] detects endogenous levels of total α-Synuclein protein only when it is phosphorylated at Ser129. |
| Clone |
|---|
| G19P15 |
| Synonym(s) |
|---|
| Alpha-synuclein; Non-A beta component of AD amyloid; Non-A4 component of amyloid precursor; SNCA |
| Background |
|---|
| Phospho‑α‑synuclein (Ser129) is a major post‑translational modification of α‑synuclein in the central nervous system, where the soluble, intrinsically disordered α‑synuclein natively adopts an extended conformation that can adopt α‑helical or β‑sheet‑rich configurations depending on lipid and oligomerization context; in pathological aggregates such as Lewy bodies and Lewy neurites, nearly all deposited α‑synuclein is phosphorylated at Ser129, contrasting with low basal Ser129‑phosphorylation levels in healthy brain tissue. The Ser129 residue lies within the C‑terminal region of α‑synuclein, adjacent to clusters of acidic residues that influence charge‑dependent interactions with membranes, chaperones, and components of the ubiquitin–proteasome and autophagy systems, and its phosphorylation alters both net charge and local conformation of the C terminus. Phosphorylation at Ser129 is catalyzed by multiple kinases, including GSK‑3β, which directly phosphorylates α‑synuclein at this site in vitro and in cellular models, and whose hyperactivity in parkinsonian systems correlates with increased Ser129‑phosphorylation and accumulation of α‑synuclein pathology. In parallel, other kinases such as Polo‑like kinases 2 and 3, GRK family members, and casein kinase 2 can also phosphorylate Ser129 under different activity or stress conditions, thereby linking neuronal activity, proteostatic stress, and inflammatory signaling to the phosphorylation state of α‑synuclein. Extensive Ser129 phosphorylation is associated with altered aggregation kinetics, increased persistence of fibrillar species, and localization of phospho‑α‑synuclein to Lewy‑body‑like inclusions, while phosphorylation‑deficient or phospho‑mimetic mutants can modulate aggregation propensity and toxicity in a context‑dependent fashion. Accumulation of Ser129‑phosphorylated α‑synuclein correlates with mitochondrial dysfunction, impaired axonal transport, and synaptic failure, and its detection in nigral and cortical regions is widely used as a pathological marker in Parkinson’s disease and related synucleinopathies. |
| References |
|---|
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.