research use only
AMPKγ1 Antibody [M8F13]
Cat.No.: F2347
Application:
Reactivity:
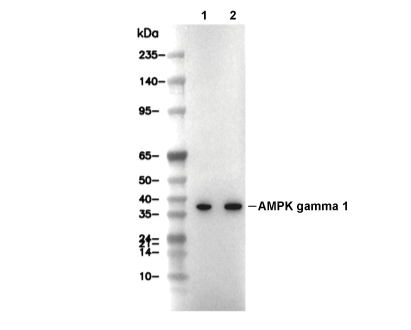
Experiment Essentials
WB
Recommended wet transfer conditions: 200 mA, 60 min.
Recommended wet transfer conditions: 200 mA, 60 min.
Usage Information
| Dilution |
|---|
|
| Application |
|---|
| WB, IP, FCM |
| Reactivity |
|---|
| Human |
| Source |
|---|
| Rabbit Monoclonal Antibody |
| Storage Buffer |
|---|
| PBS, pH 7.2+50% Glycerol+0.05% BSA+0.01% NaN3 |
| Storage (from the date of receipt) |
|---|
| -20°C (avoid freeze-thaw cycles), 2 years |
| Predicted MW Observed MW |
|---|
| 38 kDa 38 kDa |
| *Why do the predicted and actual molecular weights differ? The following reasons may explain differences between the predicted and actual protein molecular weight. Post-translational modifications(e.g., phosphorylation, glycosylation); Splice variants and isoforms; Relative charge; Multimerization. |
| Positive Control | HEK293 cells, Jurkat cells, HeLa cells |
|---|---|
| Negative Control |
Experimental Methods
| WB |
|---|
Experimental Protocol:
Sample preparation
1. Tissue: Lyse the tissue sample by adding an appropriate volume of ice-cold RIPA/NP-40 Lysis Buffer (containing Protease Inhibitor Cocktail),and homogenize the tissue at a low temperature. 2. Adherent cell: Aspirate the culture medium and wash the cells with ice-cold PBS twice. Lyse the cells by adding an appropriate volume of RIPA/NP-40 Lysis Buffer (containing Protease Inhibitor Cocktail) and put the sample on ice for 5 min. 3. Suspension cell: Transfer the culture medium to a pre-cooled centrifuge tube. Centrifuge and aspirate the supernatant. Wash the cells with ice-cold PBS twice. Lyse the cells by adding an appropriate volume of RIPA/NP-40 Lysis Buffer (containing Protease Inhibitor Cocktail) and put the sample on ice for 5 min. 4. Place the lysate into a pre-cooled microcentrifuge tube. Centrifuge at 4°C for 15 min. Collect the supernatant;
5. Remove a small volume of lysate to determine the protein concentration;
6. Combine the lysate with protein loading buffer. Boil 20 µL sample under 95-100°C for 5 min. Centrifuge for 5 min after cool down on ice.
2. Power up 80V for 30 minutes. Then the power supply is adjusted (110 V~150 V), the Marker is observed, and the electrophoresis can be stopped when the indicator band of the predyed protein Marker where the protein is located is properly separated. (Note that the current should not be too large when electrophoresis, too large current (more than 150 mA) will cause the temperature to rise, affecting the result of running glue. If high currents cannot be avoided, an ice bath can be used to cool the bath.)
Transfer membrane
1. Take out the converter, soak the clip and consumables in the pre-cooled converter;
2. Activate PVDF membrane with methanol for 1 min and rinse with transfer buffer;
3. Install it in the order of "black edge of clip - sponge - filter paper - filter paper - glue -PVDF membrane - filter paper - filter paper - sponge - white edge of clip"; 4. The protein was electrotransferred to PVDF membrane. ( 0.45 µm PVDF membrane is recommended ) Reference Table for Selecting PVDF Membrane Pore Size Specifications Recommended conditions for wet transfer: 200 mA, 60 min. ( Note that the transfer conditions can be adjusted according to the protein size. For high-molecular-weight proteins, a higher current and longer transfer time are recommended. However, ensure that the transfer tank remains at a low temperature to prevent gel melting.)
