research use only
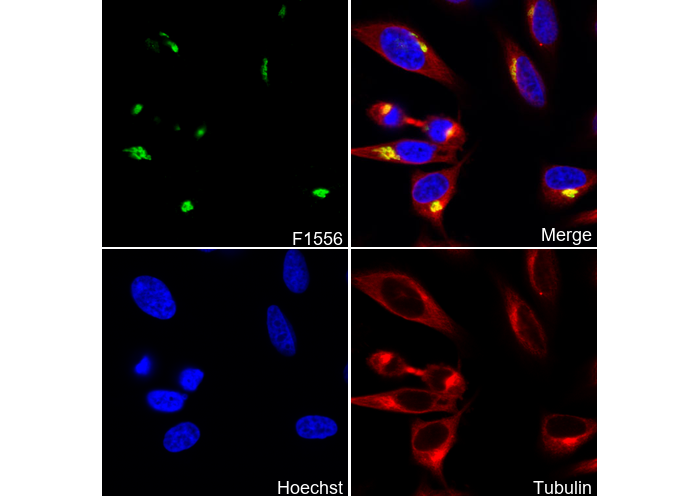
Giantin (Golgi Marker) Antibody [F5N17]
Cat.No.: F1556
Application:
Reactivity:
Usage Information
| Dilution |
|---|
|
| Application |
|---|
| IF |
| Reactivity |
|---|
| Human |
| Source |
|---|
| Mouse Monoclonal Antibody |
| Storage Buffer |
|---|
| PBS, pH 7.2+50% Glycerol+0.05% BSA+0.01% NaN3 |
| Storage (from the date of receipt) |
|---|
| -20°C (avoid freeze-thaw cycles), 2 years |
| Positive Control | HeLa cells; Hek293 cells |
|---|---|
| Negative Control |
Experimental Methods
| IF |
|---|
Experimental Protocol:
Sample Preparation
1. Adherent Cells: Place a clean, sterile coverslip in a culture dish. Once the cells grow to near confluence as a monolayer, remove the coverslip for further use.
2. Suspension Cells: Seed the cells onto a clean, sterile slide coated with poly-L-lysine.
3. Frozen Sections: Allow the slide to thaw at room temperature. Wash it with pure water or PBS for 2 times, 3 minutes each time.
4. Paraffin Sections: Deparaffinization and rehydration. Wash the slide with pure water or PBS for 3 times, 3 minutes each time. Then perform antigen retrieval.
Fixation
1. Fix the cell coverslips/spots or tissue sections at room temperature using a fixative such as 4% paraformaldehyde (4% PFA) for 10-15 minutes.
2. Wash the sample with PBS for 3 times, 3 minutes each time.
Permeabilization
1.Add a detergent such as 0.1–0.3% Triton X-100 to the sample and incubate at room temperature for 10–20 minutes.
(Note: This step is only required for intracellular antigens. For antigens expressed on the cell membrane, this step is unnecessary.)
Wash the sample with PBS for 3 times, 3 minutes each time.
Blocking
Add blocking solution and incubate at room temperature for at least 1 hour. (Common blocking solutions include: serum from the same source as the secondary antibody, BSA, or goat serum.)
Note: Ensure the sample remains moist during and after the blocking step to prevent drying, which can lead to high background.
Immunofluorescence Staining (Day 1)
1. Remove the blocking solution and add the diluted primary antibody.
2. Incubate the sample in a humidified chamber at 4°C overnight.
Immunofluorescence Staining (Day 2)
1. Remove the primary antibody and wash with PBST for 3 times, 5 minutes each time.
2. Add the diluted fluorescent secondary antibody and incubate in the dark at 4°C for 1–2 hours.
3. Remove the secondary antibody and wash with PBST for 3 times, 5 minutes each time.
4. Add diluted DAPI and incubate at room temperature in the dark for 5–10 minutes.
5. Wash with PBST for 3 times, 5 minutes each time.
Mounting
1. Mount the sample with an anti-fade mounting medium.
2. Allow the slide to dry at room temperature overnight in the dark.
3. Store the slide in a slide storage box at 4°C, protected from light.
|
Biological Description
| Specificity |
|---|
| Giantin (Golgi Marker) Antibody [F5N17] detects endogenous levels of total Giantin protein. |
| Subcellular Location |
|---|
| Golgi apparatus, Membrane |
| Uniprot ID |
|---|
| Q14789 |
| Clone |
|---|
| F5N17 |
| Synonym(s) |
|---|
| Golgin subfamily B member 1; 372 kDa Golgi complex-associated protein (GCP372); Giantin; Macrogolgin; GOLGB1 |
| Background |
|---|
| Giantin (GOLGB1), also known as Giantin Golgi protein, is the largest mammalian golgin, a family of long coiled-coil proteins localized primarily to the cis/medial Golgi and serving as a key structural marker of Golgi integrity. Giantin consists of an extended rod-like coiled-coil cytoplasmic domain that forms a rigid scaffold, a C-terminal transmembrane anchor, and interaction motifs for proteins such as p115, GM130, GRASP65, Rab1, and SNAREs, enabling it to tether vesicles without reliance on specific key residues. It regulates Golgi architecture by inhibiting excessive fusion and interconnections between stacks and cisternae, ensuring the formation of discrete reaction compartments for sequential glycosylation; knockdown of Giantin leads to reduced fenestrae size and number, accelerated membrane protein diffusion, and increased cisternal connectivity, which slows intra-Golgi transport and impacts protein and lipid glycosylation. Biologically, Giantin tethers COPI vesicles to the cis-Golgi, facilitates ER-Golgi and intra-Golgi trafficking through complexes with coat proteins, and supports Golgi reassembly after mitosis or disruption by agents like brefeldin A or ethanol. It coordinates the secretory pathway for surface and lysosomal protein sorting, with its loss altering glycosylation patterns and cargo kinetics. Disease associations include Congenital Disorders of Glycosylation (CDG), connective tissue disorders, ciliopathies (from dynein-2 mislocalization), and impaired secretion due to trafficking defects. |
| References |
|---|
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.