research use only
JARID1C Antibody [J9D7]
Cat.No.: F5827
Application:
Reactivity:
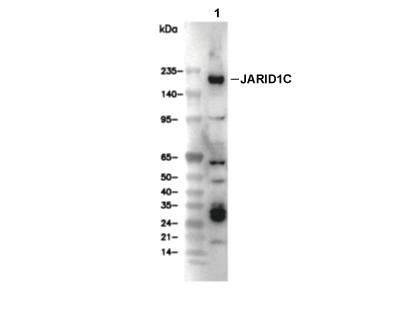
Experiment Essentials
WB
Recommended SDS-PAGE separating gel concentration: 5%.
Exposure time of at least 90s is recommended.
Recommended SDS-PAGE separating gel concentration: 5%.
Exposure time of at least 90s is recommended.
Usage Information
| Dilution |
|---|
|
| Application |
|---|
| WB, IP |
| Reactivity |
|---|
| Human, Mouse |
| Source |
|---|
| Rabbit Monoclonal Antibody |
| Storage Buffer |
|---|
| PBS, pH 7.2+50% Glycerol+0.05% BSA+0.01% NaN3 |
| Storage (from the date of receipt) |
|---|
| -20°C (avoid freeze-thaw cycles), 2 years |
| Predicted MW |
|---|
| 180 kDa |
| Positive Control | NCCIT cells; F9 cells |
|---|---|
| Negative Control |
Experimental Methods
| WB |
|---|
Experimental Protocol:
Sample preparation
1. Tissue: Lyse the tissue sample by adding an appropriate volume of ice-cold RIPA/Nuclear Lysis Buffer (containing Protease Inhibitor Cocktail),and homogenize the tissue at a low temperature. 2. Adherent cell: Aspirate the culture medium and wash the cells with ice-cold PBS twice. Lyse the cells by adding an appropriate volume of RIPA/Nuclear Lysis Buffer (containing Protease Inhibitor Cocktail) and put the sample on ice for 5 min. 3. Suspension cell: Transfer the culture medium to a pre-cooled centrifuge tube. Centrifuge and aspirate the supernatant. Wash the cells with ice-cold PBS twice. Lyse the cells by adding an appropriate volume of RIPA/Nuclear Lysis Buffer (containing Protease Inhibitor Cocktail) and put the sample on ice for 5 min. 4. Place the lysate into a pre-cooled microcentrifuge tube. Centrifuge at 4°C for 15 min. Collect the supernatant;
5. Remove a small volume of lysate to determine the protein concentration;
6. Combine the lysate with protein loading buffer. Boil 20 µL sample under 95-100°C for 5 min. Centrifuge for 5 min after cool down on ice.
Electrophoretic separation
1. According to the concentration of extracted protein, load appropriate amount of protein sample and marker onto SDS-PAGE gels for electrophoresis. Recommended separating gel (lower gel) concentration: 5%. Reference Table for Selecting SDS-PAGE Separation Gel Concentrations 2. Power up 80V for 30 minutes. Then the power supply is adjusted (110 V~150 V), the Marker is observed, and the electrophoresis can be stopped when the indicator band of the predyed protein Marker where the protein is located is properly separated. (Note that the current should not be too large when electrophoresis, too large current (more than 150 mA) will cause the temperature to rise, affecting the result of running glue. If high currents cannot be avoided, an ice bath can be used to cool the bath.)
Transfer membrane
1. Take out the converter, soak the clip and consumables in the pre-cooled converter;
2. Activate PVDF membrane with methanol for 1 min and rinse with transfer buffer;
3. Install it in the order of "black edge of clip - sponge - filter paper - filter paper - glue -PVDF membrane - filter paper - filter paper - sponge - white edge of clip"; 4. The protein was electrotransferred to PVDF membrane. ( 0.45 µm PVDF membrane is recommended ) Reference Table for Selecting PVDF Membrane Pore Size Specifications Recommended conditions for wet transfer: 200 mA, 120 min. ( Note that the transfer conditions can be adjusted according to the protein size. For high-molecular-weight proteins, a higher current and longer transfer time are recommended. However, ensure that the transfer tank remains at a low temperature to prevent gel melting.)
Block
1. After electrotransfer, wash the film with TBST at room temperature for 5 minutes;
2. Incubate the film in the blocking solution for 1 hour at room temperature;
3. Wash the film with TBST for 3 times, 5 minutes each time.
Antibody incubation
1. Use 5% skim milk powder to prepare the primary antibody working liquid (recommended dilution ratio for primary antibody 1:1000), gently shake and incubate with the film at 4°C overnight; 2. Wash the film with TBST 3 times, 5 minutes each time;
3. Add the secondary antibody to the blocking solution and incubate with the film gently at room temperature for 1 hour;
4. After incubation, wash the film with TBST 3 times for 5 minutes each time.
Antibody staining
1. Add the prepared ECL luminescent substrate (or select other color developing substrate according to the second antibody) and mix evenly;
2. Incubate with the film for 1 minute, remove excess substrate (keep the film moist), wrap with plastic film, and expose in the imaging system. (Exposure time of at least 90s is recommended) |
Biological Description
| Specificity |
|---|
| JARID1C Antibody [J9D7] detects endogenous levels of total JARID1C protein. |
| Subcellular Location |
|---|
| Nucleus |
| Uniprot ID |
|---|
| P41229 |
| Clone |
|---|
| J9D7 |
| Synonym(s) |
|---|
| Lysine-specific demethylase 5C; Histone demethylase JARID1C; Jumonji/ARID domain-containing protein 1C; Protein SmcX; Protein Xe169; [histone H3]-trimethyl-L-lysine(4) demethylase 5C; KDM5C; DXS1272E; JARID1C; SMCX; XE169 |
| Background |
|---|
| JARID1C (also known as KDM5C or SMCX) is a histone demethylase of the JARID1/KDM5 family that belongs to the larger JmjC‑domain‑containing class of lysine‑demethylating enzymes and functions as an H3K4me3/me2‑specific eraser of transcription‑activating marks. It contains a JmjC catalytic domain, a JmjN domain, a BRIGHT domain, a C5HC2 zinc‑finger region, and multiple PHD domains that bind methylated histone tails, including H3K4me3, and these modules together enable sequence‑specific recruitment to chromatin and cooperative regulation of target promoters. JARID1C is a core component of repressive complexes that include HDAC1, HDAC2, G9a (EHMT2), and the transcription factor REST, and through this association, it binds REST‑responsive elements in non‑neuronal cells and stem cells to silence neuronal‑specific genes such as BDNF, SCG10, and SCN2A, thereby preventing premature neuronal differentiation and maintaining non‑neural lineage identity. By demethylating H3K4me3 and H3K4me2 at these loci, JARID1C reduces RNA Polymerase II occupancy and transcriptional output, linking its enzymatic activity directly to chromatin‑condensed, low‑expression states. In humans, JARID1C is X‑linked, and mutations in the JARID1C gene are associated with X‑linked intellectual disability, short stature, hyperreflexia, and epilepsy, and many disease‑linked variants map to the JmjC domain, impairing demethylase activity or complex‑binding capacity. In several cancers, including clear‑cell renal cell carcinoma, reduced JARID1C function correlates with derepression of growth‑promoting loci and genomic instability, and in other contexts, JARID1C has been shown to repress HPV E6/E7 oncogenes. |
| References |
|---|
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.