research use only
CBS Antibody [J23E1]
Cat.No.: F5031
Application:
Reactivity:
Usage Information
| Dilution |
|---|
|
| Application |
|---|
| WB |
| Reactivity |
|---|
| Human, Mouse, Rat |
| Source |
|---|
| Rabbit Monoclonal Antibody |
| Storage Buffer |
|---|
| PBS, pH 7.2+50% Glycerol+0.05% BSA+0.01% NaN3 |
| Storage (from the date of receipt) |
|---|
| -20°C (avoid freeze-thaw cycles), 2 years |
| Predicted MW |
|---|
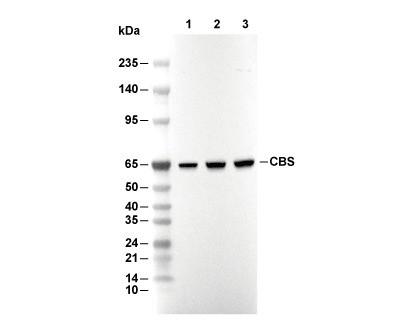
| 61 kDa |
| Positive Control | MCF7 cells; HeLa cells; Hs578T cells; LNCaP cells |
|---|---|
| Negative Control |
Experimental Methods
| WB |
|---|
Experimental Protocol:
Sample preparation
1. Tissue: Lyse the tissue sample by adding an appropriate volume of ice-cold RIPA/NP-40 Lysis Buffer (containing Protease Inhibitor Cocktail),and homogenize the tissue at a low temperature. 2. Adherent cell: Aspirate the culture medium and wash the cells with ice-cold PBS twice. Lyse the cells by adding an appropriate volume of RIPA/NP-40 Lysis Buffer (containing Protease Inhibitor Cocktail) and put the sample on ice for 5 min. 3. Suspension cell: Transfer the culture medium to a pre-cooled centrifuge tube. Centrifuge and aspirate the supernatant. Wash the cells with ice-cold PBS twice. Lyse the cells by adding an appropriate volume of RIPA/NP-40 Lysis Buffer (containing Protease Inhibitor Cocktail) and put the sample on ice for 5 min. 4. Place the lysate into a pre-cooled microcentrifuge tube. Centrifuge at 4°C for 15 min. Collect the supernatant;
5. Remove a small volume of lysate to determine the protein concentration;
6. Combine the lysate with protein loading buffer. Boil 20 µL sample under 95-100°C for 5 min. Centrifuge for 5 min after cool down on ice.
Electrophoretic separation
1. According to the concentration of extracted protein, load appropriate amount of protein sample and marker onto SDS-PAGE gels for electrophoresis. Recommended separating gel (lower gel) concentration: 10%. Reference Table for Selecting SDS-PAGE Separation Gel Concentrations 2. Power up 80V for 30 minutes. Then the power supply is adjusted (110 V~150 V), the Marker is observed, and the electrophoresis can be stopped when the indicator band of the predyed protein Marker where the protein is located is properly separated. (Note that the current should not be too large when electrophoresis, too large current (more than 150 mA) will cause the temperature to rise, affecting the result of running glue. If high currents cannot be avoided, an ice bath can be used to cool the bath.)
Transfer membrane
1. Take out the converter, soak the clip and consumables in the pre-cooled converter;
2. Activate PVDF membrane with methanol for 1 min and rinse with transfer buffer;
3. Install it in the order of "black edge of clip - sponge - filter paper - filter paper - glue -PVDF membrane - filter paper - filter paper - sponge - white edge of clip"; 4. The protein was electrotransferred to PVDF membrane. ( 0.45 µm PVDF membrane is recommended ) Reference Table for Selecting PVDF Membrane Pore Size Specifications Recommended conditions for wet transfer: 200 mA, 120 min. ( Note that the transfer conditions can be adjusted according to the protein size. For high-molecular-weight proteins, a higher current and longer transfer time are recommended. However, ensure that the transfer tank remains at a low temperature to prevent gel melting.)
Block
1. After electrotransfer, wash the film with TBST at room temperature for 5 minutes;
2. Incubate the film in the blocking solution for 1 hour at room temperature;
3. Wash the film with TBST for 3 times, 5 minutes each time.
Antibody incubation
1. Use 5% skim milk powder to prepare the primary antibody working liquid (recommended dilution ratio for primary antibody 1:1000), gently shake and incubate with the film at 4°C overnight; 2. Wash the film with TBST 3 times, 5 minutes each time;
3. Add the secondary antibody to the blocking solution and incubate with the film gently at room temperature for 1 hour;
4. After incubation, wash the film with TBST 3 times for 5 minutes each time.
Antibody staining
1. Add the prepared ECL luminescent substrate (or select other color developing substrate according to the second antibody) and mix evenly;
2. Incubate with the film for 1 minute, remove excess substrate (keep the film moist), wrap with plastic film, and expose in the imaging system. |
Biological Description
| Specificity |
|---|
| CBS Antibody [J23E1] detects endogenous levels of total CBS protein. |
| Subcellular Location |
|---|
| Cytoplasm, Nucleus |
| Uniprot ID |
|---|
| P35520 |
| Clone |
|---|
| J23E1 |
| Synonym(s) |
|---|
| Cystathionine beta-synthase; Beta-thionase; Serine sulfhydrase; CBS |
| Background |
|---|
| Cystathionine beta-synthase (CBS) is the rate-limiting pyridoxal 5'-phosphate (PLP)-dependent enzyme in the transsulfuration pathway. It catalyzes the β-replacement of serine's hydroxyl group by homocysteine to form cystathionine, channeling sulfur from methionine to cysteine biosynthesis, and generates hydrogen sulfide (H₂S) via alternative cysteine-dependent reactions. Human CBS forms a homotetramer with an N-terminal heme-binding domain that binds Fe-porphyrin to sense redox and CO changes, a central PLP catalytic domain containing the active site where Lysine 47 forms Schiff base intermediates and stabilizes carbanion or aminoacrylate species through electrostatic interactions, and C-terminal tandem CBS domains that bind S-adenosylmethionine (AdoMet) for allosteric activation by rigidifying the tetramer and enhancing PLP site accessibility. CBS maintains homocysteine homeostasis, which is essential for vascular health, with AdoMet-induced activation boosting flux under high-methionine conditions. Heme modulates activity by shifting PLP tautomerism to inhibit the enzyme under oxidative stress, while H₂S production acts as a gasotransmitter regulating vasodilation, neuromodulation, and cytoprotection through sulfhydration of targets such as K(ATP) channels and protein kinases. The enzyme's tetrameric assembly positions regulatory CBS domains away from the catalytic core, transmitting conformational changes that reposition active site residues such as lysine and arginine to stabilize zwitterionic intermediates during β-replacement. CBS deficiency causes homocystinuria, leading to hyperhomocysteinemia, thromboembolism, lens dislocation, skeletal abnormalities, and cognitive impairment due to toxic homocysteine accumulation and impaired H₂S and glutathione synthesis. Pathogenic mutations often disrupt tetramerization, heme or PLP binding, or AdoMet responsiveness. |
| References |
|---|
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.