research use only
NRF1 Antibody [A16D19]
Cat.No.: F4713
Application:
Reactivity:
Usage Information
| Dilution |
|---|
|
| Application |
|---|
| WB, IP, ChIP |
| Reactivity |
|---|
| Human, Mouse, Rat, Monkey |
| Source |
|---|
| Rabbit Monoclonal Antibody |
| Storage Buffer |
|---|
| PBS, pH 7.2+50% Glycerol+0.05% BSA+0.01% NaN3 |
| Storage (from the date of receipt) |
|---|
| -20°C (avoid freeze-thaw cycles), 2 years |
| Predicted MW |
|---|
| 68 kDa |
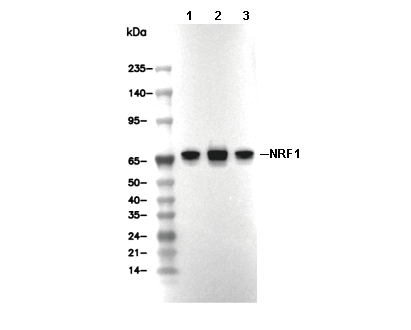
| Positive Control | 293 cells; HepG2 cells; HeLa cells; PC3 cells |
|---|---|
| Negative Control |
Experimental Methods
| WB |
|---|
Experimental Protocol:
Sample preparation
1. Tissue: Lyse the tissue sample by adding an appropriate volume of ice-cold RIPA/Nuclear Lysis Buffer (containing Protease Inhibitor Cocktail),and homogenize the tissue at a low temperature. 2. Adherent cell: Aspirate the culture medium and wash the cells with ice-cold PBS twice. Lyse the cells by adding an appropriate volume of RIPA/Nuclear Lysis Buffer (containing Protease Inhibitor Cocktail) and put the sample on ice for 5 min. 3. Suspension cell: Transfer the culture medium to a pre-cooled centrifuge tube. Centrifuge and aspirate the supernatant. Wash the cells with ice-cold PBS twice. Lyse the cells by adding an appropriate volume of RIPA/Nuclear Lysis Buffer (containing Protease Inhibitor Cocktail) and put the sample on ice for 5 min. 4. Place the lysate into a pre-cooled microcentrifuge tube. Centrifuge at 4°C for 15 min. Collect the supernatant;
5. Remove a small volume of lysate to determine the protein concentration;
6. Combine the lysate with protein loading buffer. Boil 20 µL sample under 95-100°C for 5 min. Centrifuge for 5 min after cool down on ice.
Electrophoretic separation
1. According to the concentration of extracted protein, load appropriate amount of protein sample and marker onto SDS-PAGE gels for electrophoresis. Recommended separating gel (lower gel) concentration: 10%. Reference Table for Selecting SDS-PAGE Separation Gel Concentrations 2. Power up 80V for 30 minutes. Then the power supply is adjusted (110 V~150 V), the Marker is observed, and the electrophoresis can be stopped when the indicator band of the predyed protein Marker where the protein is located is properly separated. (Note that the current should not be too large when electrophoresis, too large current (more than 150 mA) will cause the temperature to rise, affecting the result of running glue. If high currents cannot be avoided, an ice bath can be used to cool the bath.)
Transfer membrane
1. Take out the converter, soak the clip and consumables in the pre-cooled converter;
2. Activate PVDF membrane with methanol for 1 min and rinse with transfer buffer;
3. Install it in the order of "black edge of clip - sponge - filter paper - filter paper - glue -PVDF membrane - filter paper - filter paper - sponge - white edge of clip"; 4. The protein was electrotransferred to PVDF membrane. ( 0.45 µm PVDF membrane is recommended ) Reference Table for Selecting PVDF Membrane Pore Size Specifications Recommended conditions for wet transfer: 200 mA, 120 min. ( Note that the transfer conditions can be adjusted according to the protein size. For high-molecular-weight proteins, a higher current and longer transfer time are recommended. However, ensure that the transfer tank remains at a low temperature to prevent gel melting.)
Block
1. After electrotransfer, wash the film with TBST at room temperature for 5 minutes;
2. Incubate the film in the blocking solution for 1 hour at room temperature;
3. Wash the film with TBST for 3 times, 5 minutes each time.
Antibody incubation
1. Use 5% skim milk powder to prepare the primary antibody working liquid (recommended dilution ratio for primary antibody 1:1000), gently shake and incubate with the film at 4°C overnight; 2. Wash the film with TBST 3 times, 5 minutes each time;
3. Add the secondary antibody to the blocking solution and incubate with the film gently at room temperature for 1 hour;
4. After incubation, wash the film with TBST 3 times for 5 minutes each time.
Antibody staining
1. Add the prepared ECL luminescent substrate (or select other color developing substrate according to the second antibody) and mix evenly;
2. Incubate with the film for 1 minute, remove excess substrate (keep the film moist), wrap with plastic film, and expose in the imaging system. |
Biological Description
| Specificity |
|---|
| NRF1 Antibody [A16D19] detects endogenous levels of total NRF1 protein. |
| Subcellular Location |
|---|
| Nucleus |
| Uniprot ID |
|---|
| Q16656 |
| Clone |
|---|
| A16D19 |
| Synonym(s) |
|---|
| Nuclear respiratory factor 1; NRF-1; Alpha palindromic-binding protein (Alpha-pal); NRF1 |
| Background |
|---|
| NRF1, a member of the cap'n'collar CNC basic leucine zipper bZIP transcription factor family, coordinates nuclear-mitochondrial communication by activating around 50 nuclear genes encoding respiratory chain subunits, mitochondrial transcription factors such as TFAM, and regulators of mitochondrial biogenesis essential for oxidative phosphorylation and cellular energy homeostasis. It contains an N-terminal acidic transactivation domain, STD, spanning amino acids 1 to 120 and responsible for recruiting CBP and p300 coactivators, a central Neh1 domain that functions in bZIP DNA binding and dimerization with partners such as NRF1, NRF2, GABPβ, and MAFG, recognizing GC dyads in T(C/G)GCCGCGCAG palindromic motifs, and is flanked by conserved serine-rich NST and acidic AD regions. A neo-epitope neo-NRF1, about 41 kDa, is generated by proteolytic processing through DDP5 or DBD1 cleavage after ER stress, exposing the N-terminal neo-STD for constitutive activity and bypassing canonical N-glycosylation inhibition. NRF1 is promoter-specific transactivation, as it binds both canonical cytochrome c oxidase subunit promoters, such as COXIV and COXVa, and non-canonical sites like TFAM and TFB1M or TFB2, inducing mitochondrial mass with about a two-fold increase in mtDNA to nuclear DNA ratio, increasing cristae density, and boosting respiratory capacity by around 30 percent for Complex I and IV. Neo-NRF1 maintains OXPHOS under proteasome inhibition, while the NST and AD motifs mediate p21 repression and link to cell cycle arrest. BNRF1 drives myogenesis by regulating myogenin and Pax7, supports neuronal maturation through mitochondrial trafficking, and is essential for erythroid differentiation and heme biosynthesis, with PGC1α amplifying NRF1 action via ERRα and ERRγ coactivation. NRF1 haploinsufficiency leads to mitochondrial myopathy due to mtDNA depletion, while overexpression in liver cancer correlates with E2F1 activation and poor prognosis. |
| References |
|---|
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.