Block
1. After electrotransfer, wash the film with TBST at room temperature for 5 minutes;
2. Incubate the film in the blocking solution for 1 hour at room temperature;
3. Wash the film with TBST for 3 times, 5 minutes each time.
Antibody incubation
1. Use 5% skim milk powder to prepare the primary antibody working liquid (recommended dilution ratio for primary antibody 1:1000), gently shake and incubate with the film at 4°C overnight; 2. Wash the film with TBST 3 times, 5 minutes each time;
3. Add the secondary antibody to the blocking solution and incubate with the film gently at room temperature for 1 hour;
4. After incubation, wash the film with TBST 3 times for 5 minutes each time.
Antibody staining
1. Add the prepared ECL luminescent substrate (or select other color developing substrate according to the second antibody) and mix evenly;
2. Incubate with the film for 1 minute, remove excess substrate (keep the film moist), wrap with plastic film, and expose in the imaging system. |
Biological Description
| Specificity |
|---|
| AMPKγ1 Antibody [M8F13] detects endogenous levels of total AMPKγ1 protein. |
| Uniprot ID |
|---|
| P54619 |
| Clone |
|---|
| M8F13 |
| Synonym(s) |
|---|
| 5'-AMP-activated protein kinase subunit gamma-1; AMPK gamma1; AMPK subunit gamma-1; AMPKg; PRKAG1 |
| Background |
|---|
| AMPKγ1, also known as PRKAG1, is the regulatory gamma subunit of AMP-activated protein kinase (AMPK), a heterotrimeric energy sensor complex composed of alpha catalytic and beta scaffold subunits. This protein contains three canonical cystathionine-β-synthase (CBS) domains, which pair to form four adenine nucleotide-binding sites. The first CBS domain houses a binding site with key arginine and histidine residues that are critical for AMP and ATP coordination. An N-terminal extension and the second CBS domain link the structure, while a C-terminal tail helps stabilize the assembly of the heterotrimer. When AMP binds to its primary site, AMPKγ1 undergoes conformational changes that expose the activation loop (T-loop) on the alpha subunit, allowing phosphorylation by upstream kinases such as LKB1 and CaMKKβ. This modification amplifies AMPK’s kinase activity dramatically and protects it from dephosphorylation by protein phosphatases. ADP binding at alternative sites provides moderate activation, whereas ATP binding inhibits the enzyme through intrasteric occlusion. Activated AMPK phosphorylates key metabolic targets, including ACC and HMGCR, to suppress lipid and cholesterol synthesis, and TSC2 to limit anabolic processes. It simultaneously promotes catabolic pathways by activating PGC1α and ULK1 for mitochondrial biogenesis and autophagy, increasing glucose uptake via GLUT4 translocation, and restraining gluconeogenesis through FOXO signaling. AMPK also influences cell polarity by remodeling actin structures through phosphorylation of myosin light chain kinase and cofilin. Isoform specificity is notable: the γ1 subunit is predominant in hepatocytes and the liver, where it is crucial for mediating the effects of metformin, particularly through its CBS domains. Loss of γ1 abolishes activation loop phosphorylation and disrupts glucose-suppressing effects. In animal models, the absence of γ1 impairs the anti-hyperglycemic action of metformin under high-fat diets. Mutations that disrupt AMP binding, such as substitutions at critical arginine residues, can mimic cardiac pathologies seen with other AMPK gamma isoforms, including Wolff-Parkinson-White syndrome. In cancer, tumor cells may take advantage of constitutive AMPK activity for survival under metabolic stress, while silencing γ1 impairs proliferation in such conditions. Defective hepatic γ1 function in diabetes leads to elevated glucose output and impaired metabolic regulation. |
| References |
|---|
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.